Erythrocytes by Dilauroylphosphatidylcholine-Induced
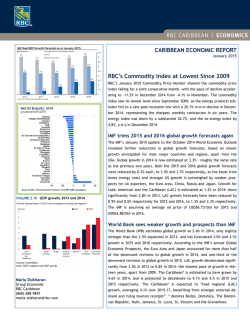
From www.bloodjournal.org by guest on February 6, 2015. For personal use only. Generation of Phenotypically Aged Phosphatidylserine-Expressing Erythrocytes by Dilauroylphosphatidylcholine-InducedVesiculation By Cecilia Diaz, Jerzy Morkowski, and Alan J. Schroit In vitro stored red blood cells (RBC) and RBC artificially induced to vesiculate by incubation with dilauroylphosphatidyl-choline were monitored for age- and vesiculation-dependent changes in celldensity, membrane lipid asymmetry, and their ability to berecognized and cleared by reticuloendothelial cells. RBC demonstrated a progressive increase in density on self-forming Percoll gradients upon vesiculation and in vitro "aging." Uptake of vesiculated RBC by in vitro cultivated macrophages was increased threefold over nonvesiculated control RBC. The clearance rate of dense vesiculated RBC was biphasic and contained a rapid component and a slower second component consistent with the clear- ance rates of normal control populations. Determination of phosphatidylserine (PS) in the outer leaflet ofRBC by the PS-dependent prothrombinase assay revealed that PS redistributed to thecell's outer leaflet upon in vitro storage and vesiculation. Inhibition of PS movement by oxidation of membrane sulfhydryls with pyridyldithioethylamine resulted in higher prothrombinase levels and enhanced.clearance of vesiculated RBC. These experiments suggest that vesiculation contributes to alterations in membrane lipid asymmetry and cell density characteristic of the aged RBC phenotype. 0 1996 by The American Society of Hematology. T Denselold RBC also exhibit significant reductions in volume/surface area that can be explained by decreased electrolyte content associated with osmotic water loss and loss of membraneandcytoplasm by microvesiculation.' '.'i~z4 Because spontaneousformation of vesicles occurs in vivo'5 and in i n vitro stored RBC,'"'' we hypothesized that vesiculation could. at feast i n part, be responsible for the age-dependent alterations in membrane lipid asymmetry associated with macrophage recognition. To test this hypothesis, RBC were artificially vcsiculated") with dilauroylphosphatidylcholine (DLPC) and assessed for alterations in density, membrane lipid asymmetry, and their propensity to be recognized by macrophages in vitro and the RES in vivo. HE MECHANISM(S) responsible for the recognition of aged red blood cells (RBC) by the reticuloendothelial system (RES) that results in their elimination from the circulation are not completely understood. Various moieties have been shown to play a role in the determination of the aged phenotype. These include several normal RBC components that have undergone specific modifications such as loss of carbohydrates that result in decreased surface charge'.' and the appearance of asialoglycophorin,' irreversible oxidative damage causing recognition of glycophorin A: proteolytic 3,"'' and formation of "senescent cell antigen" from band loss of plasma membrane phospholipid asymmetry resulting in the progressive appearance of phosphatidylserine (PS) at the cellsouter leaflet." Because theseprocesses are not mutually exclusive, the mechanisms underlying phagocytic recognition could be complex involving multiple receptorligand interactions. These could include the Fc receptor for antibody-mediated recognition.'.' VCAM- I/vitronectin receptor for VLA-4 expressing apoptotic cells""" and differentiating erythroblasts," scavenger receptors," oxidized LDL receptors.""" and the putative PS receptor.' i.'""" Because there seems to be a direct relationship between RBC density and age,'"." most studies o n erythrocyte aging have been bdsed on comparisons between density-separated RBC populations. Using self-forming Percoll gradients, we have recently shown direct a relationshipbetween the amounts of PS exposed on the surface of RBC, their ahility to transport lipids between membrane leaflets, and their propensity to be bound by macrophages in vitro and cleared by the RES in vivo.' ___ " " Fronl the Departrrrent of' Cell Biology, Tire Utlirc,r.\in; of Textrs M.D.Anderson Cancer Cerfter. Houston. Suhnritted June 7, 199.5: uccepted Novewher 7, 1995. S~cpportedin part by Nutiond fnstitutes of Health Grunt No. D K 41714. Address reprint requests to Alun J . Schroit, PhD, Department o f CellBiology, B M 173, TheUniver.sity of Texas M.D. Anderson C a m e r Center, 1515 Holcombe Blvd, Houston, TX 77030. The publication costs of this article were defrayedin parr by puge churgepuyment.Thisarticlemustthereforebeherebymarked "advertisement" i n accordance with I8 U.S.C. section 1734 solely t o indtcate this fact. 0 I996 by The American Socieiy of Hematology. OUU~-4Y71/96/~707-0U47~3.00/0 2956 MATERIALS AND METHODS Muterirr1.r t r l d routine prowdure.\. Pyridyldithioethylamine as previously described.'" Factor V was (PDA)wassynthesized isolated from bovine plasma and activatedas previously described.'' Prothrombin and factor X were from Sigma (St Louis, MO) and the throlnbin-sensitivcchronwgen. S223X. was purchased from Kabi Laboratories (Franklin. OH), Percoll was obtained from Pharmacia (Uppsala. Sweden). KBC were collectcd into heparinized tubes by venipuncture frorn healthy volunteer5 or from the tail vein of mice. l h e blood was diluted with HEPES-saline buffer (144 Inrnol/L NaCI, I O mmol/L HEPES. I O mmol/L glucose, pH 7.3) and washed twice. Percoll was prepared by mixing 213.5 g of Percoll with 25 niL of 1Ox concentrated phosphate-buffered saline (PBS) andwater to 250 nlL. The pH was adjusted to 7.4 and the osmolarity to 310 to 320 nlOsmdkg. Radiation was monitored by scintillation counting using a Packard AutoGamma spectrometer (Packard, Downers Grove, IL). &,,.sit! w p m t ; [ m of KBC. RBC were separated by density on self-forming Pcl-coll gradientsessentially B'\ described by Lutr et al." Briefly. 0.5 tnL o f packed RBC were mixed with 9 mL of Percoll (320 InOsnI, pH 7.4) at room temperature. The suspension was then centrifuged at 38.700g for 20 minutes. RBC were obtained from the gradients by puncturing the bottom o f the tube and washed free of Percoll. Vc,.sicu/nrionof' RBC. Smallunilamellarvesicles were prepared by sonication of DLPC (1.5 mmol/L) in HEPES-saline buffer followed by centrifugation at 30,OOOg to remove metal fragments and largevesicles.WashedRBCwereresuspended in HEPES-saline to a 10% hematocrit and incubated with DLPC (38 pmoIL final concentration) at 4°C on an orbital shaker for 45 minutes. Remnant RBC were collected by centrifugation at 85Og for I O minutes. The renlnantcellswere then washed with 1% bovineserumalbumin (BSA) to remove residual DLPC from the cell membranes. ~ r o t l i r r ~ ~ ~ t hccc./ivit\' i r ~ t r . c~s.sa\'. ~~~ Vesiculated andnonvesiculated Blood, Vol 87, No 7 (April I), 1996: pp 2956-2961 From www.bloodjournal.org by guest on February 6, 2015. For personal use only. VESICULATION-INDUCED PS EXPRESSION ANDAGING RBC (2 X IO' cells) were incubated in 0.2 mLof prothrombinase assay buffer (1 35 mmol/L NaCI, 3 mmol/L CaCl,, I O mmol/L HEPES, pH 7.4) at 37°C for 3 minutes. A total of 0.05 mL of factor Va (60 nmol/L) and 0.1 mL of factor Xa (0.25 U) was then added. The suspension was incubated at 37°C for another 3 to 4 minutes after which 0.1 mL of prothrombin (0.1 U) was added. After 5 minutes, a 0.1-mL aliquot of the suspension was transferred to a cuvette containing I mL of TrisEDTA buffer ( I 75 mmol/L NaCI, 50 mmol/L Tris, 2 mmol/L EDTA, pH 7.9) to stop the production of thrombin. The thrombin-dependent chromogen, S2238, was added to the cuvettes (to 0.2 mmol/L), and the rate of chromophore formation was monitored at 405 nm with a Gilford Response Spectrophotometer using appropriate kinetic software. The initial rate of thrombin-dependent chromophore production, which is proportional to the amount of PS present on the cell was determined from the slope of theabsorbance curve. To quantify the amount of endogenous PS exposed on the surface of these cells, a standard curve of the rates of thrombin productionlchromophore formation was generated using PDA-treated RBC that contained known amounts of I-oleoyl-2-[N-(7-nitro-2,1,3-benzoxadiazol-4-yI)amino]caproyl] (NBD)labeled PS exposed at the cells's outer Thrombin-dependent chromophore production was directly proportional to the amount of PS present in the cell's outer leaflet and linear within the range of interest. In vitro binding of 'ZsI-labeledRBC. Macrophages were obtained from the peritoneal cavity of 8- to IO-week-old BalblC mice 4 days after an intraperitoneal injection of 3 mL of thioglycolate medium. The cells were collected and washed in serum-free RPMI-1640 medium. Cells (3X IO'lwell) were plated in 24-wellFalcon plastic culture plates (Becton Dickinson, Lincoln Park, NJ). The nonadherent cells were removed from the plates after several hours. RBC were labeled for I hour at 0°C with 0.25 mCi "'1 in the presence of one iodobead (Pierce Chemical CO, Rockford, IL). The iodinelabeled RBC were then vesiculated and added to the macrophage monolayers at a ratio of 10 RBChacrophage (-3 X 10" RBC) for the indicated intervals. The cultures were then washed with HEPESsaline and lysed with 0.1% Triton X-100. The lysate was collected and monitored for radiation. In vivo clearanceof "Cr-labeled RBC. Two milliliters of mouse RBC (50% hematocrit) were incubated with -0.5 mCi of "Cr for 45 minutes at 37°C and washed. The cells were vesiculated as described above and injected into the tail vein of syngeneic Balblc mice (0.2 mL; 25% hematocrit). Mice were bled at the indicated times and the fraction of "Cr-labeled RBC remaining in the circulationwas determined by scintillation counting. Livers and spleens were collected at theend of the experiment to determine the presence of cleared RBC. The same experiment was carried out with cells pretreated with PDA and PDNdithiothreitol and with Percoll-separated in vitro-aged cells. The data points were fitted using Slidewrite Plus (Advanced Graphics Software, Carlsbad, CA) curve-fitting routines. Initial rates were estimated from linear fits of data points within the first hour after injection. RESULTS Clearance of in vitro stored RBC. To determine if in vitro-stored RBCs exhibit density-dependent differences in their ability to be cleared in vivo, mouse RBC were stored at 4°C for 1 week, "Cr-labeled, separated into light and heavy fractions on self-forming Percoll gradients (Fig l ) , and injected into syngeneic mice. Light cells separated from freshly obtained blood were cleared with a Tln of -100 hours, whereas heavy cells from the same gradient were cleared with half times of about 40 hours (Fig 2). Percoll gradient centrifugation ofin vitro-stored cells showed a 2957 Fig 1. Percoll gradient separation of fresh and in vitro stored RBC. Murine RBC were collected into heparin and stored for l week at 4°C. These cells and freshly obtained RBC were mixed with Percoll and centrifuged at 38,7009 for 20 minutes at 20°C. (AI Fresh RBC; (B) in vitro stored RBC. Arrow shows released vesicles. prominent hemoglobin-containing band of vesicles at the top of the gradient and RBC that localized at densities greater than fresh RBC (Fig IB). In contrast to fresh cells, cells recovered from these fractions exhibited TIQclearance rates less than IO hours in a manner that was independent of cell density (Fig 2). The rates of PS-dependent thrombin production produced by the stored cells was also threefold faster than the rates obtained with fresh cells (not shown), suggesting that PS asymmetry is not rigourously maintained after prolonged in vitro storage. Effect of DLPC-induced vesiculation on RBC density and PS asymmetry. The results presented above raise the possibility that a relationship between membrane vesiculation and the appearance of the dense/aged phenotype with perturbed membrane lipid distribution exists. To test this, freshly obtained RBC were induced to vesiculate with DLPC andseparated on Percoll (Fig 3). Similar to the pattern obtained after in vitro aging, an overall shift in the distribution of the cells to heavier densities occurred. The shift in density was also dependent on the formation of microvesicles which accumulated near the top of the gradient (Fig 3B). From www.bloodjournal.org by guest on February 6, 2015. For personal use only. 2958 DIM, MORKOWSKI, AND SCHROIT n same kinetics observed for control, nonvesiculated RBC. Extrapolation of the calculated curves to the limit of percent remaining cells approaching zero (Fig 6 inset) yielded a lifespan of about 20 days for control cells andthe slow component of vesiculated cells compared with approximately 5 days for the fast component of vesiculated cells. Clearance of %"labeled RBC vesicles was also determined. The data in Fig 6B shows that RBC vesicles were immedi- A S A 0 W 0 A m 0 lo 0 I I 25 50 75 Time (hr) Fig 2. Clearance of Percoll-separated fresh and stored RBC. Top and bottom fractions of RBC separated by density on self-forming Percoll gradients (Fig 1) were isolated, washed, and labeled with "Cr. The labeled cell suspension was adjustedt o a 2546 hematocrit and0.2 mL was injected intravenously into syngeneic mice. At the indicated intervals minimal amounts of blood were collected from tailthe vein and the fraction of "Cr-labeled RBC remaining in the peripheralcirculation was determined by scintillation counting. Fresh RBC, top fraction (A), bottom fraction (0); in vitro-aged RBC, top fraction (A), bottom fraction (0). The effects of vesiculation on endogenous PS distribution was determined by the PS-dependent prothrombinase assay. Figure 4 shows that remnant DLPC-treated cells exhibited significantly higher levels ofPS at the cell's outer leaflet. PS content in the outer leaflet of the vesiculated RBC was higher when cells were treated with PDA, a known inhibitor of aminophospholipid transport. Upon the addition of dithiothreitol (DIT), the prothrombinase activity of the remnant cells returned to values similar to vesiculated, non-PDAtreated cells suggesting that a fraction of the PS exposed at the cell surface was transported back to the inner leaflet (Fig 4). In vitro recognition and clearance of vesiculated RBC. To determine whether RBC vesiculation results in their uptake by macrophages, '251-labeledcells were incubated with macrophage monolayers. Figure 5 shows that the uptake of the vesiculated cells was about threefold higher than the uptake of nonvesiculated controls. Additional evidence suggesting that vesciculated RBC are recognized by cells of the RES was obtained by determining the clearance rates of vesiculated RBC in vivo. Figure 6 shows that control RBC were cleared with a Tln of about 80 hours, whereas the clearance kinetics of vesiculated cells suggested a two-component system. It can be seen that about half of the vesiculated cells were cleared within 12 hours (corresponding to a T l n of <2 hours) with about 80% of the cleared radiation recovered in the animals' liver and spleen (data not shown). The remaining population seemed to be cleared withthe Fig 3. Percoll gradient separation of DLPC-vesiculated RBC. Human RBC were collected into heparin and vesiculated as described in the Materials andMethods. The cells were thenwashed and separated in Percoll. (A) ControlRBC; (B1 vesiculated RBC. Arrow denotes vesicles that localized at the top of the gradient. From www.bloodjournal.org by guest on February 6, 2015. For personal use only. 2959 VESICULATION-INDUCED PS EXPRESSION AND AGING 600 r control 500 0remnant B r e m n a n t + DTT 400 h CJ, c 300 v) a 200 100 changes lead to their recognition by macrophages and rapid clearance from the peripheral circulation of syngeneic animals. a property characteristic of normal Percoll-separated dense (aged) cells.' Thus. there seems to be a strong relationship between the presence of PS on cell surfaces and macrophage recognition/clearance mechanisms. Indeed, PS has been shown to participate in various cell-cell interactions,"." cell activation and hemostasis,24"" cell aging,"." membrane fusion and apoptosis."." Similar to the redistribution ofPS obtained during Ca" ionophore-mediated vesiculation of RBC,'" R B C vesiculation with DLPC is associated with a rapid redistribution of cell-surface lipids. Analysis of cell-surface PS by prothrombinase activity showed that the cells immediately started to correct abnormal lipid distributions. This can be seen from the results showing that inhibition of PS transport byPDA resulted in 1.7-fold higher prothrombinase levels which, on treatment with DTT, returned to values of remnant cells not treated with PDA. This conclusion is also supported by the in vivo clearance studies which showed that removal of vesiculated R B C from the circulation was a two-component system: a fast component that was measurable immediately after injection and a component with rates identical to control values after -30 minutes. R B C were treated with PDA at 0°C to monitor the clearance of vesiculated and transport inhibited cells. Although PDA induced PS independent membrane alterations that re- - 0 normal RBC PDA-treated RBC Fig 4. Prothrombinaseactivity of vesiculated RBC.Normal human RBC and PDA-treated (2 mmol/L for 20 minutes at 0°C). PStransport inhibited RBC were vesiculated as described in Fig 3. The cells were then washed with 1% BSA at 8509 to remove vesicles and residual DLPC. The PS concentration in the outer leaflet was then measured by the prothrombinase activity assay. Some cells, before the prothrombinase assay, were washed with DTT (5 mmol/LI to reverse the inhibitory effects of PDA. The resultsare expressed asnanograms of PS equivalents/10' cells. ately cleared with a TI/?of -20 min and completely eliminated from the peripheral circulation within 6 hours. The data presented above are consistent with the concept that vesiculation results in membrane scrambling that leads to the exposure ofPSat the cell's outer leaflet. However, the results shown in Fig 6 are consistent with the existence ofat least two distinct R B C populations. Assuming that vesiculation generates uniformly scrambled cells, then normal lipid transport mechanisms must be concurrently correcting vesiculation-dependent membrane disorganization. To examine whether lipid asymmetry was reestablished allowing the cells to escape recognition, the clearance of vesiculated and transport-inhibited R B C was determined. Figure 7 shows that vesiculated cells that were unable to transport PS (PDA-treated RBC) were cleared faster (TI/?-3 hours) than vesiculated cells not treated with the transport inhibitor (TI/?"14 hours). The addition of DTT, but not glutathion, abrogated the inhibitory effects of PDA (Fig 7; see Discussion). DISCUSSION It has previously been shown that dense R B C separated on field-formed isodensity gradients correspond to distinct cell populations of increasing age as determined by cell area and cell volume" and relatively short lifespans in vivo.' In this study we have shown that artificially induced vesiculation of R B C in vitro, a phenomenon that spontaneously occurs in vivo" and upon long-term storage of cells in vitro,'".2x results in an increase in cell density associated with disruption of the normally asymmetric distribution of PS. These 0 1 2 3 Incubation time (hr) Fig 5. Uptake of '*'I-labeled remnant RBC by macrophages. Murine RBC were labeled with '*'l and vesiculated with DLPCas described in the Materials and Methods section. A 10-fold excessof RBC was incubatedwith themonolayersand washed at theindicated times. The fraction of RBC remaining in the wells was determined by scintillation counting. Control normal RBC ( 0 )and vesiculated remnant RBC ( W . From www.bloodjournal.org by guest on February 6, 2015. For personal use only. 2960 DIAZ, MORKOWSKI, AND SCHROIT Time (hrs) Fig 6. Clearance of S’Cr-labeled remnant RBC and RBC vesicles. Murine RBC were labeled with and vesiculated with DLPC as described. The cells and vesicles were separated by differential centrifugation, washed with 1% BSA t o remova residualDLPC, and injectedintravenously into syngeneic mice. At the indicated intervals aliquots of blood were collected from the tail vein and the fraction of“Cr remaining in the peripheral circulation was determined by scintillation counting. Initial rates of clearance were estimatedfrom theslopes obtained fromlinear fits of the initial datapoints. (A) control normalRBC (0)and vesiculated remnant RBC (m); (B) vesicles. Inset shows the slopes of clearance extrapolated t o 99% clearance (see text). sulted in cell clearance, vesiculated and PDA-treated cells were recognized and cleared more rapidly than the nonvesiculated PDA-treated controls. Consistent with the results obtained in the prothrombinase assay (Fig 4),DTT abolished the effects of PDA causing the cells to be cleared at rates identical to vesiculated cells (Fig 6). Reduced glutathion, on A the other hand, was without effect. Because glutathion cannot penetrate the membrane bilayer, this result indicates that, similar to sulfhydryls critical to PS tran~port,~’ sites critical to oxidation-dependent recognition of RBC also reside on the luminal side of the membrane. These data are consistent with results showing that in vivo clearance43and binding of diamide-treated RBC to macrophages4 is inhibitable with DTT and that oxidation of RBC with H,O, or malonyldialdehyde decreases PS-transport and induces partial scrambling of phospholipids across the bilayer membrane.38 In summary, the results presented here suggest that vesiculation of RBC scrambles membrane lipids which results in the exposure of PS at the cell surface. Although these data suggest that PS can participate either directly or indirectly in cell recognition, they do not rule out the participation of other specific mechanisms in this process. ACKNOWLEDGMENT The assistance of Elizabeth Bruckheimer and Polina Khaskina is gratefully acknowledged. L REFERENCES 1 0 25 50 Time (hr) Fig 7. Effect of PDA on the clearance of normal and vesiculated RBC. “Cr-labeled PDA-treated l2 mmol/L for 20 minutes at 0°C) mouse RBC were vesiculated, washed with 1% BSA, and injected of blood were into syngeneic mice. At theindicated intervals aliquots collected and the fractionof 61Cr remaining in the peripheralcirculation was determind by acintillation counting. (A),control normal RBC; (W, normal vesiculated RBC; (W, PDA-treated RBC; W, vesiculated, PDA-treated RBC; (0).DlT-treated, veaiculatod PDA-treated RBC. 1 . Bartosz G, Grzelinska E, Bartkowiak A: Aging of the erythroof bovine erythrocytes. cyte. XIX. Decrease in surface charge density Mech Ageing Dev 24:1, 1984 2 . Bartosz G: Erythrocyteaging:Physicalandchemicalmembrane changes. Gerontology 37:33, 1991 3. Dhermy D, Simeon J, WautierMP, Boivin P,Wautier JL: Role of membrane sialic acid content in the adhesiveness of aged erythrocytes to human cultured endothelial cells. Biochim Biophys Acta 904:201, 1987 4. Beppu M, Takahashi T, Hyashai T, Kikugawa K: Mechanism of macrophage recognition of SH-oxidized erythrocytes: Recognition of glycophorin A on erythrocytesby a macrophage receptorfor sialosaccharides. Biochim Biophys Acta 1223:47, 1994 5. KayMMB: Role of physiologic autoantibodyintheremoval of senescent human red cells. J Supramol Struct 9555, 1978 From www.bloodjournal.org by guest on February 6, 2015. For personal use only. VESICULATION-INDUCED PS EXPRESSION AND AGING 6. Kay MMB, Goodman S , Whitfield C, Wong P, Zaki L, Rudloff V: The senescent cell antigen is immunologically related to band 3. Proc Natl Acad Sci USA 80:1631, 1984 7. Allen TM, Williamson P, Schlegel RA: Phosphatidylserine as a determinant of reticuloendothelial recognition of liposome models of the erythrocyte surface. Roc Natl Acad Sci USA 85:8067, 1988 8. Connor J, Pak CC, Schroit AJ: Exposure of phosphatidylserine in the outer l e d e t of human red blood cells: Relationship to cell density, cell age, and clearance by mononuclear cells. J Biol Chem 269:2399, 1994 9. Schroit A J , Madsen J, Tanaka Y: In vivo recognition and clearance of red blood cells containing phosphatidylserine in their plasma membranes. J Biol Chem 2605131, 1985 10. Savill J, Dransfield I, Hogg N, Haslett C: Vitronectin receptormediated phagocytosis of cells undergoing apoptosis. Nature 343:170, 1990 11. Fadok VA, Savill JS, Haslett C, Bratton DL, Doherty DE, Campbell PA, Henson PM: Different populations of macrophages use either the vitronectin receptor or the PS receptor to recognize and remove apoptotic cells. J Immunol 149:4029, 1992 12. Sadahira Y, Yoshino T, Monobe Y: Very late activation antigen 4-vascular cell adhesion molecule I interaction is involved in the formation of erythroblastic islands. J Exp Med 18 1:411, 1995 13. Nishikawa K, Arai H, Inoue K: Scavenger receptor-mediated uptake and metabolism of lipid vesicles containing acidic phospholipids by mouse peritoneal macrophages. J Biol Chem 265:5226, 1990 14. Stanton LW, White RT, Bryant CM, Protter AA, Endemann G: A macrophage Fc receptor for IgG is also a receptor for oxidized low density lipoprotein. J Biol Chem 267:22446, 1992 15. Sambrano GR, Steinberg D: Recognition of oxidatively damaged and apoptotic cells by an oxidized low density lipoprotein receptor on mouse peritoneal macrophages: Role of membrane phosphatidylserine. Proc Natl Acad Sci USA 92:1396, 1995 16. Sambrano GR, Parthasarathy S , Steinberg D: Recognition of oxidatively damaged erythrocytes by a macrophage receptor with specificity for oxidized low density lipoprotein. Proc Natl Acad Sci USA 91:3265, 1994 17. Savill J, Fadok V, Henson P, Haslett C: Phagocyte recognition of cells undergoing apoptosis. Immunol Today 14:131, 1993 18. Fadok VA, Voelker DR, Campbell PA, Cohen JJ, Bratton DL, Henson PM: Exposure of PS on the surface of apoptotic lymphocytes triggers specific recognition and removal by macrophages. 148:2207, 1992 19. Pradhan D, Williamson P, Schlegel RA: Phosphatidylserine vesicles inhibit phagocytosis of erythrocytes with a symmetric transbilayer distribution of phospholipids. Mol Memb Biol 11:181, 1994 20. Piomelli S, Seaman C, Reibman J, Tytun A, Graziano J, Tabachnik N, Corash L: Separation of younger red cells with improved survival in vivo: An approach to chronic transfusion therapy. Proc Natl Acad Sci USA 75:3474, 1978 21. Piomelli S , Seaman C: Mechanism of red blood cell aging: Relationship of cell density and cell age. Am J Hematol42:46, 1993 22. Lutz HU, Stammler P, Fasler S , Ingold M, Fehr J: Density separation of human red blood cells on self forming Percol1 gradients: Correlation with cell age. Biochim Biophys Acta 11 16:1,1992 23. Mohandas N, Groner W: Cell membrane and volume changes during red cell development and aging. Ann NY Acad Sci 554:217, 1989 24. Waugh RE, Narla M, Jackson CW, Mueller TJ, Suzuki T, Dale GL: Rheologic properties of senescent erythrocytes: Loss of surface area and volume with red blood cell age. Blood 79: 1351, 1992 25. Dumaswala UJ, Greenwalt TJ: Human erythrocytes shed exocytic vesicles in vivo. Transfusion 25:490, 1984 2961 26. Rumsby MG, Trotter J, Allan D, Michell RH: Recovery of membrane micro-vesicles from human erythrocytes stored for transfusion: A mechanism for the erythrocyte discocyte-to-spherocyte shape transformation. Biochem Soc Trans 5:126, 1977 27. Lutz HU, Liu S-Ch, Palek J Release of spectrin-free vesicles from human erythrocytes during ATP depletion. J Cell Biol 73548, 1977 28. Greenwalt TJ, Dumaswala UJ: Effect of red cell age on vesiculation in vitro. Br J Haematol 684.65, 1988 29. Ott P, Hope MJ, Verkleij AJ, Roelofsen B, Brodbeck U, van Deenen LLM: Effect of dimyristoylphosphatidylcholine on intact erythrocytes. Biochim Biophys Acta 641:79, 1981 30. Connor J, Schroit AJ: Transbilayer movement of PS in erythrocytes: Inhibition of transport and preferential labeling of a 3 1,OOO Da protein by sulfydryl reactive reagents. Biochemistry 272348, 1988 31. Connor J, Bucana C, Fidler U, Schroit M: Differentiationdependent expression of phosphatidylserine in mammalian plasma membranes: Quantitative assessment of outer leaflet lipid by prothrombinase complex formation. Proc Natl Acad Sci USA 86:3184, 1989 32. Utsugi T, Schroit AJ, Connor J, Bucana CD, Fidler U: Elevated expression of phosphatidylserine in the outer membrane leaflet of human tumor cells and recognition by activated human blood monocytes. Cancer Res 51:3062, 1991 33. Schlegel RA, Prendergast TW, Williamson P: Membrane phospholipid asymmetry as a factor in erythrocyte-endothelial cell interactions. J Cell Physiol 1123:215, 1985 34. Bevers EM, Comfurius P, Zwaal RFA: Changes in membrane phospholipid distribution during platelet activation. Biochim Biophys Acta 73657, 1983 35. Rosing J, Bevers EM, Comfurius P, Hemker HC, van-Dieijen G , Weiss HJ, Zwaal RFA: Impaired factor X and prothrombin activation associated with decreased phospholipid exposure in platelets from a patient with a bleeding disorder. Blood 651.557, 1985 36. Sims PJ, Wiedmer T, Esmon CT, Weiss HJ, Shattil SJ: Assembly of the platelet prothrombinase complex is linked to vesiculation of the platelet plasma membrane. Studies in Scott syndrome: An isolated defect in platelet procoagulant activity. J Biol Chem 264:17049, 1989 37. Shukla SD, Hanahan DJ: Membrane alteration in cellular aging: Susceptibility of phospholipids in density (age)-related human erythrocytes to phospholipase A*. Arch Biochem Biophys 214:335, 1982 38. Henmann A, Devaux PF: Alteration of the aminophospholipid translocase activity during in vivo and artificial aging in human erythrocytes. Biochim Biophys Acta 1027:41, 1990 39. Farooqui SM, Wali RK, Baker RF, Kaka VK: Effect of cell shape, membrane deformability and phospholipid organization on phosphate-calcium-induced fusion of erythrocytes. Biochim Biophys Acta 904:239, 1987 40. Song LY, Baldwin JM, O’Reilly R,LucyJA: Relationship between surface exposure of acidic phospholipids and cell fusion in erythrocytes subjected to electrical breakdown. Biochim Biophys Acta 1104:1, 1992 41. Schewe M, Muller P, Korte T, Herrmann A: The role of phospholipid asymmetry in calcium-phosphate-inducedfusion of human erythrocytes. J Biol Chem 2675910, 1992 42. Comfurius P, Senden JMG, Tilly RHJ, Schroit AJ, Bevers EM, Zwaal RFA: 1990. Loss of membrane phospholipid asymmetry in platelets and red cells may be associated with calcium-induced shedding of plasma membrane and inhibition. Biochim Biophys Acta 1026:153, 1990 43. Johnson GJ, Allen DW, Flynn TP, Finkel B, White JG: Decrease survival in vivo of diamide-incubated dog erythrocytes. A model of oxidant induced hemolysis. J Clin Invest 66:955, 1980 From www.bloodjournal.org by guest on February 6, 2015. For personal use only. 1996 87: 2956-2961 Generation of phenotypically aged phosphatidylserine-expressing erythrocytes by dilauroylphosphatidylcholine-induced vesiculation C Diaz, J Morkowski and AJ Schroit Updated information and services can be found at: http://www.bloodjournal.org/content/87/7/2956.full.html Articles on similar topics can be found in the following Blood collections Information about reproducing this article in parts or in its entirety may be found online at: http://www.bloodjournal.org/site/misc/rights.xhtml#repub_requests Information about ordering reprints may be found online at: http://www.bloodjournal.org/site/misc/rights.xhtml#reprints Information about subscriptions and ASH membership may be found online at: http://www.bloodjournal.org/site/subscriptions/index.xhtml Blood (print ISSN 0006-4971, online ISSN 1528-0020), is published weekly by the American Society of Hematology, 2021 L St, NW, Suite 900, Washington DC 20036. Copyright 2011 by The American Society of Hematology; all rights reserved.
© Copyright 2026