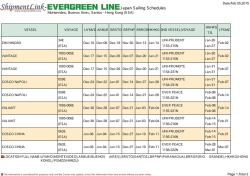
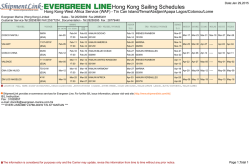
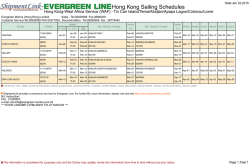
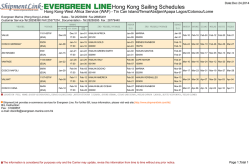
Erythropoiesis stimulating agents in Chronic Kidney
CIN 2011 6th Congress of Nephrology in Internet Erythropoiesis stimulating agents in Chronic Kidney Disease Holger Schmid KfH Nierenzentrum Muenchen – Laim, Munich, Germany and Department of Internal Medicine, Medicine, University Hospital Munich – Downtown Campus , University of Munich, Munich, Germany Erythropoiesis stimulating agents in Chronic Kidney Disease -Outline Outline-► Anemia in CKD: pathogenesis and consequences ► Treatment: ESAESA-classes, strategies and target Hb levels ► Risks of ESA use ► ESA hyporesponsiveness ► Challenges in clinical research ► Conclusions Abbreviations ► ► ► ► ► ► ► ► ► ► ► ► ► ► ► ► CAD CAPD CHF CKD CV EPO ESA ESRD Hb Hct HD HIF LVH PRCA QOL rHU EPO coronary artery disease continuous ambulatory peritoneal dialysis congestive heart failure chronic kidney disease cardiovascular erythropoietin erythropoiesis stimulating agents endend-stage renal disease hemoglobin hematocrit hemodialysis hypoxia inducible factor left ventricular hypertrophy pure red cell aplasia quality of life recombinant human erythropoietin Anemia in CKD ► Prevalence of anemia in CKD depends on severity of renal insufficiency and definition ► WHO definition of anemia ► Hb < 13.0 g/dl for adult men and postmenopausal women ► Hb < 12.0 g/dl for pre pre--menopausal women ► Predominantly normocytic and normochromic ► At every CKD stage: Higher prevalence and more pronounced severity of renal anemia in diabetic compared to nonnon-diabetic patients Anemia in CKD: Pathogenesis ► Human Erythropoietin (EPO) circulating 30.4 kDa glycoprotein growth factor (165 amino acids) acts as primary stimulus to erythropoiesis promotes terminal differentiation of erythroid progenitor (CFU(CFU-E) into normoblasts and then erythrocytes inhibits apoptosis of erythroid progenitors produced primarily by peritubular interstitial cells in kidney`s deep cortex and outer medulla (liver with only a minor contribution!) ► Anemia predominantly due to reduced erythropoietin production and only to a lesser degree due to shortened red cell survival ► Reduced availability of iron and chronic inflammation frequent contributory factors Anemia in CKD: Pathogenesis ► Almost all CKD patients without iron deficiency, inflammation or infection, have an appropriate erthyropoietic response to exogenous EPO → uraemic inhibition of erythropoiesis plays a minor role in pathogenesis of renal anemia ► Other anemia forms may coexist in CKD iron-, folate ironfolate--, and vitamin B12 B12-- deficiency chronic blood loss from gastrointestinal tract, blood drawing, HD technique Anemia in CKD: Consequences ► ► ► ► ► ► Reduced oxygen delivery to tissues → adverse impact on organ function (particularly in patients with chronic organ dysfunction) Increased risk for faster progression of renal failure ischemic adverse events in patients with CHF or CAD Reduced brain and cognitive function Reduced quality of life Increased LVH and left ventricular dilatation NOTE: Although a number of observational studies have associated anaemia in CKD with an increased risk of death to date no prospective studies have rigorously proven that renal anemia causes an increase in mortality in patients with CKD Anemia in CKD: Treatment in the PrePre-ESA Era ► Androgens Increase endogenous erythropoietin production, sensitivity of erythroid progenitors to EPO effects and prolong red blood cell survival Side effects: virilisation, liver function test abnormalities, increased risk of hepatocellular carcinoma ! ► Red blood cell transfusions transfusions Rapid raise of Hb levels and amelioration of symptoms in ESRD Significant complications: transfusiontransfusion-transmitted infection, immunologic sensitization, iron overload syndromes, volume overload and /or transfusion reactions ! Anemia in CKD: ESA Treatment ► rHu EPO Produced by gene transfer into suitable mammalian cell lines (e.g. Chinese hamster ovary cells) Approved in 1989 by FDA for treatment of anemia associated with CKD NOTE: EPO is a generic term that encompasses all genetically produced forms of erythropoietin ► Currently available and future forms of erythropoietic agents for treatment of ESRD Unmodified recombinant rhu EPOs (“short(“short-acting”) Long--acting ESAs Long EPO analogues (Biosimilar EPOs) Investigational ESAs Anemia in CKD: ESA Treatment Unmodified recombinant rhu EPOs (“short-acting”) and Long-acting ESAs ESA--Class ESA ESA -Type Mol. Weight (KDa) Serum half life (h) / administration route BioavaiBioavailability (%) Periodicity Unmodified Recombinant Rhu EPOs (“Short-(“Short Acting”) Epoetin alfa 32 32--40 8.8 / i.v. 24 24..2 / s.c. 30 30--36 1-3 times/wk 6.8 / i.v. 19 19..4 / s.c. 23 23--42 1-3 times/wk Epoetin beta Epoetin omega Epoetin delta LongLongActing ESAs DarbDarbEpoetin alpha 40 25 / i.v. 49 / s.c. 37 Every 1-2 wk C.E.R.A. 60 133 / i.v. 137 / s.c. 47 47--52 Every 2-4 wk Anemia in CKD: ESA Treatment EPO analogues (“Biosimilar” EPOs) and Investigational ESAs ESA--Class ESA ESA ESA-Type Mol. Weight (KDa) Serum half life (h)/ administration route BioavaiBioavailability (%) PerioPeriodicity EPO analogues (Biosimilar EPOs) Epoetin alfa ? n.a. ? 1-3 times/wk Epoetin zeta 30 30..6 4-5 / i.v. 24 / s.c. n .a . 1-3 times/wk Synthetic, peptidepeptide- n.a. based erythropoietin-erythropoietin receptor agonists 14 14--60 / p.o. n .a . Every 4 wks Prolyl hydroxylase n.a. inhibitors (HIF stabilizers) 7-8 / p.o. >75 Clinical Trials stopped by FDA ! InvestigaInvestigational ESAs Epoetin Biosimilars ► ► ► Biosimilars = “follow“follow-on biologics” Developed as consequence of patent expirations for rHU EPO European Medicines Agency definition “a biological medicinal product referring to an existing one and submitted to regulatory authorities for marketing authorization by an independent applicant after the existing agents' protection expires. Biosimilars can resemble the agents on which they are modeled but cannot fully copy their properties”. (Locatelli F. Oncologist 2009) ► ► ► Rapidly increasing number worldwide (except in the United States of North America) Should provide cost savings and greater accessibility NOTE: Inherent differences may produce dissimilarities in clinical efficacy, safety and immunogenicity as compared to the innovator rHU EPO More thorough knowledge of biosimilars needed to ensure appropriate use of these drugs Investigational ESAs ► Hematide Synthetic peptidepeptide-based erythropoietin receptor agonist with no structural homology to erythropoietin Activates erythropoietin receptor and stimulates erythropoiesis (Macdougall IC Curr Opin Inv Drugs 2008) ► HIF (Hypoxia (Hypoxia--inducible factor) stabilizers Orally active antianemic therapies Competitive inhibitors of HIF prolylprolyl-hydroyxlases and asparagyl asparagyl-hydroxylase enzymes Involved in the metabolisms of HIF and its transcriptional activity Increase endogenous EPO production by preventing proteasomeproteasomemediated HIF degradation Examples: prolyl hydroxylase inhibitors FGFG-2216 and FG FG--4592 NOTE: FDA has suspended any further clinical trials with HIF stabilizers, as a female patient developed fatal hepatic necrosis temporally related to the introduction of FGFG-2216 Recommended Target Hb levels Target/dosage EBPG II 2004 KDOQI 2006 Authors recommendation Definition of Anemia by Hb (g/dl) < 12. 12.0 males < 11 11..5 females < 13. 13.5 males < 12. 12.0 females n.a. Target Hb (g/dl) > 11 11..0; Hb > 14. 14.0 not desirable in HD patients 11 11..0 – 12. 12.0; caution when maintaining Hb >13 13..0 10 10..5 – 12 12..0; avoid Hb >13 13..0 Target Iron Status TSAT (%) Ferritin (ng/ml) TSAT: lower limit 20, TSAT: 20, target 3030-50 Ferritin:: lower limit 100, Ferritin 100, target 200200-500 TSAT: n.a. TSAT: Ferritin:: lower limit 200 Ferritin 200,, >500 not routinely recommended TSAT: ≥ 20 TSAT: Ferritin:: lower limit 200 Ferritin 200,, target 300300-500, 500, >800 not recommended Recommended EPO starting dose 5050-150 IU/kg per wk (≈ 4000 4000--8000 IU per wk) no specifically starting dose, but individualized dosing recommended 100-125 IU/kg per wk 100(≈ 9000 IU per wk) Definition of large EPO requirement 300 IU/kg per wk (≈ 20. 20.000 IU per wk) 450 IU/kg per wk i.v. (≈ 30. 30.000 IU per wk) 375 IU/kg per wk (≈ 24 24..000 IU per wk) adapted from Schmid H. et al. Cardiovasc Hematol Agents Med Chem. 2010 NOTE: KDIGO Clinical Practice guideline for Anemia expected in early 2012 ESA intervention in CKD Selection of randomized controlled trials ► Besarab et al al.. NEJM 1998 Normal Haematocrit trial Patients:: 1233 Patients 1233,, ESRD treated with HD + clinical evidence of CHF or IHD Study Design Design,, arms /primary endpoint: endpoint: iv / sc Epoetin alfa; alfa; High: High: target Hct 42 ± 3 (≈ Hb 14 14)); Low: Low: target Hct 30 ± 3 (≈ Hb 10 10)); EP: EP: composite of death and 1st non non--fatal MI (time to 1st event) Main Results: Results: Study terminated early (futility, safety concerns) concerns);; more patients in the higher arm reached the endpoint (n (n..s.); physical function score increased with Hct; Hct; incidence of vascular access thrombosis higher in high arm (243 vs. vs. 176; 176; р= р=0 0.001) 001) ► Singh et al al.. NEJM 2006 CHOIR Patients:: 1432 Patients 1432,, eGFR 15 15--50 50,, Hb < 11 Study Design Design,, arms /primary endpoint: endpoint: sc Epoetin alfa: alfa: High: High: target Hb 11..3: EP: EP: composite of 4 CV events (time to 1st event) 13 13..5; Low: Low: target Hb 11 Main results: results: Study terminated early (futility, safety ?) ?);; more patients in the higher arm had at least one CV event; event; no improvement in QOL; QOL; trend towards a higher rate of progression to RRT in high arm ESA intervention in CKD Selection of randomized controlled trials ► Drüeke et al. NEJM 2006 CREATE Patients: 603, eGFR 1515-35, Hb 1111-12.5 Study Design, Design, arms /primary endpoint: sc Epoetin beta; High: High: target Hb 13.013.0-15.0; 15.0; Low: Low: when Hb < 10.5→ 10.5→ target 10.510.5-11.5; EP: EP: composite of 8 CV events (time to 1st event) Main Results: Results: No difference in primary EP; EP; improvement in QOL; QOL; time to dialysis shorter in higher arm ► Pfeffer et al al.. NEJM 2009 TREAT Patients:: 4038 Patients 4038,, CKD not undergoing HD + type 2 diabetes + anemia Study Design Design,, arms /primary endpoint: endpoint: sc darbepoetin alfa; alfa; High: High: Hb 13 13..0; Low: Low: rescue when < 9.0; EP: EP: composite outcomes of death or 4 CV events and of death or ESRD Main Results: Results: No difference in primary EP: EP: only modest improvement in QOL;; Darbepoetin alfa rescue associated with increased risk of stroke QOL (р< (р<0 0.001) 001); nonsignificant trend towards cumulation of cancercancer-related adverse events in darbepoetin alfa group (6.9% vs. vs. 6.4%, p= p=0 0.53 53)) ESA intervention in CKD summary ► Quality Quality--ofof-life improvements maximize in target Hb 10– 10–12 g/dl (Palmer SC Annals of Int Med 2010) ► Following the findings of TREAT → Second position statement/paper of the European Renal Best Practice (ERBP) Anemia Working Group (Locatelli F et al. NDT 2010) 'Hb values of 1111-12 g/dL should be generally sought in the CKD population without intentionally exceeding 13 g/dL' doses of ESA therapy to achieve target Hb should also be considered Important risks of ESA use ► Hypertension ► Thrombosis ► Anti Anti--EPO antibody mediated pure red cell aplasia (PRCA) ► ESA ESA--associated Tumor progression ESA--induced Hypertension ESA ► In 25--35 % of rHu EPO treated patients, 25 particularly in those with use of titration doses ► Additional antihypertensive therapy often required, but discontinuitation of rHU EPO normally not needed (Exceptional case: hypertensive encephalopathy!) ► aetiology incompletely understood ► probably multifactorial ► NOTE: attaining a normal haemoglobin does not confer an added risk for hypertension ESA--induced Thrombosis ESA ► ► ESA treatment increases hematocrit and blood viscosity → increased risk for vascular or graft thrombosis United States Normal Haematocrit trial (Besarab et al. NEJM 1998, 1233 HD patients) Higher incidence of access thrombosis with normal Hb levels ► ► ► rHU EPO treatment associated with increased thrombosis events in arteriovenous grafts but not in fistulas (Churchill DN et al. JASN 1994) NOTE: Smaller studies did not report an increased incidence of access thrombosis! Increase in thrombotic events has been noted in patients with malignancy who have received ESA ESA--induced PRCA ESA ► ► ► Associated with profound fall in Hb level and reticulocyte count Diagnosis bone marrow aspirate with demonstration of severe erythroid hypoplasia Anti--EPO antibodies demonstrated by radioimmunoprecipitation Anti assay (higher sensitivity and specifity than ELISA) PRCA management Transfusions for symptomatic anemia and ESA discontinuation Immunosuppressive therapy (corticosteroids ± i.v. immunoglobulins, corticosteroids + oral cyclophophamide or cyclosporine alone) s.c. injection of hematide NOTE: NOTE: Optimal duration of therapy and subsequent monitoring for antiantiEPO antibody mediated PRCA currently unknown! ESA--induced Tumor progression ESA ► ► ► Cancer patients Observational trials showed more rapid cancer progression and reduced survival in ESA treated patients (e.g. head and neck cancer, breast cancer patients) Renal patients no evidence that treatment of renal anemia results in higher rates of malignant tumors stable cancercancer-specific one one--year death rates among prevalent HD patients from 1991 to 2005, arguing for no increase since ESA introduction in 1989 NOTE: TREAT study (Pfeffer et al. NEJM 2009) revealed a nonsignificant trend towards cumulation of cancercancer-related adverse events in the darbepoetin alfa group (6.9% vs. 6.4%, p=0.53)! ESA hyporesponsiveness ► ► Definition: Definition: (i) requirement of excessive doses during initiation of ESAESAtherapy, or (ii) inability to achieve or maintain target Hb levels of 11 g/dl despite large EPO doses in ironiron-replete patients Recommendations to achieve HB of 10 - 11 g/dl NKF-DOQI Clinical Practice Guidelines NKFGuidelines:: 450 U/kg/wk i.v. EPO or 300 U/kg/wk s.c. EPO revised European Best Practice Guidelines Guidelines:: 300 U/kg/wk EPO (~ 20 20..000 U/wk) or 1.5 mcg/kg/wk darbepoetin alfa (~ 100 mcg/wk) ► NOTE: Despite high doses of ESA approximately 5 - 10 % NOTE: of ESRD patients show a less than satisfactory response ESA hyporesponsiveness ► Major causes of EPO resistance iron deficiency, infection or inflammation, underdialysis Self--administering patients: poor adherence Self Exclude Vitamin B12, folate, thyroxin deficiency and severe hyperparathyroidism Aluminum toxicity no longer significant cause of partial ESA resistance ► ► ► Depending on ethnic origin hemoglobinopathy should be excluded by Hb electrophoresis Some patients on ACE inhibitors/angiotensin II Receptor antagonists may require higher ESA doses NOTE: Primary bone marrow disorders should be investigated by bone marrow investigation only if all other causes have been excluded Challenges in clinical research ► Large Large--scale randomized clinical trials comparing anemia management strategies to assess safety, efficacy, and cost ? ► Do ESA doses rather than targeted Hb itself mediate increased risk for adverse vascular outcomes in ESRD ? ► Influence of fixed ESA doses and frequency of ESA administration ? Challenges in clinical research ► Evaluation of ESA responsiveness and Hb variability in association with mortality: is the degree of hematopoietic responsiveness to ESA treatment and not Hb target critical ? ► Contribution of Hepcidin to severity of anemia and resistance to ESAs ? ► Candidate biomarkers for ESAESA- hyporesponders ? ► Impact of pharmacological interventions targeted to prevent or reduce LVH in anemic or hypertensive CKD patients ? Challenges in clinical research ► Harm associated with more intensive ESA dosing due to higher Hb, higher ESA doses or both ? ► Contribution of intrinsic disease (coexisting illness, glycemic control in diabetic patients) or extrinsic factors ? ► More individualized ESAESA-treatment strategies needed ? Conclusions I ► ESAs have become the mainstay of anemia treatment in ESRD ► Traditional recombinant human erythropoietin, second generation erythropoietin analogues including darbepoetin alfa and C.E.R.A. as well as "biosimilar" erythropoietins currently available ► Search goes on for orally active antianaermic drugs ► In ESRD observational studies revealed a strong positive correlation between severity of anemia and risk of poor outcome Conclusions II ► ► ► ► ► In ESRD correction of renal anemia under ESA treatment reduced morbidity, mortality and hospitalization Integration of ESA and i.v. iron therapy into standard anemia management resulted in target Hb levels in vast majority of ESRD patients Optimal Hb target, dosing algorithm, and monitoring approach for anemic ESRD patients currently unclear Mortality data suggest that treating CKD patients to a Hb level >13 g/dl can be harmful Target Hb levels of 1010-12 g/dl seem reasonable, but increasing ESA doses in hyporesponsive patients to achieve a specific target is not Conclusions III ► ► ► ► ► Each patient should be treated according to a Hb target with the lowest effective ESA dose Avoid large fluctuations in Hb levels or prolonged periods outside the Hb target Most important complications in ESA treated patients are hypertension, thrombosis, and PRCA Despite great success of ESAs in clinical practice, ESAESAresistance and ESAESA-hyporesponsiveness are common clinical problems Large--scale randomized clinical trials comparing anemia Large management strategies are urgently needed, as adaption of the Framingham approach seems not feasible in the CKD patient
© Copyright 2026