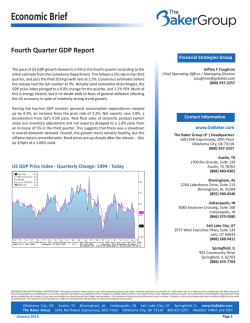
Hemoglobin Birmingham and Hemoglobin Galicia: Two
From www.bloodjournal.org by guest on February 6, 2015. For personal use only. Hemoglobin Birmingham and Hemoglobin Galicia: Two Unstable B Chain Variants Characterized by Small Deletions and Insertions By J.B. Wilson, B.B. Webber, H. Hu, A. Kutlar, F. Kutlar, J.F. Codrington, J.T. Prchal, K.M. Hall, J.M!. de Pablos, I. Rodriguez, and T.M.J. Huisman Two unstable hemoglobins (Hbs) causing rather severe hemolytic anemia have been characterized. The B chain of Hb Birmingham, found in an adult black man, is characterized by the loss of -Leu-Ala-His-Lys- at positions 141,142, 143, and 144 and their replacement by one Gln residue. These changes are the result of a deletion of nine nucleotides, namely two base pairs (bp) of codon 141, all of codons 142 and 143. and one bp of codon 144; the remaining CAG triplet (C from codon 141 and AG from codon 144) codas for the inserted glutamine. In the B chain of Hb Galicia from a Spanish patient, His and Val at D ELETIONS OF one to five amino acid residues in either the a or the j3 chain of human hemoglobin (Hb) cause a change in the stability of the protein and a mild to severe hemolytic anemia. Two deletions in the a chain and 12 such mutations in the @ chain have been reported; these are listed' and many are discussed in detail by Bunn and Forget.' The abnormal Hbs are considered to be the result of small deletions in the coding sequences of the appropriate chains; in one case ( H b Natal) it is caused by a base pair (bp) substitution leading to a stop codon at the penultimate codon of the a2-globin gene,3 and in a second case (Hb McKees Rocks) by an analogous substitution in the @-globin gene.' Here we describe two additional unstable @ chain variants caused by deletions of nine and three nucleotides from coding sequences of the @-globin gene; the observation is of some special interest because these specific deletions result in the insertion of one amino acid residue in addition to the deletion of four and two amino acid residues, respectively. MATERIALS AND METHODS Blood samples were collected in vacutainers with EDTA as anticoagulant and sent in ice by express mail to Augusta, GA. Informed consent was obtained. Hematologic values were determined with an automated cell counter. Hb analyses included isoelectrofocusing (IEF), heat and isopropanol stability tests, and cation and reversed phase high performance liquid chromatography (HPLC); methodology was as described before?.' Structural analyses were by methods routinely in use in our laborat~ries.~,~ The abnormal ,9 chain (,9' or fly) was separated from the (Y chain by CM-cellulose chromatography' and digested with trypsin for 6 hours at room temperature and at pH 8.5. The resulting peptides were separated by reversed phase HPLC,8 and some were rechromatographed by a similar procedure but with a different developing system.' Amino acid analyses were made in 24-hour acid hydrolysates with the Waters Pico Tag amino acid analyzer (Waters Chromatography Division, Milford, MA) (see manufacturer's manual). DNA was isolated by the method of Poncz et a1."' Appropriate segments of genomic DNA were amplified with Taq polymerase as described before"; the nucleotide deletions,inferred from the protein data, were verified by dot-blot analysis with 32P-labeledprobes containing sequences listed elsewhere in this report. The methodology has been detailed before." Blood, Vol 75, No 9 (May 11, 1990: pp 1883-1 887 positions97 and 98 are replaced by one Leu residue. This is due to an ACG deletion in codons 97 and 98, which causes the removal of one His and one Val residue, while the remaining CTG triplet (C from codon 97 and TG from codon 98) codes for the inserted leucine residue. Two mechanisms, namely slipped mispairing in the presence of short repeats, and misreading by DNA polymerase due to a local distortion of the DNA helix, are considered in explaining the origin of the small deletions. 0 1990 b y The American S o c i e t y of Hematology. RESULTS Cuse Report. Cuse 1. H b Birmingham or a2&( [Leu-AlaHis-Lys]+Gln at positions 141 through 144). The propositus is a 27-year-old Black man (A.J.) who was referred to a hematologist for evaluation of suspected lymphoma because of splenomegaly, anemia, and thrombocytopenia (1 10 x 10I2/ L). Further evaluation showed that the anemia was hemolytic with reticulocytosis ranging from 6% to 12%, Coombs negative. The subject was referred to the Special Red Cell and Hemoglobinopathy Laboratory at the University of Alabama at Birmingham (UAB) for red cell enzyme studies. The evaluation of the blood film showed numerous Heinz bodies. Consequently, unstable H b studies were performed; the isopropanol screen was positive and a heat test for unstable H b showed a precipitate (about 20%). Electrophoretic evaluation of a freshly drawn blood sample showed a mutant H b with a migration identical to that of H b S; however, on citrate agar electrophoresis (pH 6), H b X did not separate from H b A. On the globin electrophoresis at pH 8.4 and pH 6, a band anodic to PAwas observed. The metHb level was 0.9%; sulfH'b, however, was increased a t 2.45%.12 From the Department of Cell and Molecular Biology. Medical College of Georgia, Augusta; Division of Hematology and Oncology, University of Alabama at Birmingham and the Veterans Administration Hospital, and the Special Red Cell and Hemoglobinopathy Laboratory, Birmingham: Servicio de Hematologia, Hospital General de Especialidades, Granada; and Servicio de Hematologia, Hospital Jeran Canalejo, La Coruna, Spain. Submitted November 6,1989; accepted January 12.1990. Supported by National Institutes of Health Research Grants HLB-05168 and HLB-41544 (to T.H.J.H.)and by support of the Veterans Administration IFCAP/2237 (to J. T.P.). This is contribution no. 1193 from the Department of Cell and Molecular Biology at the Medical College of Georgia, Augusta. Address reprint requests to Titus H.J. Huisman, PhD, Department of Cell and Molecular Biology, Medical College of Georgia, Augusta, GA 3091 2-21 00. The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked "advertisement" in accordance with 18 U.S.C.section 1734 solely to indicate this fact. 6 I990 by The American Society of Hematology. 0OO6-4971/90/7509-0012$3.OO/0 1883 From www.bloodjournal.org by guest on February 6, 2015. For personal use only. WILSON ET AL 1084 Estimation of the P,, from venous blood gasesi3 showed a Birmingham after the area of family residence and original evaluation. slightly decreased level at 22.0 mm Hg (normal range 22.6 to 29.4). The erythrocyte 2,3-DPG leveli4 was in the normal Case Report. Case 2. H b Galicia or a,&([His-Val]-Leu at positions 97 and 98). This variant was present in a young range of 12,270 5 1,870 nmol/g Hb). A somewhat variable pattern was noted by reversed phase HPLC (Fig 1); two or Spanish patient (0.0.)with a hemolytic anemia, hepatothree abnormal ' 0 zones eluted ahead of the normal /3* peak. splenomegaly, and a history of jaundice. Hematologic findH b F was slightly elevated at 3.5%, which was confirmed by ings for a blood sample that had been in transit for 5 days the presence of small amounts of 'y and Aychain in the HPL were as follows: H b 7.5 g/dL; PCV 0.290 1/L; RBC 2.22 x lO"/L; MCV 131 fL;M C H 33.8 pg; MCHC 26.0 g/dL; chromatogram (Fig 1). these data indicated a rather severe anemia. The reticulocyte The unstable H b was isolated by preparative isopropanol count in a freshly collected sample was 25%; total bilirubin precipitation (20 mL 17% isopropanol in 0.2 mol/L TrisHCI, pH 7.4, mixed with 2 mL red cell lysate and incubated 1.94 mg/ 100 mL; indirect bilirubin 1.08 mg/ 100 mL. Heinz bodies were positive (2 hours at 37OC). A slow band was for 30 minutes at 37OC). The fix chain was isolated by detectable by IEF, which was slightly faster than Hb A,. CM-cellulose chromatography and digested with trypsin. Fig Heat stability and isopropanol tests were positive. Reversed 2 (top) illustrates the separation of the tryptic peptides. Most peptides eluted at their expected positions and had normal phase HPLC (Fig 1) detected a distinct zone that eluted amino acid compositions (data not shown). The peptides behind the a chain; its quantity (as percent of 6" + BY + y) T-14 and T-15 were absent, while a combination of these two was 26.7%. H b F was elevated; its total quantity (as fragments (T-14',15) eluted together with peptide T-3. The 'y + Ay)was 10.3%. two peptides were easily separated by rechromatography The variant was isolated by DEAE-cellulose chromatogra(Fig 2, top insert). The amino acid composition of the 0') was studied by the phy and the abnormal 0 chain ( methods described above for the 6" of H b Birmingham. T-14',15 fragment was Asp 0.99(1); Glu 1.12(0); Gly Figure 2 (bottom) illustrates an HPL chromatogram of the 0.98(1); His 0.92(2); Ala 3.02(4); Tyr 0.82(1); Val 2.15(3); tryptic peptides of the fly chain. All soluble peptides were Leu O( 1); Lys O( 1). Expected values are between parentheses; present and had the expected compositions (data not shown). the valine value was increased to nearly three residues when a 72-hour acid hydrolysate was analyzed. These results sugAn extra zone eluted in front of T-13; its amino acid gested a loss of one histidine, one alanine, one leucine, and composition was Asp 2.08(2); Glu l.Ol(1); His O(1); Arg one lysine residue and the addition of one glutamic acid or 0.93(1); Pro l.OO(1); Val O(1); Leu 1.91(1); Phe 0.94(1). This peptide resembled PT-11, which is normally not recovglutamine residue. Sequence analyses gave the following ered in notable quantities from a digest of a nonaminoethsequence: Val-Val-Ala-Gly-Val-Ala-Asn-Ala-Gln-Tyr-His, confirming the deletion of Leu-Ala-His-Lys at positions 141, ylated /3 chain. The sequence of this T-11' peptide, Leu-LeuAsp-Pro-Glu-Asn-Phe-Arg, confirmed a deletion of His-Val 142, 143, and 144 and the substitution of one glutamine at positions 97 and 98 of the 0 chain and the insertion of an residue. extra leucine residue. The variant was named H b Galicia; The patient has two sons and a living mother; none were family studies were not conducted. available for the study. However, electrophoretic studies were performed on the propositus' parents in routine laboraDISCUSSION tories in the past; the father was said to have H b A and the Each of the two H b variants described here is charactermother was told she had sickle cell disease with only H b S ized by a deletion and an insertion and by an instability that and Hb F detected; however, she has never had a clinical causes a rather severe hemolytic anemia. It was not possible history of sickle cell crisis or other stigmata of sickle cell to determine whether one or both anomalies were the result disease. She has been known to be anemic with increased of de novo genetic events because family studies could not be reticulocyte counts, The abnormal variant was named H b E Heme Heme PATIENT 0.0. t h 0 Jb cu cu (63.0%) a -4- cu a, 0 C ea (26.7%) 8 (6.8%) a 10 20 30 40 50 60 70 I 10 I 20 Time in Minutes (1 ml/min) I 30 I 40 I 50 Av (3.5%) I 60 I 70 Fig 1. Separation of globin chains by reversed phase HPLC with a Vydac C, column (The SeplAIRations Group. Hesperia, CA)."," (Left) Patient A.J. with the &Birmingham chain (@"I. (Right) Patient 0.0. with the 8-Galicia chain (By). The gradients used in the two chromatograms were slightly different. which explains the differences in the elution times. From www.bloodjournal.org by guest on February 6, 2015. For personal use only. khS BIRMINGHAM AND GALlClA 1886 - Fb2. 8.pmknd.dubk p.ptLb..*"dig.md abnormal B ctuinr by r w u n d phew W L C with C,,cdumn.' (Top) Potimt A.J. with tho 8-8irmingh.m c h i n WV. Tho Inoort illustrotos tho ..p.mtkn of 8 1 - 3 and BT-14'.16 h, roporoto orpuimont. IBottom) P~tkm 0.0.with tho B - a l k h c h i n (L?'). Time in Minutes (1.5 ml/min) conducted. However. in the case of Hb Birmingham. it is almost certain that the mutant was inherited from the mother whose phenotype was HbS-Hb Birmingham. Figure 3 provides additional details about the deletions responsible for the formation of the two unstable 6 chains. Patient A.J. with Hb Birmingham had the deletion of four residues (Leu-Ala-His-Lys) and the insertion of a glutamine residue at positions 141 through 144 of the 6 chain. This change is explained by the deletion of nine nucleotides as indicated in Fig 3: the C of codon 141 and the AG of codon 144 combine to code for a glutamine residue. Further proof -*Q) cooo( was obtained from hybridization experiments with specific synthetic oligonucleotide p r o k , only the amplified DNA of patient A.J. hybridized with the 9-bpdeletion probe (Fig 4). The data for patient 0.0. arc comparable (Fig 3). The replacement of a His-Val dipeptide at positions 97 and 98 by a leucine residue results from the deletion of three nucle- 139 140 141 142 143 144 1 4 s 146 .ASN.ALA.LEU.ALA.HlS.LYSIYR.nlS.IER .AAI.OCC.CIO~OCC~CAC.AAO~TAI.CAC.TAA o(LcIID:- 139 140 14s IR.mA J ) W rcD .ASN*ALA-OLN coooN -*Q) . A I 1 .OCC-CAO-----TAT 140 TVR.HlS.TER . C A C * TAA 91 05 00 07 00 89 1 0 0 101 .ASP.LVS.LEU.HlS.VALASP.PRO.OLU. coooN .OAC.AAO.ClO.CAC.OIO.OAT.CCT.OAO. Qur(D:- (Rrr 0 0 )W IcD coooN Flg3. 9 1 0 5 0 0 89 loo 101 . A S P . L V S . LEV-LEU-ASP*PRO.OLU~ .01C~AAO~C10-CTO-O~T~CCT.OAO. pWadth.nornrlmd.bnanr(.mlno#IdmdoNA ~ d ~ h u r w r , B d u lTh.d.(.tkodn(m n . nuckocid.. (TQGCCCACAI 0k.rmd in pnkm A.J. k.dr to t h d.kckn ~ d L ~ - A ) . - H h - L ~ . t p 0 & t i 0 ~ 1 4 1* h r o u g h l U m d t h . m k n o t ~ Qln. t8ottom) Tho d.ktkn of ACQ in potkm 0.0. u u... tho d.ktkn of Hh-V.1 .t pooitkm97 and 90. and th.hloutkn o f 0 LW. Dar-bkr~dmpl(kdoNAwlthPP#.kd fh. antiro B - u W n 0.m horn ch. -4. s"th .- promotor r o g h 10 tho -ban Jgcw( woo wnp(l(kd. Hybridtntbn w n w t t h prok.dur.ctwhtk tor th.Brwd.ot(d. d.kckn (oxpukrrmt I) 0. tor tho 3-nud.otld.d.kt&n (0rp.r)mw 11). Probow Expt.1 " k 6' mvunt:C ~rpt.w"k 6' w o n t : 6' CCTaacccAcMoTATCAC Y QQCTMTQCCC AOTATCAC Y AAQCTQCACOTOOATCCTO S' OACAAQCTOC-TOQATCCTO S' oNA from pmhta A.J. nd 0.0. md from lour nu)y..d. -- From www.bloodjournal.org by guest on February 6, 2015. For personal use only. WILSON ET AL 1886 otides (ACG); the remaining nucleotides form codon CTG, which codes for Leu. Data from hybridization experiments with specific probes indicate that amplified D N A from patient 0.0.hybridizes to the probe containing this deletion (Fig 4). As many as 11 different H b types have been described that are characterized by the deletion of 1 (6 variants), 2 (2 variants), 3 (1 variant), 4 (1 variant), or 5 (1 variant) amino acid residues in the (3 chain.’ None of these seems to have an insertion’af an amino acid residue as do the two (3 chain variants described here. There are at least two different mechanisms that may account for the origin of variants with deleted residues. One is based on data by Farabaugh et al” and Albertini et all8 who studied spontaneously occurring small deletions in the lac I gene of Escherichia coli, and on considerations by Efstratiadis et all9 concerning deletions occurring in the human globin gene family. Small deletions apparently can result from slippage and mispairing of two homologous D N A sequences during DNA replication. This removes the DNA between these sequences as well as one copy of the repeated sequence. This slipped mispairing mechanism may readily account for the deletion of Leu-His-Cys-Asp-Lys at (391-95 in the (3 chain of H b Gun Hill,” of Phe-Glu-Ser a t (343-45 in the B chain of Hb Niteroi,2’of the seven nucleotides causing a p-thalassemia in a Turkish family,22and of four nucleotides in the Ay-globin gene p r ~ m o t e r . ~When ’ the same rules are applied to the deletions causing the variant (3 chains described here, only the one present in patient A.J. can be partially explained by this mechanism. As indicated in Fig 3, a repeat of GCCC is observed, and slipped mispairing would account for the loss of six nucleotides; ie, one GCCC sequence and the T G dinucleotide located between the repeated sequences. The additional loss of the ACA trinucleotide is difficult to explain; it is to be noted that loss of ACA has also been observed a t codon (387 in H b Tours, which results in the deletion of a Thr residue in the (3 chain of this variant.24 The ACG deletion present in the (3 gene of patient 0.0. can also not readily be explained. This small deletion is at the edge of the eight nucleotide sequence (AGCTGCAC; Fig 3) that is repeated a t codons 90,91, and 92, and may be used to account for the 15-nucleotide H b Gun Hill deletion. Perhaps the ACG deletion is the result of a local distortion of the D N A helix and misreading by D N A polymerase leading to the deletion of the three nucleotides. This mechanism has been considered as an explanation for the occurrence of small deletions leading to frameshifts. It is also possible that the deletion of ACG is the direct result of mispairing of these closely related sequences. For instance, the 11-nucleotide sequence AG-CTG-CAC-GTC- in codons 95 (two nucleotides), 96, 97, 98 is similar to AG-CTG-CAC-TGT-G in codons 90 (2 nucleotides), 91, 92, 93, 94 (1 nucleotide) except for the first T in codon 93. When these sequences are mispaired, this T might remain unpaired and interfere with the pairing between the ACG sequence in codons 97 and 98 and the ACG in codons 92 and 93; this could result in the deletion of ACG from codons 97 and 98 and in the observed abnormal (3 chain sequence. ACKNOWLEDGMENT We thank Dr T.A. Stoming for providing the primers and probes. REFERENCES 1. The International Hemoglobin Information Center Variant List. Hemoglobin 13:223, 1989 2. Bunn HF, Forget BG: Hemoglobin: Molecular, Genetic and Clinical Aspects. Philadelphia, PA, Saunders, 1986 3. Jogessar VB, Westermeyer K, Webber BB, Wilson JB, Hu H, Gonzalez-Redondo JM, Kutlar A, Huisman THJ: Hb Natal or a2 (minus Tyr-Arg)& a high oxygen affinity a chain variant with a deleted carboxy-terminus resulting from a TAC-TAA (Tyrterminating codon) mutation in codon a140. Biochim Biophys Acta hemoglobins chains by high-performanceliquid chromatography. J Chromatogr 179:271,1979 9. Schroeder WA: High performance liquid chromatography used in structural analyses of hemoglobin variants, in Huisman THJ (ed): The Hemoglobinopathies, Methods in Hematology Series, vol 15. Edinburgh, UK, Churchill Livingstone, 1986, p 142 10. Poncz M, Solowiejczyk D, Harpel B, Mory Y,Schwartz E, Surrey S: Constructionof human gene libraries from small amounts of peripheral blood: Analysis of @-likeglobin genes. Hemoglobin 951:36, 1988 4. Huisman THJ: Introduction and review of standard methodol- 6:27, 1982 11. Gonzalez-Redondo JM, Stoming TA, Lanclos KD, Gu YC, ogy for the detection of hemoglobin abnormalities,in Huisman THJ (ed): The Hemoglobinopathies, Methods in Hematology Series, vol 15. Edinburgh, UK, Churchill Livingstone, 1986, p 32 5. Huisman THJ: Separation of hemoglobins and hemoglobin chains by high-performanceliquid chromatography. J Chromatogr Kutlar A, Kutlar F, Nakatsuji T, Deng B, Han IS, McKie VC, Huisman THJ: Clinical and genetic heterogeneity in black patients with homozygous &thalassemia from the Southeastern United States. Blood 72:1007,1988 12. Evelyn KA, Malloy HT: Microdeterminationof oxyhemoglobin, methemoglobin, and sulfhemoglobin in a single sample of blood. J Biol Chem 126:655,1938 13. Lichtman MA, Murphy MS, Adamson JW: Detection of mutant hemoglobins with altered affinity for oxygen. Ann Intern Med 84517.1976 14. Beutler E: Metabolism: A Manual of Biochemical Methods (ed 2). Orlando, FL, Grune and Stratton, 1975 15. Shelton JB, Shelton JR, Schroeder W A High performance liquid chromatographic separation of globin chains on a large-pore C, column. J Liquid Chromatogr 7:1969, 1984 16. Kutlar F, Kutlar A, Huisman THJ: Separation of normal and 418:277, 1987 6. Wilson JB, Chen SS, Webber BB, Kutlar A, Kutlar F, Villegas A, Huisman THJ: The identification of five rare 6-chain abnormal hemoglobins by high performance liquid chromatographic procedures. Hemoglobin 10:49, 1986 7. Clegg JB, Naughton MA, Weatherall DJ: Abnormal human haemoglobins. Separation and characterization of the a and 6 chains by chromatography,and the determination of two new variants, Hb Chesapeakeand Hb J (Bangkok).J Mol Biol 19:91,1966 8. Wilson JB, Lam H, Pravatmuang P, Huisman THJ: Separation of tryptic peptides of normal and abnormal a, 0, y, and 6 From www.bloodjournal.org by guest on February 6, 2015. For personal use only. Hes BIRMINGHAM AND GALlClA abnormal hemoglobin chains by reversed-phase high-performance liquid chromatography. J Chromatogr 357:147,1986 17. Farabaugh PJ, Schmeissner U,Hofer M, Miller JH: Genetic studies of the lac repressor. VII. On the molecular nature of spontaneous hotspots of the lac1 gene of Eschericia coli. J Mol Biol 126:847, 1978 18. Albertini AM, Hofer M, Calm MP, Miller JH: On the formation of spontaneous deletions: The importance of short sequence homologies in the generation of large deletions. Cell 29:3 19, 1982 19. Efstratiadis A, Posakony JW, Maniatis T, Lawn RM, OConneil C, Spritz RA, DeRiel JK, Forget BG, Weissman SM, Slightom JL, Blechl AE, Smithies 0, Baralle FE, Shoulders CC, Proudfoot NJ: The structure and evolution of the human &globin gene family. Cell 21:653, 1980 20. Bradley TB, Wohl RC, Rieder RF: Hemoglobin Gun Hill: 1887 Deletion of five amino acid residues and impaired heme-globin binding. Science 157:1581, 1967 21. Praxedes H, Lehmann H: Haemoglobin Niteroi-A new unstable variant. Proc 14th Int Congr Hematol, Sau Paulo, Brazil, 1972 22. Schnee J, Griese E-U, Eigel A, Horst J: @-Thalassemiagene analysis in a Turkish family reveals a 7 bp deletion in the coding region. Blood 73:2224, 1989 23. Gilman JG, Johnson ME, Mishima N: Four base-pair DNA deletion in human Ay-globingene promoter associated with low Ay expression in adults. Br J Haematol68:455, 1988 24. Wajcman H, Labie D, Schapira G: Two new hemoglobin variants with deletion. Hemoglobin tours: Thr @87(F3) deleted and Hemoglobin St Antoine: Gly-Leu 074-75 (E18-19) deleted. Consequences for oxygen affinity and protein stability. Biochim Biophys Acta 295:495, 1973 From www.bloodjournal.org by guest on February 6, 2015. For personal use only. 1990 75: 1883-1887 Hemoglobin Birmingham and hemoglobin Galicia: two unstable beta chain variants characterized by small deletions and insertions JB Wilson, BB Webber, H Hu, A Kutlar, F Kutlar, JF Codrington, JT Prchal, KM Hall, JM de Pablos and I Rodriguez Updated information and services can be found at: http://www.bloodjournal.org/content/75/9/1883.full.html Articles on similar topics can be found in the following Blood collections Information about reproducing this article in parts or in its entirety may be found online at: http://www.bloodjournal.org/site/misc/rights.xhtml#repub_requests Information about ordering reprints may be found online at: http://www.bloodjournal.org/site/misc/rights.xhtml#reprints Information about subscriptions and ASH membership may be found online at: http://www.bloodjournal.org/site/subscriptions/index.xhtml Blood (print ISSN 0006-4971, online ISSN 1528-0020), is published weekly by the American Society of Hematology, 2021 L St, NW, Suite 900, Washington DC 20036. Copyright 2011 by The American Society of Hematology; all rights reserved.
© Copyright 2026