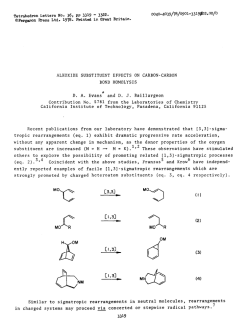
Adrenergic Modulation of Human Polymorphonuclear
From www.bloodjournal.org by guest on February 6, 2015. For personal use only. Adrenergic Modulation of Human Polymorphonuclear Leukocyte Activation. Potentiating Effect of Adenosine By Gianfranco Bazzoni, Elisabetta Dejana, and Aldo Del Maschio The activation of polymorphonuclear leukocytes (PMN) is an important step in the development of tissue damage associated with inflammatory and ischemic conditions. Catecholamines have been reported t o inhibit PMN functions, but the high concentrations required cast doubt on their actual relevance as a defense mechanism. We report here that adenosine, which is actively released in ischemic conditions, potentiates the effect of epinephrine and reduces the minimal active concentration required t o inhibit PMN activation by at least two orders of magnitude. Epinephrine caused a dose-related reduction of chemiluminescence, superoxide anion generation, enzyme release (lysozyme and p-glucuronidase), and adhesion t o endothelial cell (EC)monolayers in human PMN activated by N-formyl-methionyl-leucyl-phenylto alanine (fMLP). This effect was only apparent at mol/L. As expected, adenosine caused dose-dependent reductions of superoxide anion production and PMN adhesion t o EC. Adenosine and epinephrine combined had an additive effect on PMN superoxide production and adhesion t o EC. The minimal effective concentration of epinephrine in combination with I O - * mol/L adenosine was in the range of lo-’’ t o IO-’ mol/L. In contrast, adenosine inhibited only slightly enzyme release and did not significantly enhance the inhibition by epinephrine on this parameter. Studies with adenosine analogs suggested that the potentiating effect of adenosine was mediated by A, receptors. The mechanism of potentiation was not related t o additive effect on intracellular cyclic adenosine monophosphate levels. Epinephrine‘s ability to modulate PMN activation and the potentiating effect of adenosine may constitute a form of physiologic protection against tissue injury in inflammatory and ischemic processes. o 1991 b y The American Society of Hematology. R sion to endothelial cells (EC).l3.l4Therefore, it was of interest to investigate whether adenosine potentiated the inhibitory effect of epinephrine on PMN responsiveness. The results reported here show that at high concentrations epinephrine inhibited the PMN respiratory burst, enzyme release, and adhesion to EC. Adenosine, at concentrations equal to or lower than those in blood, markedly potentiated the inhibitory effect of epinephrine on PMN activation bringing the active concentration of epinephrine down to within the physiologic range. These data support the hypothesis that distinct endogenous mediators released during tissue ischemia may cooperate to constitute a more effective defense mechanism against harmful PMN activation. ELEASE OF TOXIC oxygen free radicals and proteolytic enzymes by activated polymorphonuclear leukocytes (PMN) accounts for much of the tissue damage described in inflammatory reactions’ and in ischemic conditions like myocardial infarction and “ischemia-reperfusion Biochemical modifications that can interfere with PMN functions might consequently condition the pathologic course of these diseases. The heightened sympathetic activity that parallels the early phases of acute myocardial infarction4 and results in elevated catecholamine levels in blood’ might limit the effects of excessive PMN activation in infarcted areas. Indeed, PMN have &-adrenergic receptors6whose stimulation by isoprenaline results in an adenyl cyclase-mediated inhibitory effect on PMN activation.’.’” However, very high doses of a selective synthetic agonist, such as isoprenaline, needed to produce appreciable in vitro inhibition raise some doubt on the physiologic relevance of the catecholamines’ effect on PMN.” Little is known about whether non-adrenergic agents, endogenously released in conditions of acute vascular occlusion, influence the adrenergic control of leukocyte activation. Among the biochemical responses to tissue ischemia, the release of adenosine is one of the most important.’* Adenosine can act as a physiologic defense mechanism by inhibiting the PMN oxygen burst and adhe- From the Laboratory of Vascular Biology, Istituto di Ricerche Farmacologiche, “Mario Negri; ’’Milano, Italy. Submitted June 26,1990; accepted January 4, 1991. Supported by the Italian National Research Council (Project CNR No. 89.01277.04). Address reprint requests to Gianfranco Bazzoni, MD, Laboratory of VascularBiology, Istituto di Ricerche Farmacologiche, “Mario Negn”’ E a Eritrea 62, 20157Milan0, Italy. The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. section 1734 solely to indicate this fact. 0 1991 by The American Society of Hematology. 0006-4971191J7709-0023$3.OOlO 2042 MATERIALS AND METHODS Chemicals and stimuli. The suppliers for chemicals were Sigma Chemical Co (St Louis, MO) for N-formyl-methionyl-leucylphenylalanine (NLP), zymosan A (from Saccharomyces cerevisiae), epinephrine, isoprenaline, adenosine, NECA, CPA, phenylephrine, cytochalasin B, phenolphthalein standard solution, phenolphthalein glucuronic acid, xanthine, xanthine oxidase, horseradish peroxidase, cytochrome c type 111, 4-(2-hydroxyethyl)-lpiperazine-ethane sulfonic acid (HEPES), and 5-amino-2,3-dihydro1,4-phthalazinedione (Luminol); Imperial Chemical Industries (Macclesfield, England) for propranolol; Nycomed AS (Oslo, Norway) for Lymphoprep; Boehringer Mannheim (Mannheim, Germany) for Micrococcus luteus, lysozyme, and superoxide dismutase (SOD); Pharmacia Fine Chemical (Uppsala, Sweden) for Dextran T 500; Kabi Vitrum (Stockholm, Sweden) for human fibrinogen; Amersham (Buckinghamshire, England) for Na;’CrO, and the cyclic adenosine monophosphate (CAMP)radioimmunoassay kit. Ro 20-1724 was a generous gift of Prodotti Roche (Milano, Italy). All reagents for EC culture were purchased from GIBCOEurope (Paisley, Scotland). Tissue culture plates and flasks were obtained from Cell Cult, Flow Laboratories (Milano, Italy). All other materials were pure reagent grade. fMLP was dissolved in dimethyl sulfoxide (DMSO) to a final concentration of 50 mmol/L, stored at -20°C and diluted in isotonic saline just before use; zymosan and opsonized zymosan particles were prepared as previously described.15Epinephrine and isoprenaline (the L -isomers of the bitartrate salt), propranolol (the Blood, Vol77, No 9 (May 1). 1991: pp 2042-2048 From www.bloodjournal.org by guest on February 6, 2015. For personal use only. 2043 EPINEPHRINE,ADENOSINE, AND LEUKOCYTE FUNCTIONS D,L-isomer of the hydrochloride salt), and adenosine were dissolved in isotonic saline as stock solutions (10 mmol/L) just before use. Luminol was dissolved as stock solution (90 mmol/L) in DMSO, further diluted in isotonic saline and stored at 4°C protected from light. The final DMSO concentration never exceeded 0.001%. PMNisolation. Venous blood from healthy donors who had not received any medication for at least 2 weeks was anticoagulated with trisodium citrate (3.8%, 1:9 vol/vol). PMN were isolated by Dextran sedimentation followed by Lymphoprep gradient and hypotonic lysis of erythrocytes.16PMN were washed and resuspended (5 x lo6 cells/mL) in ice-cold HEPES-Tyrode buffer (pH 7.4) containing (mmol/L): 129 NaC1, 9.9 NaHCO,, 2.8 KCI, 0.8 KH,PO,, 0.8 MgCI, . 6H,O, 5.6 dextrose, 1 CaCl,, and 10 HEPES. Cell suspensions contained more than 97% viable PMN, as evaluated by the Trypan blue exclusion test. PMN were used within 2 hours after their isolation. Chemiluminescenceassay. PMN suspensions (5 x lo6cells/mL, in a final volume of 1 mL), exposed to 90 nmol/L luminol and 2.5 pg/mL cytochalasin B, were preincubated at 37°C for the time indicated under constant stirring, in the Chrono-Log lumiaggregometer (Havertown, PA), in the presence or absence of the adrenergic agents. Changes in chemiluminescence (CL) were recorded for 7 minutes after fMLP or opsonized zymosan stimulation.17 CL values reported in the present work are expressed in millivolts and correspond to the maximal amplitude of the signal recorded. Peak amplitude was measured in millimeters and transformed into millivolts, the maximal tracing excursion (160 mm) corresponding to 1,600 mV. CL increased linearly with the concentration of the stimulus and the number of cells (ED,,: 68 f 8, 460 f 30, and > 1,000 x M fMLP, for 5, 2.5, and 1.25 x lo6 PMN/mL, respectively). Basal CL was always negligible. Control experiments showed that CL generated in a cell-free system by xanthine mol/L) plus xanthine oxidase (1 U/mL), in the presence of horseradish peroxidase (9 U/mL), was not reduced by epinephrine (483 f 26 mV and 494 f 35 mV, respectively, in the presence and absence of mol/L epinephrine (n = 3) or by any of the other compounds used, thus excluding that light quenching accounted for the inhibitory effect described. Superoxide union generation. Superoxide anion generation was determined by measuring the reduction of cytochrome c, as described.'* PMN suspensions (5 x lo6 cells/mL) were preincubated at 22°C for 3 minutes with cytochrome c mol/L) in the presence or absence of epinephrine, stimulated with fMLP mol/L), and then incubated for different times (10 to 60 minutes). Cell suspensions were centrifuged (12,OOOg for 1 minute) and aliquots of the supernatants were transferred into a 96-well microtiter plate. Absorbance was measured simultaneously at 550 and 540 nm with a Multiskan spectrophotometer (Titertek, Flow Laboratories, Great Britain). In some experiments, PMN were preincubated (10 minutes at 22°C) with adenosine (lo-'' to mol/L) or with the adenosine analogs NECA or CPA (10-lo to mol/L) before exposure to epinephrine. In the presence of every compound tested in this study, SOD (50 U/mL) almost completely abolished cytochrome c reduction in both resting and fMLPstimulated PMN (eg, 0.8 f 0.2 v 15.7 f 0.5 nmol reduced, respectively, in the presence or absence of SOD). Adhesion of PMN to EC. EC isolated from bovine thoracic aorta were cultured in minimal essential medium (MEM) with 15% fetal bovine serum, 10 mmol/L HEPES, 2 mmol/L glutamine, 100 U/mL penicillin, 100 pg/mL streptomycin, and 2.5 &mL fungizone. Cells were maintained in a 37"C, 5% CO, humidified atmosphere and used between 8 and 16 in vitro passages. For the adhesion assays, EC detached by brief exposure to trypsin (0.25%)- EDTA (0.022%) were plated and grown to confluence in 96-well plates. PMN, prepared as described above, were radiolabeled for 1hour at room temperature with Na,s1Cr04 (1 pCi/106 cells), washed twice, and resuspended at 3 x lo6 cells/mL in HEPES-Tyrode buffer. As described," radiolabeled PMN suspensions (100 p,L) were added to each well with 0.38 mg/mL fibrinogen and 2.5 p,g/mL cytochalasin B in the presence or absence of epinephrine. Three minutes after exposure to epinephrine, cells were stimulated with mol/L fMLP and incubated for 15 minutes at 37°C. Wells were washed three times to remove nonadherent cells, and the remaining bound cells were lysed with sodium dodecyl sulfate (0.1% in 25 mmol/L NaOH) and the individual lysates were counted in a gamma 5500 counter (Beckman, Fullerton, CA). Average binding of unstimulated PMN was 86 f 7 cpmiwell, compared with 544 f 27 cpm/well for fMLP-treated PMN. In some experiments, PMN were preincubated (for 10 minutes at 22°C) with adenosine (lO-'to lo-' mol/L) before exposure to epinephrine. Enzymatic release. P-glucuronidase enzymatic activity was determined using phenolphthalein glucuronate as substrate, monitoring its cleavage at 540 nm.20Lysozyme concentration was determined by measuring the lysis of Micrococcus luteus and comparing the results with those obtained with known amounts of lysozyme.'' CAMP assay. cAMP determination was performed as described?' Briefly, PMN suspensions (5 X lo6 cells/mL) were incubated for 10 minutes at 37°C in the presence or absence of Ro 20-1724 (2 x mow). At the indicated times after the addition of the different compounds, samples were centrifuged at 12,OOOg for 20 seconds at 4°C and the supernatants were decanted. Aliquots (150 p,L) of ice-cold buffer containing Tris (5 x lo-' mol/L) and EDTA (4 x lo-' mol/L) (pH 7.5) were then added to the pellet. The samples were then boiled for 5 minutes and centrifuged. The supernatants were subsequently assayed for cAMP using a competitive protein binding assay. Statistical analysis. Results reported here are means f SEM of N experiments from different donors. Unpaired Student's t-test was used to establish the significance of differences between the means. Dose-response curves were processed by computer-assisted analysis" to establish the stimulus concentration giving 50% maximal response (ED,,) and the drug concentration giving 50% maximal inhibition (ICso). RESULTS Effect of epinephrine on PMN respiratory burst and adhesion to EC. The effect of epinephrine on the fMLPinduced respiratory burst was first tested by luminoldependent CL. Stimulation of PMN by lo-' mol/L fMLP gave a luminescent signal of 1,220 80 mV (at maximal amplitude, 2 minutes after stimulation). Incubation with epinephrine to mol/L) for 2 minutes before cell stimulation reduced fMLP-induced CL in a dose-dependent manner (IC5,,,2.8 k 0.7 x lo-' mol/L). CL triggered by a particulate stimulus such as opsonized zymosan was evaluated in parallel. Unopsonized zymosan had no effect, but stimulation of PMN with 1.5 x 10' particles/mL of opsonized zymosan elicited a signal of 1,030 ? 100 mV. Also this response was inhibited by epinephrine (IC5', 3.9 ? 0.9 x mol/L) in a concentration-dependent manner (Fig 1A). The time course indicated that CL depended on the preincubation time (0 t o 10 minutes) before exposure to fMLP and that, on incubation of the cells with lo-' mol/L * From www.bloodjournal.org by guest on February 6, 2015. For personal use only. 2044 BAZZONI, DEJANA, AND DEL MASCHIO c -9 -8 -7 -6 log [ Epinephrine] ( M ) 0 1 2 3 4 5 10 Time (min) Fig 1. (A) Effect of epinephrine on CL in human PMN. PMN suspensions (5 x 10' cells/mL) were preincubated (2 minutes, 37°C) with epinephrine before stimulationwith lO-'mol/LfMLP (m) or 1.5 x 10' particles/mL of opsonized zymosan (0).Data (mean SEMI are derived from five different experiments. *P < .05 and **P < .01, significantly different from control (without epinephrine, c). (B) Time dependence of epinephrine's inhibition of fMLP-induced CL. PMN suspensions(5 x 10' cells/mL) were incubatedfor the times indicated of lo-' mol/L epinephrine before in the presence (m) or absence (0) stimulationwith mol/L fMLP. Each point represents the mean ? SEM of three different experiments. * epinephrine, maximal reduction of the response was obtained between 2 and 4 minutes of incubation (Fig 1B). Pharmacologic evidence was obtained that the inhibitory effect was mediated by selective @-receptorstimulation: the selective P-agonist isoprenaline was more effective than epinephrine (IC5o,2 f 0.9 x mol& n = 3, P < .05 v epinephrine) and the a-agonist phenylephrine did not affect the response (not shown). Moreover, the @-blocking agent propranolol reversed the inhibition of epinephrine: pretreatment of PMN with propranolol (lo-' to mol/L) before exposure to epinephrine ( mol/L) reversed the latter's inhibitory effect on the fMLP-induced CL in a concentration-dependent manner (70% ? 3% and 98% f 4% of control, respectively, in the presence of and mol/L propranolol; P < .01, n = 3). Because CL results from the reaction of luminol with multiple oxygen metabolites (eg, superoxide anion, hydrogen peroxide, and, principally, hypochlorous acid), superoxide dismutase-inhibitable cytochrome c reduction was used in parallel to specifically assess the effect of epinephrine on superoxide anion generation. As shown in Table 1, the response increased with time plateauing 30 minutes after stimulation with fMLP. Inhibition by epinephrine peaked at 10 minutes of stimulation (IC5o,6.9 f 3 x lo-* mol/L) and decreased at 30 minutes (IC5,,,4.6 & 1.5 x mol/L), while late-phase activation at 60 minutes was only slightly affected (IC5o,1.0 f: 0.2 x mol/L). Therefore, despite differences in sensitivity and specificity for the oxygen species and in the kinetics of the signals, an inhibitory effect was similarly observed with both techniques. Epinephrine's ability to modulate PMN adhesion to EC was assessed by coincubating 5'Cr-labeled PMN with cultured EC from bovine aorta in the presence or absence of epinephrine (lo-'' to lo-' mol/L). PMN adhesion in response to mol/L fMLP was maximal at 15 minutes of stimulation (62% & 6% of adherent cells). Epinephrine inhibited PMN adhesion, at 15 minutes, in a dosedependent manner (ICso, 4.3 & 0.9 x mol/L). PMN adhesion to EC was never completely inhibited, even at the highest concentration ( mol/L) of epinephrine used. Finally, we found that at micromolar concentrations epinephrine induced an appreciable inhibition of the enzymatic release in agreement with previous report^.',^ Potentiation of epinephrine's effect on PMN activation by adenosine. To assess whether adenosine potentiated the inhibitory effect of epinephrine on superoxide anion generation, PMN suspensions were preincubated with adenosine (lo-'' to mol/L), subsequently exposed to epinephrine (lo-'' to mol/L) for an additional 3 minutes of incubation and finally stimulated with mol/L fMLP. At 30 minutes of stimulation, cytochrome c reduction was dose-dependently inhibited by both epinephrine and adenosine (Fig 2). The combination of both compounds resulted in marked inhibition of the cellular response. Indeed, in the mol/L), strong presence of low doses of adenosine inhibition was achieved even at physiologic concentrations of epinephrine (IC5,,,0.48 5 0.1 x mol/L). Adenosine's ability to modulate epinephrine inhibition of PMN adhesion to EC was assessed by preincubating PMN suspensions with adenosine (W8to lod6mol/L) before layering them onto EC monolayers. After 3 minutes of incubation with epinephrine, PMN suspensions were stimulated with lo-' mol/L fMLP. Cell adhesion was dose-dependently reduced by both compounds (Fig 3). Adenosine caused marked, dose-related enhancement of epinephrine's inhibitory effect and PMN adhesion was almost completely inhibited by micromolar concentrations of both agents combined (78% f 8% of inhibition). We next examined the effect of epinephrine and adenosine on the fMLP-induced release of lysozyme and @-glucuronidase. At 15 minutes of stimulation epinephrine caused dose-dependent reduction of the release of lysozyme (Fig 4). At the highest dose of epinephrine mol/L), the release of lysozyme was reduced to 63% ? 4%. Adenosine alone to mol/L) inhibited only slightly this response and no significant enhancement of the epinephrine's effect was observed when PMN were preincubated with adenosine before exposure to epinephrine (Fig 4). Table 1. Effect of Epinephrineon fMLP-Induced Superoxide Anion Generation Time After Stimulation (min) Epinephrine (mol/L) - 10-10 10-9 10-8 10-7 10-6 10 7.4 2 6.6 2 5.9 2 4.9 2 2.6 ? 1.4 2 2.0 1.9 1.7 1.9 1.1* 0.3t 60 30 14.9 13.9 13.3 11.6 8.7 6.1 f 2.2 2 2.3 2 2.2 2 2.6 ? ? 2.6* 1.8t 15.2 f 1.2 14.4 5 1.1 14.2 5 1.1 12.0 f 1.5* 9.3 5 0.3* 8.0 5 0.4t PMN suspensions (5 x lO'cells/mL) were preincubated (3 minutes at 22°C) in the presence or absence of epinephrine, with 2.5 pg/mL mol/L cytochrome c. Cells were then stimulated cytochalasin B and with lO-'mol/L fMLP. At the indicated timrs after stimulation, cells were spun down at 12,OOOg for 1 minute, supernatants were collected, and adsorption at 540 to 550 nm was determined. Data are expressed as nanomoles of reduced cytochrome c and are means f SEM of four separate experiments each performed in duplicate. *P < .05 and t P < .01, significantly different from control values (in the absence of epinephrine). From www.bloodjournal.org by guest on February 6, 2015. For personal use only. 2045 EPINEPHRINE, ADENOSINE, AND LEUKOCYTE FUNCTIONS 100- 100- 75 - 80 - 50 - 25 60 - - 'V I C 1 I -10 I -8 -9 0 I -7 -6 c log [ Epinephrine ] ( M ) 100- - 50 - 25 - 0' I I c -10 I I -9 -8 I I -9 -8 -7 -6 -5 log [ Epinephrine ] ( M ) Fig 2. Adenosine potentiates epinephrine's inhibition of fMLPinduced superoxide anion generation. PMN suspensions (5 x lo6 cells/mL) were preincubated (10 minutes) in the absence (m) or presence of different doses of adenosine (0. lo-'" mol/L; A, lo-' mol/L; and A, lo-' mol/L), subsequently exposed t o epinephrine for another 3 minutes and finally stimulated with lo-' mol/L fMLP. Thirty minutes after stimulation, supernatants were collected and absorption was measured at 540 t o 550 nm. Data (mean ? SEM, n = 3) are expressed as percent of maximal response (in the absence of epinephrine and adenosine). 75 -10 I I -7 -6 -5 log [ Epinephrine ] ( M ) Fig 3. Adenosine potentiates epinephrine's inhibition of fMLPinduced adhesion of PMN t o EC. "Cr-labeled PMN suspensions (3 x 10' cells/mL) were preincubated (10 minutes) in the absence (m) or presence of different doses of adenosine (0. lo-' mol/L; A, mol/L; and A, mol/L), layered onto EC monolayers, exposed t o epinephrine, and incubated for another 3 minutes before stimulation mol/L fMLP. After incubation (15 minutes, 37°C. 5 % CO,), with nonadherent cells were removed and radioactivity associated with adherent cells was determined. Each point represents the mean ? SEM for three determinations in a single experiment. Two additional experiments gave comparable results. Fig 4. Adenosine does not potentiate epinephrine's inhibition of fMLP-induced release of lysozyme. PMN suspensions (5 x 10' cells/ mL) were preincubated (10 minutes) in the absence ( W ) or presence of different doses of adenosine (0. lo-' mol/L; and A, IO-' mol/L), subsequently exposed t o epinephrine for another 3 minutes and finally stimulated with lo-' mol/L fMLP. Fifteen minutes after stimulation, supernatants were collected and lysozyme release was determined as described. Data (mean f SEM, n = 3) are expressed as percent of maximal response (in the absence of epinephrine and adenosine). Similarly, the release of P-glucuronidase was inhibited by epinephrine in a dose-dependent way. Adenosine neither inhibited per se the response nor potentiated epinephrine's effect (data not shown). Effect of adenosine receptor agonists on superoxide anion generation. Adenosine modulates PMN functions through two pharmacologically distinct surface receptors, which have been identified with the A, and A, types. To define the receptor through which adenosine potentiates the action of epinephrine, we examined the effects of two highly selective adenosine analogs, alone and in combination with epinephrine. NECA (lo-'' to mol/L), the most potent agonist for Az receptor^,'^ inhibited per se superoxide anion generation and potentiated the inhibitory effect of epinephrine in a dose-dependent way (Fig 5A). In contrast, preincubation of PMN with the highly selective A, receptor agonist CPA (lo-'' to mol/L) did not result in enhancement of the inhibitory effect of epinephrine (Fig SB). These data suggest that adenosine enhances the inhibitory effect of epinephrine by occupancy of A, receptors. Effect of epinephrine and adenosine on intracellular CAMP levels. To evaluate whether amplification of the adenylate cyclase response to epinephrine is the mechanism whereby adenosine enhances the inhibitory effect of epinephrine, intracellular CAMP levels were measured in resting and fMLP-stimulated PMN (Table 2). In these experiments, inhibition of CAMPphosphodiesterase with the nonmethylxanthine CAMP phosphodiesterase inhibitor Ro 20-1724 was necessary to magnify the CAMP response to the From www.bloodjournal.org by guest on February 6, 2015. For personal use only. BAZZONI, DEJANA, AND DEL MASCHIO 2046 1oc 7 I ;ip v c ._ 6 A A 75 100 75 4 T) e, 50 50 25 25 E P .c 0 I C c -10 -9 -8 -7 -6 C log [ Epinephrine ] ( M ) -10 -9 -8 agonists. Indeed, in the absence of Ro 20-1724 no significant changes were evident after exposure to both agonists (not shown). Ro 20-1724 caused per se a substantial and persistent increase of cAMP levels in resting PMN (1.1 f 0.2 v 7.9 f 0.5 pmol/107 PMN, respectively, in the absence and presence of Ro 20-1724). In the presence of Ro 20-1724 both epinephrine and adenosine ( mol/L) increased CAMP levels and their combination was only slightly more effective than each agonist alone. Stimulation with fMLP (lo-’ mol/L) caused twofold elevation in CAMPcontent and amplified the accumulation of cAMP induced by epinephrine and adenosine. However, the combination of the two agents did not further increase cAMP levels. DISCUSSION Among the multiple interactions of the sympathoadrenergic system with immune f~nctions,’~ the effect of physiologic adrenergic agents on the functional activation of PMN is important in view of a potential role in modulating inflammatory reactions. We report here that epinephrine, at high concentrations, reduced the fMLPinduced respiratory burst and adhesion to EC of human Table 2. Effect of Epinephrine and Adenosine on cAMP Levels in Human PMN cAMP Content (pmolllO’ cells) Additive(s) Saline Epinephrine (lo-’mol/L) Adenosine mol/L) Epinephrine mol/L) mol/L) -7 log [ Epinephrine ] ( M ) -fMLP +fMLP 7.9 2 0.5 14.7 2 0.9 9.1 2 0.5 27.4 2 1.4’ 10.1 2 0.3* 27.9 2 0.5* + adenosine (10.’ 12.1 2 0.7* 28.3 2 0.5* PMN suspensions (5 x 10‘ cells/mL) were preincubated for 10 minmol/L), exposed to utes at 37°C in the presence of Ro 20-1724(2x adenosine (or to an equivalent volume of saline) and further incubated for another 2 minutes before addition of epinephrine and fMLP or control buffer. One minute after stimulation with fMLP incubations were terminated as described. Values represent the means t SEM of three determinations, each performed in duplicate. *P < .05 versus control (saline 2 fMLP). -6 Fig 5. NECA but not CPA potentiates epinephrine‘s inhibition of fMLP-induced superoxide anion generation. PMNsuspensions(5 x 106cells/mL) were preincubated (10 minutes) in the absence (M) or presence of different doses of NECA (A) or CPA ( 8 ) (0, lo-’’ mol/L; A, lo-* mol/L; and A, lo-* mol/L), subsequently exposed to epinephrine for another 3 minutes and finally stimulated with mol/L fMLP. Thirty minutes after stimulation, supernatants were collected and absorptionwas measured at 540 to 550 nm. Data (mean 2 SEM, n = 3) are expressed as percent of maximal response (in the absence of epinephrine and adenosine). PMN. Addition of low concentrations of adenosine markedly potentiated the effect of epinephrine and reduced the minimal active concentration of the catecholamine by at least two orders of magnitude. We first evaluated the effect of epinephrine alone on the activation of PMN induced by the chemotactic peptide fMLP. Short preincubation with epinephrine resulted in dose-dependent inhibition of oxygen radical generation evaluated by luminol-amplified CL and superoxide dismutase-inhibitable cytochrome c reduction. Then we obtained evidence that epinephrine dosedependently reduced fMLP-stimulated adherence of PMN to cultured endothelium. A general inhibitory effect of epinephrine on leukocyte adhesiveness to EC is suggested by the fact that epinephrine, as reported by Boxer et al,25 reduced also the basal adhesion of unstimulated PMN to EC, which probably accounts for the detachment of PMN from the marginating pool and the consequent neutrophilia observed after administration of epinephrine in vivo.26 appreciable However, in agreement with other inhibition of PMN adhesion and release of oxygen radicals and proteolytic enzymes was only seen at epinephrine concentrations greater than lo-’ mol/L, a dose approximately two orders of magnitude higher than the physiologic plasma levels of the ~atecholamine.~ Nothing is known about the effects of the interaction between adrenergic mediators and adenine derivatives on PMN activation. The role of adenosine is particularly important because this nucleoside, produced mainly in ischemic conditions,” prevents the PMN respiratory burst’’ and adhesion to EC.I4 One of the major findings of this study was that preincubation of PMN with adenosine dose-dependently potentiated the inhibitory effect of epinephrine on superoxide anion generation and on adhesion to EC (Figs 2 and 3). Circulating levels of adenosine in plasma are at least 0.3 pmol/LZ7and concentrations are higher in hypoxic and ischemic tissues.” In the presence of low doses of adenosine, even physiologic concentrations of epinephrine (subnanomolar range) appreciably inhibited both functional responses, indicating that the effects of From www.bloodjournal.org by guest on February 6, 2015. For personal use only. EPINEPHRINE, ADENOSINE, AND LEUKOCYTE FUNCTIONS 2047 catecholamines, which per se only slightly affect PMN activation, may be amplified by endogenous nonadrenergic agents. The physiopathologic relevance of this finding is underlined by the fact that adenosine derives from damaged cellsz and that oxygen-free radicals play a pivotal role in cell injury.29Sublethal concentrations of hydrogen peroxide, an oxidant produced by PMN, induce the release of cytoplasmic adenosine by EC.30Therefore, it can be postulated that the cooperative interaction of circulating epinephrine with IocalIy reIeased adenosine may dampen the harmful effects of uncontrolled phagocytic activation. The intracellular mechanism involved in the interaction between epinephrine and adenosine remains to be elucidated. The effect of adenosine is probably mediated by specific A, receptors, because the selective A, receptor agonist NECA shared the same activity while the highly selective A, receptor agonist CPA was ineffective?l A2 receptors for adenosine were first described in brain cells in vitro, where they mediated the accumulation of CAMP?' Because p-adrenoceptor stimulation also increases CAMP levels in PMN; additive stimulation of adenylate cyclase might be the mechanism underlying the interaction between catecholamines and adenosine. However, this theory was not supported by experimental evidence. We report here that, although both epinephrine and adenosine stimulated the accumulation of CAMP in resting and fMLP-activated PMN, the combination of both agents did not result in further, significant enhancement of CAMP levels. Additional indirect evidence also supports this finding. Adenosine apparently potentiates epinephrine through A2 receptor occupancy, and A, receptor modulation of PMN activation has been demonstrated to be CAMP-independent." In addition, adenosine increased the epinephrine's inhibitory effect on PMN respiratory burst and adhesion but not on degranulation, while agents that stimulate adenylate cyclase, such as prostaglandins, catecholamines, and histamine, are potent inhibitors of enzyme reIease.'~8~1' In summary, we have found that epinephrine inhibits PMN respiratory burst and adhesion to the endothelium in response to a chemoattractant stimulus, and that these effects were markedly potentiated by adenosine at concentrations normally found in plasma. Higher concentrations of adenosine (that might be reached in cases of endothelial damage or platelet a c t i ~ a t i o n ) ' ~and * ~ ~elevated levels of catecholamines (as ih pathological hyperadrenergic almost completely inhibited PMN activation. We propose that this interaction could easily be operative in many pathophysiologic conditions and could constitute an important defense mechanism against harmful PMN activation. ACKNOWLEDGMENT The authors thank Judith Baggott and Vincenzo and Felice de Ceglie for their help in the preparation of the manuscript. REFERENCES 1. Weiss SJ: Tissue destruction by neutrophils. N Engl J Med 320:365,1989 2. Werns SW, Lucchesi BR: Inflammation and myocardial infarction. Br Med Bull 43:460, 29&7 3. Ernst E, Hammerschmidt DE, Bagge U, Matrai A, Dormandy JA: Leukocytesand the risk of ischemicdiseases.JAMA 2572318,1987 4. Cryer PE: Physiology and pathophysiology of the human sympathoadrenal neuroendocrine system. N Engl J Med 303:436, 1980 5. Karlsberg RP, Cryer PE, Roberts R: Serial plasma catecholamine response early in the course of clinical acute myocardial infarction: Relationship to infarct extent and mortality. Am Heart J 102:24,1981 6. Galant SP, Underwood S, Duriseti L, Insel PA. Characterization of high-affinity @,-adrenergic receptor binding of ( -)-[3H]dihydroalprenolol to human polymorphonuclear cell particulates. J Lab Clin Med 92:613,1978 7. Zurier RB, Weissmann G, Hoffstein S, Kammermann S, Tai HH: Mechanisms of lysosomal enzyme release from human leukocytes. J Clin Invest 53:297,1974 8. Ignarro LJ, Colombo C Enzyme release from polymorphonuclear leukocyte lysosomes: Regulation by autonomic drugs and cyclic nucleotides. Science 180:1181,1973 9. Schopf RE, LemmeI EM: Control of the reduction of oxygen intermediates of human polymorphonuclear leukocytes and monocytes by p-adrenergic receptors. J Immunopharmacol5:203,1983 10. Nielson CP: @-Adrenergic modulation of polymorphonuclear leukocyte respiratory burst is dependent upon the mechanism of cell activation, J Immunol 1392392,1987 11. Marone G, Thomas LL, Lichtenstein LM: The role of agonists that activate adenylate cyclase in the control of CAMP metabolism and enzyme release by human polymorphonuclear leukocytes. J Immunol125:2277,1980 12. Headrick JP, Willis RJ: 5'-Nucleotidase activity and adenosine formation in stimulated, hypoxic and underperfused rat heart. Biochem J 261:541,1989 13. Cronstein BN, Kramer SB, Weissmann GS, Hirschhorn R: Adenosine: A physiological modulator of superoxide anion generation by human neutrophils. J Exp Med 158:1160,1983 14. Cronstein BN, Levin RI, Belanoff J, Weissmann G, Hirschhorn R: Adenosine: An endogenous inhibitor of neutrophilmediated injury to endothelial cells. J Clin Invest 78:760, 1986 15. Del Maschio A, Corvazier E, Maillet F, Kazatchkine MD, Maclouf J: Platelet-dependent induction and amplification of polymorphonuclear leucocyte lysosomal enzyme release. Br J Haematol72329,1989 16. Bqiyum A: A one-stage procedure for isolation of granulocytes and lymphocytes from human blood. General sedimentation properties of white blood cells in a 1 g gravity field. Scand J Clin Lab Invesf 21:51,1968 (suppl97) 17. DeChatelet LR, Long GI), Shirley PS, Bass DA, Thomas MJ, Henderson. FW, Cohen MS: Mechanism of the luminoldependent chemiluminescence of human neutrophils. J Immunol 129:1589,1982 18. Goldstein IM, Roos D, Kaplan HB, Weissmarm G: Complement and immunoglobulins stimulate superoxide production by human leukocytes independently of phagocytosis. J Clin Invest 56:1155,1975 19. Breviario F, Bertocchi F, Dejana E, Bussolino F IL-1induced adhesion of polymorphonuclear leukocytes to cultured human endothelial cells. Role of platelet-activating factor. J Immunol141:3391,1988 From www.bloodjournal.org by guest on February 6, 2015. For personal use only. 2048 20. Stahl PD, Fishman WH: P-D-Glucuronidase, in Bergmeyer HU (ed): Methods of Enzymatic Analysis. Weinheim, Germany, Verlag Chemie, 1984, p 246 21. De Blasi A, Lipartiti M, Motulsky HJ, Insel PA, Fratelli M: Agonist-induced redistribution of @-adrenergicreceptors on intact human mononuclear leukocyte. Redistributed receptors are nonfunctional. J Clin Endocrinol Metab 61:1081, 1985 22. Sacchi-Landriani G, Guardabasso V, Rocchetti M: NL-FIT: A microcomputer program for non-linear fitting. Comput Methods Programs Biomed 16:35,1983 23. Cronstein BN, Rosenstein ED, Kramer SB, Weissmann G, Hirschhom R: Adenosine: A physiologic modulator of superoxide anion generation by human neutrophils. Adenosine acts via an Az receptor on human neutrophils. J Immunol135:1366,1985 24. Felten DL, Felten SY, Bellinger DL, Carlson SL, Ackerman KD, Madden KS, Olschowski JA, Livnat S: Noradrenergic sympathetic neural interactions with the immune system: Structure and function. Immunol Rev 100:225, 1987 25. Boxer LA, Allen JM, Baehner RL: Diminished polymorphonuclear leukocyte adherence. Function dependent on release of cyclic AMP by endothelial cells after stimulation of @-receptorsby epinephrine. J Clin Invest 66:268, 1980 26. Steel CM, French EB, Aitchison WRC: Studies on adrenaline-induced leukocytosis in normal man. I. The role of the spleen and the thoracic duct. Br J Haematol21:307,1975 BAZZONI, DEJANA, AND DEL MASCHIO 27. Hirshhorn R, Roegner-Maniscalco V, Kuritsky L, Rosen FS: Bone marrow transplantation only partially restores purine metabolites to normal in adenosine-deaminase deficient patients. J Clin Invest 68:1387,1981 28. Sacks T, Moldow CF, Craddock PR, Bowers TK, Jacob HS: Oxygen radicals mediate endothelial cell damage by complementstimulated granulocytes. An in vitro model of immune vascular damage. J Clin Invest 61:1161,1978 29. Babior BM: Oxidants from phagocytes: Agents of defense and destruction. Blood 64:959, 1984 30. Ager A, Gordon J L Differential effects of hydrogen peroxide on indices of endothelial cell function. J Exp Med 159592,1984 31. Cronstein BN, Daguma L, Nichols D, Hutchison AJ, Williams M: The adenosine/neutrophil paradox resolved: human neutrophils possess both A, and A, receptors that promote chemotaxis and inhibit 0,-generation, respectively. J Clin Invest 85:1150, 1990 32. van Calker D, Mueller M, Hamprecht B: Adenosine regulates via two different types of receptors the accumulation of cyclic AMP in cultured brain cells. J Neurochem 33:999,1979 33. Cronstein BN, Kramer SB, Rosenstein ED, Korchack HM, Weissmann G, Hirshhorn R: Occupancy of adenosine receptors raises cyclic AMP alone and in synergy with occupancy of chemoattractant receptors and inhibits membrane depolarization. Biochem J 252:709,1988 From www.bloodjournal.org by guest on February 6, 2015. For personal use only. 1991 77: 2042-2048 Adrenergic modulation of human polymorphonuclear leukocyte activation. Potentiating effect of adenosine G Bazzoni, E Dejana and A Del Maschio Updated information and services can be found at: http://www.bloodjournal.org/content/77/9/2042.full.html Articles on similar topics can be found in the following Blood collections Information about reproducing this article in parts or in its entirety may be found online at: http://www.bloodjournal.org/site/misc/rights.xhtml#repub_requests Information about ordering reprints may be found online at: http://www.bloodjournal.org/site/misc/rights.xhtml#reprints Information about subscriptions and ASH membership may be found online at: http://www.bloodjournal.org/site/subscriptions/index.xhtml Blood (print ISSN 0006-4971, online ISSN 1528-0020), is published weekly by the American Society of Hematology, 2021 L St, NW, Suite 900, Washington DC 20036. Copyright 2011 by The American Society of Hematology; all rights reserved.
© Copyright 2026