Link to comlete article
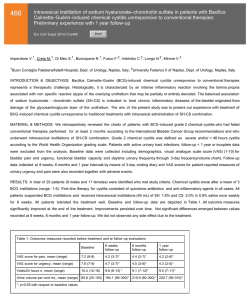
Micromedic Reports Successful Clinical Study Results for Monitoring Bladder Cancer Recurrence with the CellDetect® Non-Invasive Test - Plans to Secure CE Mark for a European Launch of the Non-Invasive Test and Submit a Pre-IDE to the U.S. FDA in 2015 – TEL-AVIV (February 2, 2015) - Micromedic Technologies Ltd. (TASE: MCTC), announced today the results of a blinded, multi-center clinical study using the CellDetect® non-invasive technology to detect bladder cancer recurrence in patients with a history of the disease. The CellDetect® test successfully identified cancerous cells in urine samples, with reported sensitivity of 84.4% and specificity of 82.7% for the study’s primary endpoint. The blinded clinical study was conducted in nine medical centers, where urine samples from 217 subjects with a history of bladder cancer were tested. The study population included 121 healthy subjects and 96 patients currently suffering from the disease. The results of the CellDetect® urine test were compared with results from biopsy or cystoscopy, in cases where biopsies were not performed. The results also indicated that the negative predictive value (NPV) was 98.5%. In addition to its high sensitivity for advanced stage tumors and high-grade malignancy, the test was also found to exhibit high sensitivity for early stage tumors and low-grade malignancies which are difficult to identify using other non-invasive tests currently available on the market. The secondary endpoint showed that the sensitivity of other non-invasive comparator tests, urine cytology, BTA stat and NMP22 BladderCheck, was 50.0%, 68.8% and 17.4%, respectively. These findings indicate that the method is adequately sensitive for the purpose of accurate and early detection of the recurrence of the disease. Following these successful study results, the company plans to secure a CE mark approval for a European launch of this non-invasive test later this year as well as to submit a Pre-IDE to the U.S. Food and Drug Administration (FDA). “We are tremendously pleased with the results of the study that confirm the high performance of the CellDetect® urine test in accurately monitoring recurrence of bladder cancer" Steven Eitan, Micromedic’s CEO, said. “By administering this test, millions of bladder cancer patients may be able to forego numerous costly and invasive tests, starting to receive treatment faster if their cancer is likely to recur. Following these successful results, we are also planning to conduct the required activities to quickly broaden the intended use to the early detection diagnostic market for additional millions of patients . The CellDetect® technology has the potential for diagnosing additional cancer indications.” Prof. Ofer Yossepowitch. M.D., Head of the Uro-Oncology Service at Rabin Medical Center: “The study results are encouraging. The accuracy of this novel assay appears 2 to be superior over any available non-invasive test, suggesting a potential to supplant some or all of the cystoscopies required for bladder cancer surveillance. This is indeed great news for patients with history of bladder cancer, which may change their management.” About CellDetect® Micromedic’s CellDetect® technology allows an accurate diagnosis of cancerous and precancerous cells based on unique combination of color and morphology. The technology may be implemented in screening and monitoring cancer patients. Micromedic has proven the product’s efficacy in diagnosing cervical cancer and bladder cancer in the framework of clinical trials, and estimates that the technology underlying the products may be implemented for use in additional cancer indications. The cervical cancer detection screening diagnostic test kit is in the initial commercial stage and Micromedic recently completed a clinical trial to prove its ability to monitor bladder cancer recurrence. Micromedic believes that the underlying technology may be adapted for other types of cancer as well. About Bladder Cancer Bladder cancer is the fourth most prevalent cancer among males in the U.S and seventh globally with nearly 430,000 new cases of the disease diagnosed worldwide in 2012. The rate of recurrence is the highest of all cancers and ranges from 50% to 80%. Because of high recurrence rates, the cost of diagnosing and treating bladder cancer is among the highest of all cancers. About Micromedic Micromedic is engaged in the development and commercialization of unique solutions addressing a real need prevailing in the field of cancer diagnostics including early detection of cancers. For more information please visit the Company’s website at www.m-medic.com CONTACTS: Micromedic Steven Eitan, CEO ([email protected]) Tel: +972-73-2753450
© Copyright 2026