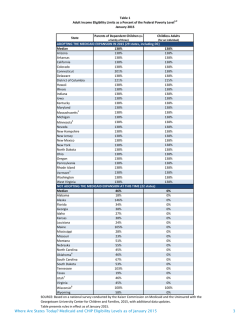
Prescription Drug Program Prior Authorization Criteria
Prescription Drug Program Prior Authorization Criteria Revised 2/03/2015 This document is an informational listing of the medications requiring a Prior Authorization through the Arkansas Medicaid Pharmacy Program, and a description of the associated criteria. Inclusion in this document does not guarantee market availability and products must meet the Centers for Medicare and Medicaid Services (CMS) definition of a covered outpatient drug and pay CMS rebate to be covered by Arkansas Medicaid. Select covered over the counter medications are covered pursuant to a valid prescription, but are not covered for Long Term Care eligible beneficiaries. Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Prescription Product or Drug Class 5-HT3 or NK1 Receptor Antagonists Abiraterone Acetate Tablet (Zytiga) Acetic Acid HC Ear Drops Acitretin Capsule (Soriatane) Aclidinium Bromide Inhaler (Tudorza Pressair) Acthar HP Gel Injection Actonel Acyclovir (Xerese and Zovirax Cream) Acyclovir Orally Disintegrating Delayed Release Tablet (Sitavig) Adempas (Riociguat) Tablet Adoxa Afatinib Dimaleate Tablet (Gilotrif) Afinitor Alagesic Liquid Oral Solution 50-325-40/15ml Albuterol Oral Tablets and Syrup Aldara Allergan Extracts Altabax 1% Ointment Amitiza Ampyra ER Angiotensin II Receptor Antagonists Anoro Ellipta Antibiotic Ophthalmic Drops (Moxeza,Vigamox, Quixin, Iquix, Azasite, Zymar) Antibiotic-Steroid Fixed-Dose Combination Ophthalmic Drops (Cortisporin, Tobradex ST, Zylet) Antibiotics, Long-acting Antidepressant, Second Generation (SGAD) Antidiabetic Agents Anti-Inflammatory Ophthalmic Drops Anti-Inflammatory Agents, Nonsteroidal Antihistamine Eye Drops, Mast Cell Stabilizers, and Combination Mast Cell Stabilizer-Antihistamine Eye Drops Antihistamines, Second Generation Antifungal Creams (Topical), Lotions, and Shampoos Antipsychotics, Injectable Long-acting Antipsychotics, Oral Aptiom Aranesp Armodafinil (Nuvigil) Aromatase Inhibitors (Femara Tablets and Arimidex) Astragraf XL Attention Deficit (ADD/ADHD) Agents for Children (Age Less than 18 Years) Attention Deficit (ADD/ADHD) Agents for Adults (Age 18 Years or greater) Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Auranolfin (Ridaura) Axitinib Tablet (Inlyta) Azelaic Acid 15% Gel (Finacea) Azithromycin (Azithromycin Powder Packets and ZMAX Suspension) Aztreonam Inhalation Solution (Cayston) Baraclude Becaplermin (Regranex) Bedaquiline Fumarate (Sirturo) Belimumab (Benlysta) Benign Prostatic Hypertrophy (BPH) Drugs Benzodiazepine Solid Oral Dosage Forms Benzodiazepine Liquid Oral Dosage Forms Benzoic Acid 6%, Salicyclic Acid 3%, Oak Bark Extract (Bensal HP) Ointment Beta Adrenergic Blocking Agents Beta2 Agonists/Corticosteroids, Inhaled Bethkis Bexarotene Gel (Targretin) BiDil Binosto Boniva Bosutinib (Bosulif) Botox Bowel Prep Kits Bronchodilators, Long-Acting Bronchodilators, Short-Acting Budesonide EC Capsule (Entocort EC) Buprenorphine (Subutex) Buprenorphine-Naloxone (Suboxone) Buproprion (Zyban) Butalbital Products C1 Esterase inhibitor (Cinryze) CII Stimulants Calcipotriene Cream, Foam, Ointment, or Scalp Solution (Dovonex, Sorilux) Calcipotriene and Betamethasone Dipropionate Ointment (Taclonex) Calcitonin-Salmon Nasal Spray (Miacalcin or Fortical) Calcitonin-Salmon Injection (Miacalcin) Calcitriol (Vectical) Calcium Channel Blockers Carbidopa (Lodosyn) Carbidopa/Levodopa/Entacapone (Stalevo) Cephalexin 750mg Capsule (Keflex) Cephalosporins – 3rd Generation Ceritinib (Zykadia) Chlorpheniramine ER 12mg Ciclopirox 8% Kit Cidofovir (Vistide) Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Cinryze Ciprofloxacin Extended-Release (Cipro XR) Clobazam (Onfi) Clonazepam Oral Disintegrating Tablet Clonidine Clonidine Vials Colchicine 0.6mg Tablets (Colcrys) Cometriq COPD Agents Corticosteroids, Nasal Inhaled Corticosteroids, Oral Inhaled Corticosteroids, Topical Crizotinib (Xalkori) Cromolyn Sodium Oral Solution (Gastrocrom) Crotamiton Cream and Lotion (Eurax) Cuprimine Cyclosporine 0.05% Eye Emulsion (Restasis) Cyproheptadine 4mg/10ml U.D. Cup Cysteamine Opthalmic Drops (Cystaran) Cysteamine DR Oral Capsule (Procysbi) Dabigatran Etexilate Mesylate (Pradaxa) Dabrafenib (Tafinlar) Dalfampridine ER (Ampyra ER) Daliresp Dasatinib (Sprycel) Tablet Deferiprone (Ferriprox) Tablet Denosumab (Prolia) Depen Desmopressin Nasal Spray/Solution (DDAVP) Dexamethasone Dose Pack (Dexpak and Zema-Pak) Dextromethorphan Hbr/Quinidine Capsule (Nuedexta) Diclegis DR 10-10 Dihydroergotamine Mesylate Nasal Spray (Migranal) Direct Renin Inhibitors Disopyramide CR (Norpace CR) Dornase Alfa Inhalation Solution (Pulmozyme) Doryx Dorzolamide-Timolol 2%-0.5% PF (Cosopt PF) Doxepin 5% Cream (Prudoxin, Zonalon) Doxycycline Doxylamine Succinate 5mg Chewable Tablet (Aldex AN) Doxylamine Succinate 10mg and Pyridoxine 10mg (Diclegis DR 10-10) Dronabinol (Marinol) Droxidopa (Northera) Capsule Efinaconazole 10% Solution (Jublia) Eliglustat (Cerdelga) Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Eliquis Emtricitabine (Emtriva) Entacapone (Comtan) Enzalutamide (Xtandi) Eslicarbazepine (Aptiom) Etanercept (Enbrel) Entecavir (Baraclude) Entocort EC Epogen Erythropoiesis Stimulating Agents Estrogen Replacement Agents Everolimus (Afinitor) Exelon Famotidine Oral Suspension (Pepcid) Febuxostat Tablet (Uloric) Fenofibrate and Fenobibric Acid Products Fentanyl Buccal Tablet (Fentora and Onsolis) Fentanyl Nasal Spray (Lazanda) Fentanyl Citrate Sublingual (Abstral) Fentanyl Citrate Sublingual Spray (Subsys) Fentanyl Citrate Oral Transmucosal (Actiq) Fibromyalgia Agents Fidaxomicin (Dificid) Finasteride Tablet (Proscar) Flagyl ER Fluorouracil 0.5% Cream (Carac) Fluorouracil 1% Cream (Fluoroplex) Fluorouracil 2% and 5% Solution (Efudex) Fluorouracil 5% Cream (Efudex) Fosamax Oral Solution Fosamax Plus D Fosamprenavir (Lexiva) Tablet Fosamprenavir (Lexiva) Suspension Forteo Fulyzaq Galantamine ER (Razadyne ER) Galantamine Solution (Razadyne) Gattex Giazo Glycerol Phenylbutyrate Oral Liquid (Ravicti) Glycophos Glycopyrrolate 0.2mg/ml vial Glycopyrrolate 1.5mg Tablet (Glycate) Guanfacine Hemorrhoid Preparations Hepatitis C Virus Medications Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria HMG-CoA Reductase Inhibitors Hydrocodone ER (Zohydro ER) Capsule Hydroxypropyl 5mg Eye Insert (Lacrisert) Ibrutinib (Imbruvica) Capsule Icatibant Syringe (Firazyr) Iclusig Idelalisib (Zydelig) Tablet Imiquimod (Aldara) Imiquimod (Zyclara) Immunologic Agents (Multiple Sclerosis) Infergen Ingenol Mebutate (Picato Gel) Inhaled Corticosteroid (ICS)/Long-Acting Beta Agonist (LABA) Combination Products Insulin Pens Isosorbide Dinitrate/Hydralazine HCl (BiDil) Isotretinoin (Accutane) Itraconazle 200mg Tablet (Onmel) Itraconazole Oral Solution (Sporanox) Ivacaftor Tablet (Kalydeco) Ivermectin 0.5% Lotion (Sklice) Juxtapid Keppra ER Ketorolac (Toradol) Kits Korlym Kynamro Lamotrigine Start Kits (Lamictal Start Kits) Lansoprazole, Amoxicillin, and Clarithromycin Combination (Prevpac) Leukotriene Receptor Antagonists Levetiracetam ER (Keppra ER) Levofloxacin 500mg/20ml U.D. Cup Levothyroxine Capsule (Tirosint) Levothyroxine Vial Lexiva Oral Suspension Lexiva Tablets Lidocaine 5% Patch (Lidoderm) Lidocaine-Prilocaine2.5%-2.5% Topical Cream (Emla) Linacoltide (Linzess) Lithium ER Lithobid SA Lindane Lovaza Low Molecular Weight Heparins Lubiprostone (Amitiza) Lupaneta Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Lupron Malathion (Ovide) Macitentan (Opsumit) Marinol Maraviroc (Selzentry) Mecamylamine HCL Tablet (Vecamyl) Mechlorethamine HCL Gel (Valchlor) Medroxyprogesterone (Depo-Provera) Megestrol (Megace and Megace ES) Mekinist Memantine XR (Namenda XR) Memantine Solution (Namenda) Meprobamate (Equanil) Tablet Mepron Mercaptopurine (Purixan) Mesalamine 1000mg (Canasa) Mestinon Timespan Methoxsalen (Oxsoralen-Ultra, 8-MOP) Metreleptin Vial (Myalept) Metformin Oral Solution(Riomet) Metformin ER (Fortamet ER and Glumetza ER) Methscopolamine (Pamine, Pamine Forte, Pamine FQ) Methotrexate Auto Inj. (Otrexup) Methotrexate (Trexall) Methylnaltrexone Bromide (Relistor) SQ Injection Metozolv Metronidazole 1% Cream (Noritate) Metronidazole 1% Gel (Metrogel) Metronidazole 375mg (Flagyl) Metronidazole ER 750mg (Flagyl) Metronidazole/Tetracycline/Bismuth (Helidac and Pylera) Miconazole 50mg Buccal Tablet (Oravig) Miconazole Nitrate/ Zinc Oxide/White Petrolatum (Vusion) Mifepristone (Korlym) Mig Lustat (Zavesca) Migranal Nasal Spray Millipred Minocycline Mirapex ER Misoprostol (Cytotec) Modafinil (Provigil) Moxatag Multiple Sclerosis Agents Mycophenolate (Cellcept, Myfortic) Nabilone (Cesamet) Nafarelin Nasal Spray (Synarel) Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Naloxone Auto-injector (Evzio) Namenda Namenda XR Nandrolone Decanoate Injection Neuropathic Pain Agents Nevirapine XR (Viramune XR) Nevirapine Oral Suspension (Viramune) Nicotine Replacement Therapy Nimodipine Solution (Nymalize) Nitisinone (Orfadin) Nitrofurantoin Suspension (Furadantin) Nitroglycerin 0.4% Rectal Ointment (Rective) Nizatadine Oral Solution (Axid) Non-Benzodiazepine Sedative Hypnotics Nonsteroidal Anti-Inflammatory Agents Norpace CR Noxafil Suspension Noxafil DR Tablet Noxafil Vial Nucort Nuzon Gel Omega-3-acid ethyl esters (Lovaza) Omeprazole/Clarithromycin/Amoxicillin (Omeclamox-Pak) Onabotulinumtoxina (Botox 200 Units) Onfi Opioids, Long-Acting Opioids, Short-Acting Opium Tinture Opsumit (Macitentan) Oracea Orapred ODT Tablets Oseltamivir Suspension (Tamiflu Suspension) Osteoporosis Drugs Otic Preparations Otrexup Overactive Bladder Agents Oxandrolone (Oxandrin) Oxymethalone (Anadrol-50) Pain Medications, Injectable Papaverine Injection Pasireotide Diaspartate (Signifor) Pazopanib (Votrient) Peg Intron Pegasys PegInterferon Alfa-2B (Sylatron) Pegvisomant Injection (Somavert) Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Penciclovir (Denavir) Penicillamine (Depen 250mg Tablet and Cuprimine 250mg Capsule) Perampanel Tablet (Fycompa) Phosphate Removing Agents Pimecrolimus (Elidel) Podofilox (Condylox 0.5% Topical Solution/Gel) Pomalyst Posaconazole (Noxafil) Potassium Chloride Capsule and Packet Pradaxa Pramipexole ER (Mirapex ER) Prednisolone Prednisone (Rayos DR) Procrit Procysbi Prolia Promacta Propafenone SR (Rythmol SR) Proquin Proton Pump Inhibitors Pyridostigmine Timespan (Mestinon Timespan) Quick Relief Medications for Asthma Quinine Sulfate (Qualaquin) Raloxifene (Evista) Ranolazine (Ranexa) Razadyne ER Razadyne Solution Rebetol Ravicti Regorafenib (Stivarga) Requip XL Respiratory Syncytial Virus (RSV) Medications Restasis Ribapak Ribasphere Ribavirin Ridaura Rifaximin Tablet (Xifaxan) Rilonacept Sub-Q Vial (Arcalyst) Riociguat (Adempas) Tablet Rivaroxaban10mg Tablet (Xarelto) Rivaroxaban 15mg and 20mg Tablet (Xarelto) Rivastigmine Soution (Exelon) Solution Ropinirole XL (Requip XL) Rosacea Treatment Rotigotine (Neupro) Patch Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Ruxolitinib (Jakafi) Rythmol SR Sapropterin HCL (Kuvan) Scopolamine Transermal Patch Sedative Hypnotics Serostim Serotonin 5-HT1 Receptor Agonists Signifor Sildenafil (Revatio) Siltuximab Vial (Sylvant) Sinecatechins (Veregen Ointment 15%) Skeletal Muscle Relaxants Sodium Chloride 7% Inhalation Solution (Hyper-Sal 7%) Sodium Oxybate (Xyrem) Somatropin (Genotropin) Somatropin (Humatrope) Somatropin (Norditropin) Somatropin (Nutropin) Somatropin (Omnitrope) Somatropin (Saizen) Somatropin (Tev-tropin) Somatropin (Zorbtive) Soriatane Spinosad 0.9% Topical Suspension (Natroba) Spiriva Suboxone Subutex Sucralfate Suspension (Carafate) Sulfamethoxazole-Trimethoprim U.D. Cup Sunitinib (Sutent) Symbicort Synagis Syprine Tacrolimus (Astagraf XL) Tacrolimus (Protopic) Tadalafil (Adcirca) Tafinlar Tamiflu Suspension Tamoxifen Solution (Soltamox) Targeted Immune Modulators Targretin Tasimelteon (Hetlioz) Tavaborole (Kerydin) Tazarotene Gel/Cream (Tazorac) Tedizolid (Sivextro) Telithromycin (Ketek) Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Temazepam 7.5mg and 22.5mg Capsule (Restoril) Tenofovir (Viread) Tenofovir/Emtricitabine (Truvada) Terbinafine (Lamisil) Terbinex Teriparatide (Forteo) Testosterone Replacement Products Tetrabenazine (Xenazine) Timolol Maleate/Dorzolamide HCL (Cosopt PF) Tiotropium Bormide Inhaler (Spiriva) Tirosint Tobacco Cessation Products Tobramycin Inhalation Solution (TOBI - Brand) Tobramycin Inhalation Solution (Generic) (Bethkis) Tobramycin Inhalation Powder (TOBI Podhaler) Topical Corticosteroids Topical Products Topiramate XL (Qudexy XL or Trokendi XL) Tramadol ER Tramadol IR Tramadol-Acetaminophen (Ultracet) Trametinib (Mekinist) Tranexamic Acid (Lysteda) Transdermal Scopolamine Patch Trazodone (Trazodone 300mg, Oleptro ER) Trexall Trientine HCl (Syprine) Tudorza Pressair Inhaler Ulesfia Uloric Valchlor Valcyte Solution Varenicline (Chantix) Vascepa Vecamyl Vemurafenib (Zelboraf) Veregen Veripred Solution Viramune XR Viramune Oral Suspension Vismodegib Capsule (Erivedge) Vorapaxar (Zontivity) Votrient Xarelto 10mg, 15mg & 20mg, Starter Pack Xgeva Xyrem Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Xolair Zohyrdro ER Appendix A – Nil per os (NPO) Appendix B – Approved Tracheostomy Codes Appendix C – Topical Corticosteroids, No PA Required Appendix D – Congestive Heart Failure Diagnoses Appendix E – Malignant Cancer Diagnoses Appendix F – Antineoplastics to Infer Malignant Cancer Appendix G – Chronic Obstruction Pulmonary Disease Diagnoses Appendix H – Approved Diagnoses for nonpreferred Antiepileptic Agents in Neuropathic Pain Agent Class Appendix I – Approved Endoscopy Code Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local.) The Arkansas Medicaid Preferred Drug List may be found at this link. https://www.medicaid.state.ar.us/Download/provider/pharm/PDL.xls For assistance on all other drugs, prescribers may call the HP Pharmacy Unit at 1-800-707-3854 (toll-free) or 501-374-6609 x500 (local.) The appropriate number is indicated with the associated drug. Please refer to the Arkansas Medicaid Webpage at www.medicaid.state.ar.us for a complete list of drugs. Other claim edits for age, gender, quantity, dose and/or cumulative quantity may apply. https://www.medicaid.state.ar.us/Download/provider/pharm/ClaimEdits.xls Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria 5-HT3 or NK1 Receptor Antagonists (Implemented 09/14/2009) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents with criteria Ondansetron HCl 4mg, 8mg tablet (Zofran) Ondansetron 4mg, 8mg oral-disintegrating tablet (Zofran ODT) Ondansetron 4mg/2ml preservative-free vial (Zofran) Ondansetron 40mg/20ml vial (Zofran) Nonpreferred agents Aprepitant (Emend) Dolasetron (Anzemet) Granisetron (Kytril, Sancuso) Netupitant-Palonosetron HCl (Akynzeo) Ondansetron 24mg tablet (Zofran) Ondansetron 32mg/50ml bag (Zofran) Ondansetron 4mg/2ml ampule and syringe (Zofran) Ondansetron 4mg/5ml solution (Zofran) Ondansetron Soluble Film (Zuplenz) Approval criteria for preferred agents with criteria No therapeutic duplication with other 5-HT3 receptor antagonists Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Abiraterone AcetateTablet (Zytiga) (Implemented 12/10/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Zytiga Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Acitretin Capsule (Soriatane) (Implemented 03/26/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Soriatane Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Aclidinium Bromide Inhaler (Tudorza Pressair) (Implemented 12/19/2012) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Approval criteria Diagnosis of COPD in Medicaid history in previous 2 years; AND No therapeutic duplication with overlapping days’ supply between Anoro Ellipta, Spiriva, and/or Tudorza; AND Medicaid recipient is > 18 years of age; AND Denial criteria Lack of approval criteria; OR Diagnosis of asthma in Medicaid history in previous 2 years Additional criteria Quantity edits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Acyclovir Cream, Ointment (Implemented 03/19/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Docosanol 10% (Abreva) cream Drugs that require manual review for prior authorization Acyclovir (Zovirax) 5% cream Acyclovir (Zovirax) 5% ointment Acyclovir-Hydrocortisone (Xerese) 5%-1% cream Penciclovir (Denavir) 1% cream 5 gram (Implemented 09/23/2014) Additional criteria Quantity edits apply Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Acyclovir Orally Disintegrating Delayed Release Tablet (Sitavig) (Implemented 09/23/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Sitavig Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Afatinib Dimaleate Tablet (Gilotrif) (Implemented 12/10/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Gilotrif Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Alagesic Liquid Oral Solution 50-325-40/15ml (Implemented 01/18/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria NPO (Appendix A) Age Edit Recipients must be 12 years of age or greater Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Albuterol Oral Tablets and Syrup (Implemented 03/18/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Albuterol 2mg/5ml Syrup Albuterol 4mg ER Vospire 4mg ER Albuterol 8mg ER Vospire 8mg ER Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Allergan Extracts, (short Ragweed Pollen Allergan Extract) and (Timothy Grass Pollen Allergen Extract) (Implemented 09/23/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Ragwitek Grastek Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Angiotensin II Receptor Antagonists (Implemented 10/01/2008) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents with criteria Diovan (Brand only) Irbesartan (Avapro) Irbesartan/HCTZ (Avalide) Losartan potassium (Cozaar) Losartan potassium/HCTZ (Hyzaar) Olmesartan medoxomil (Benicar) Olmesartan medoxomil/Amlodipine besylate (Azor) Olmesartan medoxomil/Amlodipine besylate/HCTZ (Tribenzor) Olmesartan medoxomil/HCTZ (Benicar HCT) Valsartan/Amlodipine (Exforge) Valsartan/Amlodipine/HCTZ (Exforge HCT) Valsartan/HCTZ (Diovan HCT) Nonpreferred agents Azilsartan medoxomil (Edarbi) Azilsartan medoxomil/Chlorthalidone (Edarbyclor) Candesartan cilexetil/HCTZ (Atacand HCT) Eprosartan mesylate (Teveten) Eprosartan mesylate/HCTZ (Teveten HCT) Telmisartan (Micardis) Telmisartan/Amlodipine (Twynsta) Telmisartan/HCTZ (Micardis HCT) Valsartan (generic Diovan) Approval criteria for preferred agent with criteria New Starts will require a paid claim in history of an ACE-Inhibitor product (ACEInhibitor, ACE-Inhibitor/HCTZ, or ACE-Inhibitor/Calcium Channel Blocker) in the previous 27-45 days OR Continuation of therapy will require at least one paid claim in history of a “Preferred with criteria” angiotensin receptor blocker or direct renin inhibitor in the past 60 days Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Nonpreferred agent with criteria Candesartan cilexetil (Atacand) Approval criteria for nonpreferred agent with criteria Candesartan cilexetil (Atacand) Congestive heart failure (Appendix D) Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Antibiotic Ophthalmic Drops (Implemented 08/21/2009) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria History of at least two claims for two different products that do not require prior authorization within the previous 30 days Gentamicin 3mg/ml eye drops, Sulfacetamide 10% eye drops, and Tobramycin 0.3% eye drops are available agents that do not require PA but are not included in this approval criteria. ANTIBIOTIC OPHTHALMIC DROPS GENERIC NAME COMMON TRADE NAME* PA STATUS Ciprofloxacin 0.3% eye drops Ciloxan® 0.3% eye drops No PA Neomycin-Polymyxin B-Gramicidin eye drops Neosporin® eye drops No PA Ofloxacin 0.3% eye drops Ocuflox® 0.3% eye drops No PA GENERIC NAME COMMON TRADE NAME* PA STATUS Azithromycin 1% eye drops Azasite® 1% eye drops PA Besifloxacin HCl 0.6% eye drops Besivance® 0.6% eye drops PA Gatifloxacin 0.5% eye drops Zymaxid® 0.5% eye drops PA Levofloxacin 0.5% eye drops Quixin® 0.5% eye drops PA Moxifloxacin 0.5% eye drops Moxeza® 0.5% eye drops PA Moxifloxacin HCl 0.5% eye drops Vigamox® 0.5% eye drops PA *TRADE NAMES ARE FOR REFERENCE ONLY Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Antibiotic-Steroid Fixed-Dose Combination Opthalmic Drops (Cortisporin, Tobradex ST, Zylet) (Implemented 10/11/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria History of at least two claims for two different products that do not require prior authorization within the previous 31 days o One claim must have been in the previous 14-31 days, AND o One claim must have been in the previous 5-13 days. ANTIBIOTIC OPHTHALMIC DROPS GENERIC NAME COMMON TRADE NAME* PA STATUS Gentamicin-Prednisolone eye drops Pred-G® 1% eye drops No PA Neomycin-Polymyxin B-Dexamethasone eye drops Maxitrol® eye drops No PA Sodium Sulfacetamide-Prednisolone eye drops Blephamide® eye drops No PA Sodium Sulfacetamide-Prednisolone eye drops Sulf-Pred® 10-0.23% eye drops No PA Tobramycin/Dexamethasone eye drops Tobradex® eye drops No PA GENERIC NAME COMMON TRADE NAME* PA STATUS Neomycin-Polymyxin B-Hydrocortisone eye drops Cortisporin® eye drops PA Tobramycin-Dexamethasone eye drops Tobradex ST® eye drops PA Tobramycin-Loteprednol eye drops Zylet® eye drops PA *TRADE NAMES ARE FOR REFERENCE ONLY Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Antibiotics, Long-acting Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). (Implemented 09/21/2009 and 8/17/2010) Drugs that do not require prior authorization Generic MAC’d short-acting antibiotics are available without a prior authorization. Drugs that require manual review for prior authorization Amoxicillin ER 775mg (Moxatag ER) Ciprofloxacin ER 500mg, 1000mg (Cipro XR, Proquin XR) Metronidazole ER 750mg (Flagyl ER) Link to Memorandum: Clarithromycin XL, Flagyl ER 750 mg Link to Memorandum: Removal of manual review for Clarithromycin XL Link to Memorandum: Ciprofloxacin ER, Proquin, Moxatag Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Second-generation Antidepressants (SGAD) (Implemented 01/01/2010) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents with criteria Bupropion HCl regular-release (Wellbutrin) Bupropion HCl extended-release (Wellbutrin XL) Bupropion HCl sustained-release (Wellbutrin SR) Citalopram hydrobromide (Celexa) Escitalopram oxalate (Lexapro) Fluoxetine HCl 10mg, 20mg capsule, and 20mg/5ml solution (Prozac) Fluvoxamine maleate (Luvox) Mirtazapine 15mg, 30mg, 45mg tablet (Remeron) Paroxetine HCl regular-release tablet (Paxil) Sertraline HCl (Zoloft) Venlafaxine HCl extended-release capsule (Effexor ER) Venlafaxine HCl regular-release tablet (Effexor) Nonpreferred agents with SGAD criteria Bupropion hydrobromide extended-release tablet (Aplenzin) Bupropion HCl exended-release tablet (Forfivo XL) Desvenlafaxine extended-release tablet (Khedezla ER) Desvenlafaxine fumarate extended-release tablet Desvenlafaxine succinate extended-release tablet (Pristiq ER) Duloxetine HCl (Cymbalta) Fluoxetine HCl 10mg, 15mg, 20mg Tablet; 40mg capsule; and 90mg weekly capsule (Prozac) Fluvoxamine maleate extended-release (Luvox CR) Milnacipran HCl (Savella) Mirtazapine 7.5 mg Tablet, and orally disintegrating tablet (Remeron SolTab) Nefazodone HCl (Serzone) Paroxetine HCl controlled-release tablet, and 10mg/5ml suspension (Paxil) Paroxetine mesylate (Pexeva) Vilazodone HCl (Viibryd) Vortioxetine HBr (Brintellix) Nonpreferred agents Levomilnacipran HCl extended-release tablet (Fetzima ER) Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Paroxetine mesylate (Brisdelle) Exempt SGAD combination agents with criteria Fluoxetine HCL/Olanzapine (Symbyax) Approval criteria for preferred or exempt agents Drug daily dose ≤ maximum daily dose (Table 1) Approval criteria for preferred or exempt agents resulting from a therapeutic duplication If applicable for a change in therapy or concomitant therapy of two agents and only one or neither are SSRIs and/or SSNRIs (including combinations) (Table 1.2): Drug in history reflects a minimal therapeutic dose (Table 1) for at least four weeks OR If applicable for a change in therapy for two SSRIs and/or SSNRIs (including combinations) (Table 1.2) Drug in history reflects a minimal therapeutic dose (Table 1) for at least four weeks, AND No prior therapeutic duplication for two different SSRIs and/or SSNRIs (including combinations) (Table 1.2) within the past 365 days. Approval criteria for all nonpreferred agents except milnacipran > 90 days of therapy in the previous 120 days for the same drug, strength, and daily dose with the denial exception of a therapeutic duplication between an SSRI and/or SNRI between incoming claim and history Denial criteria for all agents Preferred agents or exempt agents Therapeutic duplication of three agents Therapeutic duplication of two SSRIs and/or SSNRIs (including combinations) (Table 1.2) more than once per 365 days Nonpreferred drugs for patients who do not meet criteria of >90 days of therapy in the previous 120 days for the same drug, strength, and daily dose See Fibromyalgia agents for additional criteria on select second generation antidepressants Link to PDL Memorandum: Second Generation Antidepressants Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Table 1 – Minimum and maximum dose for second-generation antidepressants Drug Bupropion Citalopram Desvenlafaxine Duloxetine Escitalopram Fluoxetine Fluoxetine/olanzapine* Fluvoxamine Mirtazapine Nefazodone Paroxetine Sertraline Venlafaxine Minimal daily therapeutic dose 200mg 20mg 50mg 40mg 10mg 20mg 25mg 100mg 30mg 300mg 30mg 100mg 150mg Maximum daily dose 450mg 40mg 100mg 60mg 30mg 80mg 75mg 300mg 60mg 600mg 60mg (CR 62.5mg) 200mg 375mg * Minimum therapeutic dose and maximum dose based on SSRI component of the combination agent. Table 1.2 – Selective Serotonin (norepinephrine) Reuptake Inhibitors (combinations) SSRI, SSNRI or SSRI Combinations Citalopram Desvenlafaxine Duloxetine Escitalopram Fluoxetine Fluoxetine/olanzapine Fluvoxamine Paroxetine Sertraline Venlafaxine Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Antidiabetic Agents (Implemented 01/01/2009) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents Glimepiride (Amaryl) Glipizide (Glucotrol) Glipizide extended-release (Glucotrol XL) Glipizide/Metformin HCl (Metaglip) Glyburide (Diabeta, Glynase Prestab) Glyburide/Metformin HCl (Glucovance) Nateglinide (Starlix) Non-Preferred agents Albiglutide (Tanzeum) Alogliptin benzoate (Nesina) Alogliptin benzoate/Metformin HCl (Kazano) Alogliptin benzoate/Pioglitazone (Oseni) Canagliflozin (Invokana) Canagliflozin/Metformin (Invokamet) Dapagliflozin (Farxiga) Dapagliflozin/Metformin HCl (Xigduo XR) Dulaglutide (Trulicity) Empagliflozin (Jardiance) Exenatide (Byetta) Exenatide microspheres (Bydureon) Linagliptin (Tradjenta) Linagliptin/Metformin HCl (Jentadueto) Liraglutide (Victoza) Pioglitazone HCl (Actos) Pioglitazone HCl/Glimepiride (Duetact) Pioglitazone HCl/ Metformin HCl (Actoplus Met) Pramlintide Acetate (Symlin, SymlinPen) Repaglinide (Prandin) Repaglinide/Metformin HCl (Prandimet) Rosiglitazone (Avandia) Rosiglitazone/Glimepiride (Avandaryl) Rosiglitazone/Metformin HCl (Avandamet) Saxagliptin HCl (Onglyza) Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Saxagliptin HCl/Metformin HCl (Kombiglyze XR) Sitagliptin Phosphate (Januvia) Sitagliptin phosphate/Metformin HCl (Janumet) Sitagliptin phosphate/Metformin HCl extended-release (Janumet XR) Sitagliptin phosphate/Simvastatin (Juvisync) Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Anti-inflammatory Ophthalmic Drops (Implemented 01/12/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria History of at least two claims for two different products that do not require prior authorization within the previous 28 days ANTI-INFLAMMATORY OPHTHALMIC DROPS GENERIC NAME COMMON TRADE NAME* PA STATUS Dexamethasone Sodium Phosphate 0.1% eye drops Decadron® 0.1% eye drops No PA Diclofenac 0.1% eye drops Voltaren® 0.1% eye drops No PA Fluorometholone 0.1% eye drops FML Liquifilm® 0.1% eye drops No PA Fluorometholone 0.25% eye drops FML Forte® 0.25% eye drops No PA Flurbiprofen 0.03% eye drops Ocufen® 0.03% eye drops No PA Ketorolac 0.5% eye drops Acular® 0.5% eye drops No PA Ketorolac 0.4% eye drops Acular LS® 0.4% eye drops No PA Prednisolone acetate 1% eye drops Pred Forte® 1% eye drops No PA Prednisolone acetate 0.12% eye drops Pred Mild® 0.12% eye drops No PA Prednisolone sodium 1% eye drops AK-Pred® 1% eye drops No PA GENERIC NAME COMMON TRADE NAME* PA STATUS Bromfenac 0.07% eye drops Prolensa® 0.07% eye drops PA Bromfenac 0.09% eye drops Bromday® 0.09% eye drops PA Dexamethasone 0.1% suspension eye drops Maxidex® 0.1% suspension eye drops PA Difluprednate 0.05% eye drops Durezol® 0.05% eye drops PA Fluorometholone 0.1% eye drops Flarex® 0.1% eye drops PA Ketorolac 0.45% eye drops Acuvail® 0.45% eye drops PA Loteprednol 0.2% eye drops Alrex® 0.2% eye drops PA Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Loteprednol 0.5% eye drops Lotemax® 0.5% eye drops PA Loteprednol 0.5% eye gel drops Lotemax® 0.5% eye gel drops PA Nepafenac 0.1% eye drops Nevanac® 0.1% eye drops PA Nepafenac 0.3% eye drops Ilevro® 0.3% eye drops PA Rimexolone 1% eye drops Vexol® 1% eye drops PA *TRADE NAMES ARE FOR REFERENCE ONLY Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Nonsteroidal Anti-inflammatory Agents (Implemented 06/18/2007) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents Diclofenac sodium ER 100mg tablet (Voltaren XR) Ibuprofen 100mg/5ml suspension; 400mg, 600mg, 800mg tablet (Motrin) Indomethacin 25mg, 50mg capsule (Indocin) Ketoprofen 50mg, 75mg capsule (Orudis) Meloxicam 7.5mg, 15mg tablet (Mobic) Naproxen 250mg, 375mg, 500mg tablet (Naprosyn) Naproxen 375mg, 500mg enteric-coated tablet (EC-Naprosyn) Naproxen sodium 275mg and 550mg tablet (Anaprox) Salsalate 750mg (Salflex-750) Preferred agent with criteria Ketorolac tablet (Toradol) Nonpreferred agents Celecoxib (Celebrex) Diclofenac epolamine (Flector) Diclofenac potassium (Cambia, Cataflam, Zipsor) Diclofenac sodium 25mg, 50mg, and 75 mg tablet (Voltaren) Diclofenac sodium topical (Pennsaid, Voltaren Gel) Diclofenac sodium/Misoprostol (Arthrotec) Diclofenac submicronized (Zorvolex) Diflunisal (Dolobid) Etodolac (Lodine) Fenoprofen (Nalfon) Flurbiprofen (Ansaid) Ibuprofen 40mg/ml suspension; 50mg,100mg tablet (Motrin) Ibuprofen/caffeine/B1/B2/B6/B12 (IC400, IC800 Kit) Ibuprofen/famotidine (Duexis) Indomethacin 75mg SA Capsule; 50mg suppository; 25mg/5ml suspension (Indocin) Ketoprofen 200mg extended-release capsule (Oruvail) Ketorolac nasal spray (Sprix) Meclofenemate sodium (Meclomen) Mefenamic acid (Ponstel) Meloxicam suspension (Mobic) Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Nabumetone (Relafen) Naproxen/Esomeprazole magnesium (Vimovo) Naproxen Suspension (Naprosyn) Naproxen sodium 375mg and 500mg extended-release tablet (Naprelan) Oxaprozin (Daypro) Piroxicam (Feldene) Salsalate 500mg (Salflex-500) Sulindac (Clinoril) Tolmetin sodium (Tolectin) Nonpreferred agents with criteria Diclofenac Sodium 3% Gel (Solaraze) Naproxen 125mg/ml suspension (Naprosyn suspension) Approval criteria for nonpreferred agents with criteria Diclofenac Sodium 3% Gel (Soloraze) Diagnosis of Actinic Keratosis in the past two months Naproxen 125mg/ml suspension (Naprosyn suspension) < 6 years of age, OR NPO (Appendix A) Denial criteria for preferred agent with criteria Ketorolac o History of ketorolac use in the last 60 days, OR o NSAID claim in the past 30 days, OR o Dose greater than four per day, OR o Day supply greater than five, OR o Quantity greater than 20, OR o Greater than 20 units per 60 days Link to PDL Memorandum: NSAIDS Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Antihistamine Eye Drops, Mast Cell Stabilizers, and combination Mast Cell Stabilizer-Antihistamine Eye Drops (Implemented 01/12/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). EYE DROPS FOR ALLERGIC CONJUNCTIVITIS GENERIC NAME COMMON TRADE NAME* PA STATUS Azelastine HCl 0.05% eye drops Optivar® 0.05% eye drops No PA Cromolyn sodium 4% eye drops Cromolyn® 4% eye drops No PA Ketotifen fumarate 0.025% eye drops Alaway® 0.025% eye drops No PA Ketotifen fumarate 0.025% eye drops Zaditor® 0.025% eye drops No PA GENERIC NAME COMMON TRADE NAME* PA STATUS Alcaftadine 0.25% eye drops Lastacaft® 0.25% eye drops Manual PA Bepotastine besilate 1.5% eye drops Bepreve® 1.5% eye drops Manual PA Emedastine difumarate 0.05% eye drops Emadine® 0.05% eye drops Manual PA Epinastine HCl 0.05% eye drops Elestat® 0.05% eye drops Manual PA Lodoxamide tromethamine 0.1% eye drops Alomide® 0.1% eye drops Manual PA Nedocromil sodium 2% eye drops Alocril® 2% eye drops Manual PA Olopatadine HCl 0.1% eye drops Patanol® 0.1% eye drops Manual PA Olopatadine HCl 0.2% eye drops Pataday® 0.2% eye drops Manual PA Pemirolast potassium 0.1% eye drops Alamast® 0.1% eye drops Manual PA *TRADE NAMES ARE FOR REFERENCE ONLY Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Second-generation Antihistamines (Implemented 11//2007) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents Cetirizine HCl 1 mg/ml solution, 10 mg swallow tablet (Zyrtec) Loratadine (Claritin) Olopatadine HCl 6% nasal spray (Patanase) Nonpreferred agents Acrivastine w/Pseudoephedrine (Semprex-D) Azelastine HCL nasal spray (Astelin, Astepro) Azelastine/fluticasone nasal spray (Dymista) Desloratidine liquid (Clarinex) Nonpreferred agents with criteria Cetirizine Cetirizine 5 mg, 10 mg chewable tablet (Zyrtec) Cetirizine w/Pseudoephedrine (Zyrtec-D) Desloratadine Desloratadine tablet (Clarinex) Desloratadine w/Pseudoephedrine tablet (Clarinex-D) Fexofenadine Fexofenadine (Allegra) Fexofenadine w/Pseudoephedrine (Allegra-D) Levocetirizine (Xyzal) Approval criteria for nonpreferred agents with criteria All nonpreferred drugs with criteria > Four claims for the drug, strength and dosage form of any nonpreferred drug in the last six months Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Topical Antifungals (Implemented 09/21/2009) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria History of at least two claims for two different topical antifungal products containing different drug entities that do not require prior authorization within the previous 45 days. TOPICAL ANTIFUNGALS GENERIC NAME COMMON TRADE NAME* PA STATUS Ciclopirox 0.77% cream Loprox® 0.77% cream No PA Ciclopirox 0.77% topical suspension Loprox® 0.77% topical suspension No PA Clotrimazole 1% cream (OTC or Rx) Lotrimin AF® 1% cream No PA Clotrimazole 1% solution Lotrimin® 1% solution No PA Econazole nitrate 1% cream Spectazole® 1% cream No PA Ketoconazole 2% cream Nizoral® 2% cream No PA Ketoconazole 2% shampoo Nizoral® 2% shampoo No PA GENERIC NAME COMMON TRADE NAME* PA STATUS Butenafine HCl 1% cream Mentax® 1% cream PA Ciclopirox 0.77% gel Loprox® 0.77% gel PA Ciclopirox 1% shampoo Loprox® 1% shampoo PA Ketoconazole 2% foam Extina® 2% foam PA Ketoconazole 2% gel Xolegel 2% gel PA Luliconazole 1% cream Luzu® 1% cream PA Naftifine HCl 1% cream Naftin® 1% cream PA Naftifine HCl 1% cream Naftin® 1% pump cream PA Naftifine HCl 1% gel Naftin® 1% gel PA Naftifine HCl 2% cream Naftin® 2% cream PA Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Naftifine HCl 2% gel Naftin® 2% gel PA Oxiconazole nitrate 1% cream Oxistat® 1% cream PA Oxiconazole nitrate 1% lotion Oxistat® 1% lotion PA Sertaconazole nitrate 2% cream Ertaczo® 2% cream PA Sulconazole nitrate 1% cream Exelderm® 1% cream PA Sulconazole nitrate 1% solution Exelderm® 1% solution PA Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Antipsychotics, Injectable Long-acting (Implemented 01/12/2010) Prescribers may request an override by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866250-2518 (toll-free) or 501-526-4200 (local). Approval criteria All injectable long acting antipsychotics (Abilify MaintenaTM, fluphenazine decanoate, haloperidol decanoate, Risperdal® Consta®, Invega® Sustenna®, Zyprexa® RelprevvTM): Absence of denial criteria Age < 18 years requires Manual Review Additional criteria: Abilify MaintenaTM Three paid claims for oral AbilifyTM in the past 14-75 days at a dose of >/= 10 mg/day, OR One paid claim for Abilify MaintenaTM in the past 45 days Invega® Sustenna® Two paid claims for oral Invega® in the past 27-75 days, OR Two paid claims for oral Risperdal® in the past 27-75 days, OR One paid claim for Invega® Sustenna® in the past 45 days Risperdal® Consta® Two paid claims for oral Risperdal® in the past 27-75 days, OR Two paid claims for oral Invega® in the past 27-75 days, OR One paid claim for Risperdal® Consta® in the past 45 days Zyprexa® RelprevvTM Injection Two paid claims for oral Zyprexa® in the past 27-75 days, OR One paid claim for Zyprexa® RelprevvTM in the past 45 days Denial criteria All injectable long acting antipsychotics Therapeutic duplication with another long acting antipsychotic in the past 23 days Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Antipsychotics, Oral (Implemented 07/11/2009) Prescribers may request an override by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866250-2518 (toll-free) or 501-526-4200 (local). Approval criteria Patients ≥ 18 years of age Oral liquids and orally disintegrating tablets (ODTs): Patient must have an NPO code (Appendix A) in the past year Oral capsules/tablets are approved Seroquel XR requires >/= 90 days of Seroquel XR therapy in the past 120 days. Immediate-release quetiapine (Seroquel) is covered via existing criteria. Patients < 18 years of age At least one paid claim for an oral antipsychotic in the past 45 days, and monitoring for both glucose and lipid screening in the past 9 months (Table 2.3) Typical and Atypical antipsychotics: o All new start patients or patients changed to a different chemical entity will require a signed informed consent and a copy of a baseline metabolic lab test data. (Effective 11/8/2011) Medication Informed Consent Document o One therapeutic duplication for a change in therapy between two antipsychotics (oral or injectable) with > 25% remaining on the last fill on different dates of service allowed per 93 days. o PA required through manual review for recipients < 6 years of age. Oral liquids and orally disintegrating tablets (ODTs): Patient must have an NPO code (Appendix A) in the past year OR be < 7 years of age AND meet criteria for atypical antipsychotics Seroquel XR requires >/= 90 days of Seroquel XR therapy in the past 120 days. Immediate-release quetiapine (Seroquel) is covered via existing criteria. Additional dose criteria Atypical antipsychotics Requested dose must be an approved dose for age range (Table 2) Requested maximum daily dose must be approved for age range (Table 2.2). Denial criteria Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Antipsychotics Claims with a therapeutic duplication on the same date of service Requests for Loxapine, Thioridazine, Thiothixene, Fanapt®, Latuda®, or Saphris® for patients <18 years of age Requests for combination antipsychotic products for patients <18 years of age Failure to meet approval criteria Table 2 – Approved doses per age range Drug Abilify® Abilify® Abilify® Abilify® Abilify® Abilify® Abilify Dismelt® Abilify Dismelt® Abilify Solution® Chlorpromazine Chlorpromazine Chlorpromazine Chlorpromazine Chlorpromazine Fanapt® Fanapt® Fanapt® Fanapt® Fanapt® Fanapt® Fanapt® Fluphenazine Fluphenazine Fluphenazine Fluphenazine Fluphenazine Elixir Fluphenazine Soln Geodon® Geodon® Geodon® Geodon® Strength 2 mg 5 mg 10 mg 15 mg 20 mg 30 mg 10 mg 15 mg 1 mg/ml 10 mg 25 mg 50 mg 100 mg 200 mg 1 mg 2 mg 4 mg 6 mg 8 mg 10 mg 12 mg 1 mg 2.5 mg 5 mg 10 mg 2.5mg/5ml 5 mg/ml 20 mg 40 mg 60 mg 80 mg FDA dosing QD QD QD QD QD QD QD QD QD BID-QID BID-QID BID-QID BID-QID BID-QID BID BID BID BID BID BID BID BID-QID BID-QID BID-QID BID-QID BID-QID BID-QID BID BID BID BID <6* y/o 2 tabs 1 tab 6-9 y/o 2 tabs 1 tab 1 tab 1 tab 10-12y/o 2 tabs 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 15 mls 4 tabs 4 tabs 4 tabs 2 tabs 1 tab 2 tabs 2 tabs 1 tab 2 tabs 1 tab 20 mls 4 tabs 4 tabs 4 tabs 4 tabs 2 tabs 2 tabs 2 tabs 2 tabs 1 tab 1 tab 2 tabs 4 tabs 2 tabs 1 tab 4 mls 0.4 ml 2 caps 10 mls 1 ml 2 caps 1 cap 1 cap 4 tabs 4 tabs 2 tabs 1 tab 20 mls 2 mls 2 caps 2 caps 5 mls 4 tabs 4 tabs 2 tabs 1 tab 2 tabs 1 tab 1317y/o 2 tabs 1 tab 1 tab 1 tab 1 tab 1 tab 2 tabs 2 tabs 30 mls 4 tabs 4 tabs 4 tabs 4 tabs 3 tabs 2 tabs 2 tabs 2 tabs 2 tabs 2 tabs 1 tab 1 tab 4 tabs 4 tabs 4 tabs 2 tabs 40 mls 4 mls 2 caps 2 caps 2 caps 2 caps Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Drug Haloperidol Haloperidol Haloperidol Haloperidol Haloperidol Haloperidol Haloperidol Soln Invega® Invega® Invega® Invega® Latuda® Latuda® Latuda® Latuda® Latuda® Loxapine Loxapine Loxapine Loxapine Orap® Orap® Perphenazine Perphenazine Perphenazine Perphenazine Risperdal® Risperdal® Risperdal® Risperdal® Risperdal® Risperdal® Risperdal® M Tab Risperdal® M Tab Risperdal® M Tab Risperdal® M Tab Risperdal® M Tab Risperdal® M Tab Risperdal® Soln Saphris® SL Saphris® SL Seroquel® Seroquel® Strength 0.5 mg 1 mg 2 mg 5 mg 10 mg 20 mg 2 mg/ml 1.5 mg 3 mg 6 mg 9 mg 20 mg 40 mg 60 mg 80 mg 120 mg 5 mg 10 mg 25 mg 50 mg 1 mg 2 mg 2 mg 4 mg 8 mg 16 mg 0.25 mg 0.5 mg 1 mg 2 mg 3 mg 4 mg 0.25 mg 0.5 mg 1 mg 2 mg 3 mg 4 mg 1 mg/ml 5 mg 10 mg 25 mg 50 mg FDA dosing BID-TID BID-TID BID-TID BID-TID BID-TID BID-TID BID-TID QD QD QD QD QD QD QD QD QD BID BID BID BID QD-BID QD-BID BID-QID BID-QID BID-QID BID-QID BID BID BID BID BID BID BID BID BID BID BID BID BID BID BID TID TID <6* y/o 3 tabs 2 tabs 1 tab 6-9 y/o 3 tabs 3 tabs 2 tabs 1 tab 10-12y/o 3 tabs 3 tabs 3 tabs 2 tabs 1 tab 1 ml 1 tab 1 tab 2.5 ml 1 tab 1 tab 5 ml 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 2 caps 1 cap 2 caps 2 caps 2 caps 2 caps 1 tab 2 tabs 1 tab 1 tab 1 tab 3 tabs 1 tab 1 tab 2 tabs 4 tabs 2 tabs 1 tab 2 tabs 2 tabs 2 tabs 1 tab 2 tabs 2 tabs 2 tabs 2 tabs 2 tabs 2 tabs 2 tabs 1 tab 1 tab 2 tabs 2 tabs 2 tabs 2 tabs 2 mls 1 tab 1 tab 4 mls 1 tab 3 tabs 3 tabs 3 tabs 3 tabs 2 tabs 2 tabs 2 tabs 2 tabs 2 tabs 1 tab 2 tabs 2 tabs 2 tabs 2 tabs 2 tabs 1 tab 6 mls 2 tabs 1 tab 3 tabs 3 tabs 1317y/o 3 tabs 3 tabs 3 tabs 3 tabs 2 tabs 1 tab 10 ml 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 2 caps 2 caps 2 caps 1 cap 1 tab 5 tabs 4 tabs 4 tabs 2 tabs 1 tab 2 tabs 2 tabs 2 tabs 2 tabs 2 tabs 2 tabs 2 tabs 2 tabs 2 tabs 2 tabs 2 tabs 2 tabs 8 mls 2 tabs 2 tabs 3 tabs 3 tabs Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Drug Seroquel® Seroquel® Seroquel® Seroquel® Seroquel® XR Seroquel® XR Seroquel® XR Seroquel® XR Seroquel® XR Thioridazine Thioridazine Thioridazine Thioridazine Thiothixene Thiothixene Thiothixene Thiothixene Trifluoperazine Trifluoperazine Trifluoperazine Trifluoperazine Zyprexa® Zyprexa® Zyprexa® Zyprexa® Zyprexa® Zyprexa® Zyprexa® Zydis® Zyprexa® Zydis® Zyprexa® Zydis® Zyprexa® Zydis® Strength 100 mg 200 mg 300 mg 400 mg 50 mg 150 mg 200 mg 300 mg 400 mg 10 mg 25 mg 50 mg 100 mg 1 mg 2 mg 5 mg 10 mg 1 mg 2 mg 5 mg 10 mg 2.5mg 5mg 7.5mg 10mg 15mg 20mg 5mg 10mg 15mg 20mg FDA dosing TID TID TID TID QD QD QD QD QD BID-TID BID-TID BID-TID BID-TID TID TID TID TID QD-BID QD-BID QD-BID QD-BID QD QD QD QD QD QD QD QD QD QD <6* y/o 1 tab 6-9 y/o 3 tabs 1 tab 1 tab 2 tabs 1 tab 2 tabs 1 tab 1 tab 1 tab 3 tabs 2 tabs 1 tab 3 tabs 3 tabs 2 tabs 1 tab 3 caps 3 caps 1 cap 3 caps 3 caps 1 cap 10-12y/o 3 tabs 3 tabs 2 tabs 1 tab 2 tabs 1 tab 1 tab 2 tabs 1 tab 3 tabs 3 tabs 3 tabs 1 tab 3 caps 3 caps 1 cap 1 tab 2 tabs 1 tab 2 tabs 2 tabs 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1317y/o 3 tabs 3 tabs 2 tabs 2 tabs 2 tabs 1 tab 1 tab 2 tabs 2 tabs 3 tabs 3 tabs 3 tabs 2 tabs 3 caps 3 caps 3 caps 1 cap 2 tabs 2 tabs 2 tabs 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab 1 tab *Prior authorization required through manual review for recipients < 6 years of age. Table 2.2 – Max daily doses for age categories < 18 years of age. Drug Abilify® Geodon® Invega® Risperdal® <6* y/o 5 mg daily 40 mg daily 3 mg daily 2 mg daily 6*-9 y/o 15 mg daily 60 mg daily 3 mg daily 4 mg daily 10-12 y/o 20 mg daily 80 mg daily 6 mg daily 6 mg daily 13-17 y/o 30 mg daily 160 mg daily 9 mg daily 8 mg daily Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Drug Seroquel® Zyprexa® <6* y/o 150 mg daily 5 mg daily 6*-9 y/o 300 mg daily 10 mg daily 10-12 y/o 600 mg daily 15 mg daily 13-17 y/o 800 mg daily 20 mg daily *Prior authorization required through manual review for recipients < 6 years of age. Table 2.3 – CPT codes for glucose and lipid monitoring. Glucose codes: Criteria require one of the following CPT codes that contain glucose monitoring in the previous 9 months from claim date of in-process claim: 83036 (HbA1c), OR 80050 (General Health Panel), OR 80069 (Renal Function Panel), OR 80047 (Basic Metabolic Panel), OR 80048 (Basic Metabolic Panel), OR 80053 (Comprehensive metabolic panel ), OR 82962 (Glucose, blood by glucose monitoring device(s) cleared by the FDA specifically for home use) OR 82948 (Glucose; blood, reagent strip ) OR 82947 (Glucose; quantitative, blood), AND, criteria require one of the following lipid panel tests or all of the individual lipid test monitoring codes in previous 9 months from claim date of the in-process claim: Lipid codes: 80061 (Lipid panel ), OR 83701 (High resolution fractionation and quantitation of lipoproteins panel), OR 82465 (Cholesterol, serum or whole blood, total ), AND 83718 (HDL cholesterol ), AND 84478 (Triglycerides ), AND 83721 (LDL Cholesterol) Link to Memorandum (Initial Antipsychotic criteria) Link to Memorandum (Requirements of informed consent and metabolic monitoring) Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Apixaban Tablet (Eliquis) (Implemented 07/09/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis for Atrial Fibrillation in Medicaid history within past 2 years; No Therapeutic duplication allowed between different strengths of Eliquis®; One (1) therapeutic duplication with overlapping days’ supply will be allowed once per 186 days for inferred change in therapy between an Eliquis® claim and any of the following: a Xarelto® claim, a warfarin claim, OR a Pradaxa® claim, AND The Eliquis® claim and the warfarin claim, the Xarelto® claim, or the Pradaxa® claim cannot have the same date of service. Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Armodafinil (Nuvigil) (Implemented 05/27/2009) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Age > 16 years of age, AND History of at least one of the following in the last two years: o Narcolepsy, OR o Multiple sclerosis, OR o Shift work sleep disorder, OR o Obstructive sleep apnea/hypopnea, AND CPAP, OR BPAP, OR Nasal interface positive airway pressure device Denial criteria Age < 16 years of age, OR History of at least one of the following in the last 30 days: o Atomoxetine (Strattera), OR o CII Stimulants, OR o Other modafinil (Provigil) strengths, OR o Other armodafinil (Nuvigil) strengths, OR o Pemoline (Cylert) Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Aromatase Inhibitors (Arimidex and Femara) (Implemented 09/24/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require prior authorization Anastrozole oral tablet [Arimidex] Letrozole oral tablet [Femara] Approval criteria Medical history for female metastatic breast cancer in the past 3 years Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Attention Deficit Disorder and Attention Deficit Hyperactivity Disorder (ADD/ADHD) Agents for Children (Less than 18 Years of Age) (Implemented 07/21/2009) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents with criteria Adderall XR (Brand only) Amphetamine salts immediate-release tablet (Adderall) Atomoxetine HCl (Strattera) Dextroamephetamine sulfate 5mg, 10mg tablet (Dextrostat) Focalin (Brand only) Focalin XR (Brand only) Lisdexamfetamine dimesylate (Vyvanse) Methylphenidate HCl swallow tablet (Ritalin) Nonpreferred agents Amphetamine salts extended-release capsule (generic Adderall XR) Clonidine extended-release suspension (Nexiclon XR) Clonidine extended-release tablet (Kapvay ER, Nexiclon XR) Dexmethylphenidate HCl extended-release capsule (generic Focalin XR) Dexmethylphenidate HCl tablet (generic Focalin) Dextroamephetamine sulfate extended-release capsule (Dexedrine Spansule) Dextroamphetamine sulfate solution (Liquadd, Procentra) Dextroamphetamine sulfate 2.5mg, 7.5mg tablet (Zenzedi) Guanfacine HCl extended-release tablet (Intuniv ER) Methamphetamine HCl (Desoxyn) Methylphenidate HCl chewable tablet (Methylin) Methylphenidate HCl extended-release capsule (Metadate CD, Ritalin LA) Methylphenidate extended-release patch (Daytrana) Methylphenidate HCl extended-release suspension (Quillivant XR) Methylphenidate HCl extended-release tablet (Concerta) Methylphenidate HCl extended-release tablet (Metadate ER, Ritalin SR) Methylphenidate HCl solution (Methylin) Approval criteria for preferred agents with criteria for children: Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Less than18 years of age All preferred extended-release CII stimulants: ≤ One therapeutic duplication between long-acting CII stimulants with 75% of the last fill per 93 days AND If a incoming long-acting CII stimulant claim overlaps with a short-acting CII stimulant that was filled at a dose of >/= to 2 units per day, the long-acting product will require prior authorization All preferred immediate-release CII stimulants: ≤ One therapeutic duplication between short-acting CII stimulants with 75% of the last fill per 93 days AND If an incoming short-acting CII stimulant claim overlaps with a long-acting CII stimulant, the short-acting product will only be approved for a dose of one unit per day Additional criteria Age and Quantity limits apply Link to orginal Memorandum Link to current Memorandum with new quantity restrictions Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Attention Deficit Disorder and Attention Deficit Hyperactivity Disorder (ADD/ADHD) Agents for Adults (18 Years of Age or greater) (Implemented 01/18/2011) Preferred agents with criteria Adderall XR (Brand only) Amphetamine salts immediate-release tablet (Adderall) Atomoxetine HCl (Strattera) Dextroamephetamine sulfate 5mg, 10mg tablet (Dextrostat) Focalin (Brand only) Focalin XR (Brand only) Lisdexamfetamine dimesylate (Vyvanse) Methylphenidate HCl swallow tablet (Ritalin) Nonpreferred agents Amphetamine salts extended-release capsule (generic Adderall XR) Clonidine extended-release suspension (Nexiclon XR) Clonidine extended-release tablet (Kapvay ER, Nexiclon XR) Dexmethylphenidate HCl extended-release capsule (generic Focalin XR) Dexmethylphenidate HCl tablet (generic Focalin) Dextroamephetamine sulfate extended-release capsule (Dexedrine Spansule) Dextroamphetamine sulfate solution (Liquadd, Procentra) Dextroamphetamine sulfate 2.5mg, 7.5mg tablet (Zenzedi) Guanfacine HCl extended-release tablet (Intuniv ER) Methamphetamine HCl (Desoxyn) Methylphenidate HCl chewable tablet (Methylin) Methylphenidate HCl extended-release capsule (Metadate CD, Ritalin LA) Methylphenidate extended-release patch (Daytrana) Methylphenidate HCl extended-release suspension (Quillivant XR) Methylphenidate HCl extended-release tablet (Concerta) Methylphenidate HCl extended-release tablet (Metadate ER, Ritalin SR) Methylphenidate HCl solution (Methylin) Approval criteria for preferred agents with criteria for adults: 18 years of age or greater Diagnosis of Narcolepsy in Medicaid History in the previous 2 years and No therapeutic duplication with a different ADD/ADHD with different GCN in history with > 25% remaining days supply Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria No therapeutic duplication with a Provigil/Nuvigil in history with > 25% remaining days supply No therapeutic duplication with Atomoxetine in history with > 25% remaining days supply Atomoxetine for adults (18 years of age or greater) Required for all new starts with no diagnosis of Narcolepsy in Medicaid history. Denial criteria for adults (18 years of age or greater) No Diagnosis of Narcolepsy in Medicaid History in the previous 2 years will require a manual review Additional criteria Quantity limits apply Link to Memorandum: CII Stimulant for Adults Link to current Memorandum with new quantity restrictions Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Auranolfin (Ridaura) Capsule (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Ridaura Capsule Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Axitinib Tablet (Inlyta) (Implemented 07/09/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Inlyta tablet Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Azithromycin (Azithromycin Powder Packets and ZMAX) (Implemented 04/12/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Azithromycin 1 gm powder packets ZMAX 2gm/60ml suspension Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Aztreonam Inhalation Solution (Cayston) (Implemented 09/28/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Cayston Inhalation Solution Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Balsalazide Disodium Tablet (Giazo) (Implemented 07/09/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Giazo Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Becaplermin (Regranex) (Implemented 01/12/2005) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Submitted ICD-9 diagnosis of diabetes mellitus in past 365 days, AND Submitted ICD-9 diagnosis of skin ulcer in past 180 days Denial criteria Submitted ICD-9 diagnosis of osteomyelitis in past 90 days, OR > one claim in past 30 days Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Bedaquiline Fumarate Tablet (Sirturo) (Implemented 12/10/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Sirturo Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Belimumab (Benlysta) (Implemented 06/21/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Benlysta Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Benign Prostatic Hypertrophy (BPH) Drugs (Implemented 01/12/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). BENIGN PROSTATIC HYPERTROPHY (BPH) DRUGS GENERIC NAME COMMON TRADE NAME* PA STATUS Alfuzosin HCl ER 10 mg tablet Uroxatral® 10 mg tablet No PA Finasteride 5 mg tablet Proscar 5® mg tablet See Finasteride Tamsulosin HCl 0.4 mg capsule Flomax® 0.4 mg capsule No PA GENERIC NAME COMMON TRADE NAME* PA STATUS Dutasteride 0.5 mg softgel Avodart® 0.5 mg softgel PA Dutasteride 0.5 mg-Tamsulosin 0.4 mg capsule Jalyn® 0.5-0.4 mg capsule PA Silodosin 4 mg capsule Rapaflo® 4 mg capsule PA Silodosin 8 mg capsule Rapaflo® 8 mg capsule PA *TRADE NAMES ARE FOR REFERENCE ONLY Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Benzodiazepine Oral Solid Dosage Forms (Implementation Date 12/07/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Up to 124 units of any solid oral benzodiazepines paid by Medicaid per the previous 31 calendar days. Exemption from accumulation quantity limit Diastat AcuDial rectal gel Benzodiazepine injectable agents Benzodiazepine oral liquid agents. Additional criteria Daily quantity limits apply for Alprazolam ER, Clobazam, Estazolam, Flurazepam, Quazepam, Temazepam Onfi tablet requires a Manual PA (See Clobazam [Onfi] Tablet) Temazepam 22.5 mg Capsule requires a Manual PA (see Temazepam 22.5 mg) Alprazolam XR [Xanax XR] additional approval criteria: o > 18 years of age, AND o >/= 90 days of Alprazolam XR therapy in the past 120 days Alprazolam oral-disintegrating tablet [Niravam] o > 18 years of age, AND o One of the following: Long Term Care NPO (Appendix A) Denial criteria Therapeutic duplication with alprazolam Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Benzodiazepine Oral Liquid Dosage Forms (Implementation Date 12/07/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria </= 6 years of age, OR NPO (Appendix A) Additional criteria Quantity limits apply Exemption criteria Midazolam 2 mg/ml Syrup Claims for 30 ml or less will pay at point-of-sale for any age. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Beta Adrenergic Blocking Agents (Implemented 10/17/2007) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents Atenolol (Tenormin) Metoprolol tartrate (Lopressor) Propranolol HCl immediate-release (Inderal) Preferred agents with criteria Bisoprolol fumarate (Zebeta) Carvedilol tablet (Coreg) Metoprolol succinate extended-release (Toprol XL) Nonpreferred agents Acebutolol HCl (Sectral) Betaxolol HCl (Kerlone) Carvedilol phosphate capsule (Coreg CR) Labetalol HCl (Normodyne) Nadolol (Corgard) Nebivolol HCl (Bystolic) Penbutolol sulfate (Levatol) Pindolol (Visken) Propranolol HCl extended-release capsule (Inderal LA) Propranolol HCl solution (Hemangeol) Timolol maleate (Blocadren) Approval criteria for preferred agents with criteria Bisoprolol fumarate (Zebeta), Carvedilol Tablet (Coreg), and Metoprolol succinate extended-release (Toprol XL) Congestive heart failure (Appendix D) Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Inhaled Long-acting Beta2 Agonist and Corticosteroid Combination (Implemented 08/11/2009) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents with criteria Budesonide/Formoterol fumarate dihydride inhalation aerosol (Symbicort®) Mometasone furoate/Formoterol fumarate dihydride Inhalation Aerosol (Dulera®) Approval criteria for preferred agents with criteria Criterion 1: COPD diagnosis (Appendix G) in the past two years AND ≥ 18 years old AND ≥3 claims in the last 120 days of any product including: Arcapta®, Anoro™ Ellipta®, Breo® Ellipta®, Brovana®, Foradil®, Perforomist®, Serevent®, Spriva®, or Tudorza® Pressair® OR Criterion 2: Paid drug claim in drug history for Advair Diskus®, Advair® HFA, Dulera®, or Symbicort® in the last six months OR Criterion 3: One of the following criteria below: > Three inhaled corticosteroid claims in the last 120 days, OR > Three oral steroid claims in the last 120 days, OR Combination for > three claims (as defined below) in the last 120 days: One Inhaled Corticosteroid + 2 Oral Steroids Two Inhaled Corticosteroids + 1 Oral Steroids Non-Preferred agents Fluticasone furoate/Vilanterol inhalation powder (Breo® Ellipta®) Fluticasone propionate/Salmeterol inhalation aerosol (Advair® HFA) Fluticasone propionate/Salmeterol 250-50 MCG inhalation powder (Advair 250-50 Diskus®) Fluticasone propionate/Salmeterol 500-50 MCG inhalation powder (Advair 500-50 Diskus®) Nonpreferred agents with criteria Fluticasone propionate/Salmeterol 100-50 MCG inhalation powder (Advair 100-50 Diskus®) Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Approval criteria for nonpreferred agents with criteria Criterion 1: Beneficiary is between 4 years through 11 years of age AND has a paid claim in drug history for Advair Diskus®, Advair® HFA, Dulera®, or Symbicort® in the last six months OR Criterion 2: Beneficiary is between 4 years through 11 years of age AND One of the following criteria below: > Three inhaled corticosteroid claims in the last 120 days, OR > Three oral steroid claims in the last 120 days, OR Combination for > three claims (as defined below) in the last 120 days: One Inhaled Corticosteroid + 2 Oral Steroids Two Inhaled Corticosteroids + 1 Oral Steroids Denial criteria Absence of approval criteria Therapeutic duplication between different Inhaled Long-acting Beta2 Agonists-Corticosteroid Combination Products Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Bexarotene Gel (Targretin) (Implemented 10/01/2004) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Targretin Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Bosutinib (Bosulif 100mg and 500mg Tablets) (Implemented 03/19/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Bosulif Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Botulinum Toxin Type A and Onabotulinumtoxina (Botox 100 unit and 200 unit) (Implementation Date 06/21/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Denial criteria Diagnosis in Medicaid History of migraine headaches in the past 2 years, OR At least one paid claim for Triptan medication in the past 2 years Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Bowel Prep Agents and Kits (Implementation Date 10/11/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Colyte Solution Colyte with Flavor Packets Gavilyte-C Solution Gavilyte-G Soution Gavilyte-N Solution Golytely Packet Golytely Solution Moviprep Powder Kit Nulytely with Flavor Packs Solution PEG-3350 and Electrolytes Solution PEG-3350 with Flavor Packs Solution Trilyte with Flavor Packets Drugs that require manual review for prior authorization Halflytely-Bisacodyl Bowel Kit Osmoprep Tablet Prepopik Powder Packet Suclear Bowel Prep Kit Suprep Bowel Prep Kit Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Bronchodilators, Long-Acting (Implemented 08/11/2009) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Nonpreferred agents with criteria Aclidinium bromide inhaler (Tudorza Pressair) (Click for Tudorza Critiera) Tiotropium bromide inhaler (Spiriva Handihaler) (Click for Spiriva Criteria) Nonpreferred agents without criteria Arformoterol tartrate inhalation solution (Brovana) Formoterol fumarate inhaler (Foradil) Formoterol fumarate inhalation solution (Perforomist) Indacaterol maleate inhaler (Arcapta Neohaler) Salmeterol xiafoate disk with device (Serevent Diskus) Umeclidinium bromide inhaler (Incruse Ellipta) Umeclidinium-Vilanterol inhaler (Anoro Ellipta) Approval criteria for nonpreferred agents with criteria COPD (Appendix G) Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Bronchodilators, Short-Acting (Implemented 08/11/2009) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents Albuterol sulfate 5mg/ml solution Albuterol sulfate 0.83mg/ml solution Albuterol sulfate HFA inhaler (Ventolin HFA 18 GRAM) Nonpreferred agents Albuterol 2.5mg/0.5ml solution Albuterol sulfate 0.21mg/ml, 0.42mg/ml solution (Accuneb) Albuterol sulfate HFA inhaler (ProAir HFA) Albuterol sulfate HFA inhaler (Proventil HFA) Albuterol sulfate HFA inhaler (Ventolin HFA 8 GRAM) Levalbuterol HCl inhalation solution (Xopenex) Levalbuterol tartrate HFA inhaler (Xopenex HFA) Metaproterenol inhaler and solution (Alupent) Pirbuterol acetate inhaler (Maxair Autohaler) Nonpreferred agents with criteria Albuterol sulfate-Ipratropium bromide inhaler(Combivent, Combivent Respimat, Duoneb) Ipratropium bromide HFA inhaler (Atrovent HFA) Approval criteria for nonpreferred agents with criteria One of the following diagnoses or procedures: Anoxic brain injury (348.1) COPD (Appendix G) Heart transplant (V421) Quadriplegic cerebral palsy (343.2) Respiratory insufficiency 518.82 — Other pulmonary insufficiency, not elsewhere classified 518.83 — Chronic respiratory failure 518.84 — Acute and chronic respiratory failure Tracheostomy (Appendix B) Trachoemalacia congenital (748.3) Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Budesonide Extended-Release 9mg (Uceris) (Implemented 07/09/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Submitted Diagnosis of Ulcerative Colitis in the past 2 years Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Budesonide EC 3mg Capsule (Entocort EC) (Implemented 07/09/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Submitted Diagnosis of Crohn’s Disease in the past 2 years Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Buprenorphine (Subutex) (Implemented 04/21/2009) Prescribers are required to fax a letter of medical necessity and include all supporting documentation for manual review to 501-683-4124. Drugs that require manual review for prior authorization Subutex Additional criteria Quantity limits apply Link to Subutex statement of necessity—Microsoft Word format (.doc) Link to Subutex statement of necessity—Portable Document Format (.pdf) Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Buprenorphine-Naloxone (Suboxone) (Implemented 04/21/2009) Prescribers are required to fax a letter of medical necessity and include all supporting documentation for manual review to 501-683-4124. Drugs that require manual review for prior authorization Suboxone Additional criteria Quantity limits apply Link to Suboxone statement of necessity—Microsoft Word format (.doc) Link to Suboxone statement of necessity—Portable Document Format (.pdf) Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Butalbital Products (Implemented 01/18/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require a manual PA Butalbital-Acetaminophen 50-325 mg TABLET (Marten-Tab) Butalbital-Acetaminophen-Caffeine 50-325-40 mg TABLET (Esgic Tablet) Drugs that require a manual PA Butalbital-Acetaminophen 50-300 mg TABLET (Bupap 50-300 mg Tablet) Butalbital-Acetaminophen-Caffeine 50-300-40 mg CAPSULE (Fioricet Capsule) Butalbital-Acetaminophen-Caffeine 50-325-40 mg CAPSULE (Esgic Capsule) Butalbital-Aspirin-Caffeine 50-325-40 mg CAPSULE (Fiorinal Capsule) Age Edit Recipient must be at least 12 Years of Age or greater Quantity Edit Solid Oral dosage forms of butalbital products will be limited up to a maximum of 6 units per day Solid Oral dosage forms of butalbital products will have a cumulative quantity limit of 124 units per 31 days’ supply The butalibital products that contain 750mg acetaminophen per unit will be limited to a maximum of 5 units per day based on the maximum amount of acetaminophen allowed per day Oral liquid forms of butalbital will be limited to 60ml per day or up to 240ml per prescription Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria C1 Esterase Inhibitor (Cinryze) (Implemented 01/21/2011) Prescribers are required to fax a letter of medical necessity and include all supporting documentation for manual review to 501-683-4124. Drugs that require manual review for prior authorization Cinryze A letter of medical necessity must be sent, along with ALL necessary documentation substantiating ALL of the criteria listed below. 1. Documented diagnosis of hereditary angioedema by an immunologist AND 2. The recipient’s history of HAE attacks is consistent with two or more abdominal or respiratory attacks per month that require hospital ER intervention with usage of Berinert® or Kalbitor® in the previous 6 consecutive months (ER documentation is required) AND 3. The Member is NOT concurrently taking an angiotensin converting enzyme (ACE) inhibitor or estrogen replacement therapy AND 4. The recipient has had an insufficient response or contraindication to BOTH of the following classes of medication: a. 17α – alkylated androgens (e.g. danazol, stanozolol, oxandrolone, methyltestosterone) b. Antifibrinolytic agents (e.g. ε – aminocaproic acid, tranexamic acid) Note: The Arkansas Medicaid Medical Director will work with hospital emergency departments to ensure the availability of Berinert®, Kalbitor®, or Ruconest® for acute attacks of HAE for authorized recipients. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Cabozantinib Capsule (Cometriq) (Implemented 07/09/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Cometriq Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Calcitrol (Vectical), Calcipotriene (Dovonex, Sorilux) (Implemented 06/19/2006) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drug Dovonex Sorilux Vectical Approval criteria (New Start) Diagnosis of psoriasis in Medicaid history in previous 365 days, AND 2 paid claims of a topical corticosteroid in previous 27-60 days, AND At least one paid claim for a topical corticosteroid must be from the very high potency category. Approval criteria (Continuation Criteria) Diagnosis of psoriasis in Medicaid history in previous 365 days, AND The incoming claim matches claim in history in the previous 45 days, AND At least two claims of a topical corticosteroid in previous 60 days in drug claim history with at least one topical corticosteroid claim 14-60 days back in history. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Calcipotriene and Betamethasone Dipropionate (Taclonex) (Implemented 01/09/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria ≥ 18 years of age, AND History of three paid claims in the past 90 days for Calcipotriene (Dovonex), AND History of three paid claims in the past 90 days for a topical steroid Denial criteria < 18 years of age Concurrent use of a topical corticosteroid Failure to meet the approval criteria Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Calcitonin-salmon Nasal Spray (Miacalcin or Fortical) (Implemented 08/17/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria For Males: Diagnosis osteoporosis in the previous 2 years, AND Diagnosis of esophageal strictures in the previous 2 years , OR Diagnosis of esophageal achalasia in the previous 2 years For Females: Diagnosis of Postmenopause in the previous 2 years, AND Diagnosis Osteoporosis in the previous 2 years, AND Diagnosis of esophageal strictures in the previous 2 years , OR Diagnosis of esophageal achalasia in the previous 2 years Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Calcitonin-salmon Injection (Miacalcin) (Implemented 08/17/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of hypercalcemia in the past two years. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Calcium Channel Blockers (Implemented 07/12/2005) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents Amlodipine besylate (Norvasc) Amlodipine besylate/Olmesartan medoxomil* (Azor) Amlodipine besylate/Olmesartan medoxomil/Hydrochlorothiazide* (Tribenzor) Amlodipine besylate/Valsartan* (Exforge) Amlodipine besylate/Valsartan/Hydrocholorothiazide* (Exforge HCT) Diltiazem HCl extended-release 120mg, 180mg, 240mg capsule (Dilacor XR) Nifedipine extended-release (Adalat CC, Procardia XL) Verapamil extended-release tablet (Calan SR) Nonpreferred agents Amlodipine besylate/Atorvastatin calcium (Caduet) Diltiazem HCl extended-release, CD, LA, SA, XR, XT (Cardizem, Tiazac) Felodipine extended-release (Plendil) Isradipine (Dynacirc) Isradipine extended-release (Dynacirc CR) Nicardipine HCl (Cardene) Nicardipine HCL extended-release (Cardene SR) Nisoldipine extended-release (Sular ER) Verapamil extended-release capsule (Verelan) *See Angiotensin II Receptor Antagonist criteria Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Carbidopa (Lodosyn) (Implemented 01/12/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria At least 1 paid Medicaid claim for Sinemet (carbidopa/levodopa) in the previous 60 days, OR At least 1 paid Medicaid claim for Stalevo in the previous 60 days. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Carbidopa-Levodopa-Entacapone (Stalevo) (Implemented 01/12/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria No therapeutic duplication with Comtan. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Cephalexin 750mg Capsule (Keflex) (Implemented 07/09/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Keflex 750mg capsule Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Cephalosporins – 3rd Generation (Implementation Date 3/18/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization CEFDINIR 300 MG ORAL CAPSULE [OMNICEF] CEFDINIR 125 MG/5 ML ORAL SUSPENSION [OMNICEF] CEFDINIR 250 MG/5 ML ORAL SUSPENSION [OMNICEF CEFPODOXIME PROXETIL 50 MG/ML ORAL SUSPENSION [VANTIN] CEFPODOXIME PROXETIL100 MG/ML ORAL SUSPENSION [VANTIN] CEFPODOXIME PROXETIL 100 MG ORAL TABLET [VANTIN] CEFPODOXIME PROXETIL 200 MG ORAL TABLET [VANTIN] Drugs that require manual review for prior authorization CEFDITOREN PIVOXIL 200 MG ORAL TABLET [SPECTRACEF]* CEFDITOREN PIVOXIL 400 MG ORAL TABLET [SPECTRACEF]* CEFIXIME 400 MG ORAL CAPSULE [SUPRAX] CEFIXIME 100 MG ORAL CHEWABLE TABLET [SUPRAX] CEFIXIME 200 MG ORAL CHEWABLE TABLET [SUPRAX] CEFIXIME 100 MG/5 ML ORAL SUSPENSION [SUPRAX] CEFIXIME 200 MG/5 ML ORAL SUSPENSION [SUPRAX] CEFIXIME 500 MG/5 ML ORAL SUSPENSION [SUPRAX] CEFIXIME 400 MG ORAL TABLET [SUPRAX] CEFTIBUTEN DIHYDRATE 400 MG ORAL CAPSULE [CEDAX] CEFTIBUTEN DIHYDRATE 180 MG/5 ML ORAL SUSPENSION [CEDAX] *Being removed from market. Limited availability. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Ceritinib Capsule (Zykadia) (Implemented 09/23/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Zykadia Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Chlorpheniramine ER 12mg (Implemented 04/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria >/= 90 days of Chlorpheniramine ER therapy in the past 120 days Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Ciclopirox 8% Topical Solution (Implemented 01/13/2015) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Itraconazole Capsule [Sporanox] Terbinafine Tablet [Lamisil] Drugs that require manual PA Ciclopirox 8% Topical Solution [Penlac] Efinaconazole 10% Topical Solution [Jublia] Tavaborole 5% Topical Solution [Kerydin] Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Cidofovir Injection (Vistide) (Implemented 04/17/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Vistide Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Clobazam (Onfi) (Implemented 07/09/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Onfi suspension Onfi tablet Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Clonazepam Orally Disintegrating Tablet (Implemented 10/11/2005) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria < 6 years of age, OR NPO (Appendix A), OR Long Term Care Eligible Approval criteria Up to 124 Units of any solid oral benzodiazepines paid by Medicaid per the previous 31 calendar days. Exemption from accumulation quantity limit Diastat AcuDial rectal gel Benzodiazepine injectable agents Benzodiazepine oral liquid agents. Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Clonidine and Guanfacine (Implemented 07/11/2009) Prescribers may request an override by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Approval criteria Patients ≥ 18 years of age All claims are approved Patients < 18 years of age One therapeutic duplication with > 25% remaining on the last fill on different dates of service allowed per 93 days between two clonidine claims, two guanfacine claims, or one clonidine claim and one guanfacine claim Cumulative quantity edits will apply (Table 3) Maximum daily dose edits will apply (Table 3.1) Table 3 – Cumulative quantity edits Generic name Clonidine HCL 0.1mg tablet Clonidine HCL 0.2mg tablet Clonidine HCL 0.3mg tablet Guanfacine 1mg tablet Guanfacine 2mg tablet Cumulative qty < 18 y/o 124 per 31 days 62 per 31 days 31 per 31 days 93 per 31 days 62 per 31 days Table 3.1 – Maximum daily dose edits Generic name Clonidine HCL 0.1mg tablet Clonidine HCL 0.2mg tablet Clonidine HCL 0.3mg tablet Guanfacine 1mg tablet Guanfacine 2mg tablet Link to Memorandum Top of the document Dose < 18 y/o 4 tabs per day 2 tabs per day 1 tab per day 3 tabs per day 2 tabs per day Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Clonidine Vials (Implemented 07/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Clonidine HCl PF vials 5000mcg/10ml Clonidine HCl PF vials 1000mcg/10ml Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Colchicine Tablet (Colcrys) (Implemented 08/17/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of gout in the past three years. Diagnosis of Familial Mediterranean Fever (FMF) Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria COPD Agents (Implemented 09/23/2014) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Nonpreferred agents Roflumilast (Daliresp) Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Corticosteroids, Nasal Inhaled (Implemented 11/28/2006) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents Fluticasone propionate nasal spray (Flonase) Nonpreferred agents Azelastine/fluticasone nasal spray (Dymista) Beclomethasone dipropionate AQ nasal spray (Beconase AQ) Beclomethasone dipropionate nasal spray (Qnasl) Budesonide nasal spray (Rhinocort Aqua) Ciclesonide nasal spray (Omnaris, Zetonna)) Triamcinolone acetonide AQ nasal spray (Nasacort AQ) Nonpreferred agents with criteria Fluticasone furoate nasal spray (Veramyst) Mometasone furoate nasal spray (Nasonex) Approval criteria for nonpreferred agents with criteria Approvable if the beneficiary is between 2 years through 3 years of age Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Corticosteroids, Oral Inhaled (Implemented 08/11/2009) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents with criteria Budesonide inhaler (Pulmicort Flexhaler) Flunisolide inhaler (Aerospan) Fluticasone propionate disk with device (Flovent Diskus) Fluticasone proprionate HFA inhaler (Flovent HFA Inhaler) Nonpreferred agents Beclomethasone dipropionate inhaler (QVAR 7.3 gram) Beclomethasone dipropionate inhaler (QVAR 8.7 gram) Ciclesonide inhaler (Alvesco) Fluticasone furoate inhaler (Arnuity Ellipta) Mometasone furoate inhaler (Asmanex Twisthaler) Triamcinolone acetonide inhaler (Azmacort) Nonpreferred agents with criteria Budesonide ampules for nebulizer (Pulmicort Respules) Approval criteria for preferred agents with criteria For beneficiaries ≥ 18 years old, if there is a diagnosis for COPD (Appendix G) in the past two years, then it requires ≥ 3 claims in the past 120 days of any product including : Arcapta®, Anoro™ Ellipta®, Breo® Ellipta®, Brovana®, Foradil®, Perforomist®, Serevent®, Spiriva®, or Tudorza® Pressair® for approval. Approval criteria for nonpreferred agents with criteria Budesonide ampules for nebulizer < 4 years of age Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Corticotropin Gel Injection (Acthar HP) (Implemented 07/09/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Acthar HP Gel Injection Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Crizotinib Capsule (Xalkori) (Implemented 04/17/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Xalkori Information required for the manual review process Detection of ALK-positive NSCLC using an FDA-approved test, indicated for this use Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Crofelemer Delayed Release Tablet (Fulyzaq) (Implemented 07/09/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Fulyzaq Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Cromolyn Sodium Oral Solution (Gastrocrom) (Implemented 09/21/2009) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of mastocytosis (congenital pigmentary anomalies or malignant mast cell tumors) in the past three years Additional criteria Age edit : Approve > 2 years of age Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Crotamiton Cream and Lotion (Eurax) (Implemented 01/12/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Eurax cream and lotion Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Cyclosporine 0.05% Eye Emulsion (Restasis) (Implemented 01/18/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of one of the following diagnoses associated with dry eye in the past two years: Keratoconjunctivits sicca, non-Sjogren’s syndrome Keratoconjunctivits sicca, Sjogren’s syndrome Keratoconjunctivitis, exposure Tear film insufficiency, unspecified (Dry eye syndrome) Xerosis Denial criteria Therapeutic duplication with Lacrisert (hydroxypropyl cellulose) Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Cyproheptadine 4mg/10ml U.D. Cup (Implemented 04/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Currently LTC Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Cysteamine 0.44% Ophthalmic Drop (Cystaran) (Implemented 12/10/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Cystaran Ophthalmic Drop Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Cysteamine DR Capsule (Procysbi) (Implemented 03/18/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Procysbi 25mg DR Capsule Procysbi 75mg DR Capsule Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Dabigatran Etexilate Mesylate Capsules (Pradaxa) (Implemented 04/17/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of atrial fibrillation in Medicaid history in past 2 years, AND No Therapeutic duplication allowed between different strengths of Pradaxa®; One (1) therapeutic duplication with overlapping days’ supply will be allowed once per 186 days for inferred change in therapy between an Pradaxa® claim and any of the following: a Xarelto® claim, a warfarin claim, OR a Eliquis® claim, AND The Pradaxa® claim and the warfarin claim, the Xarelto® claim, or the Eliquis® claim cannot have the same date of service. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Dalfampridine Extended-Release Tablet (Ampyra ER) (Implemented 09/28/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Ampyra ER 10mg tablet Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Dabrafenib (Tafinlar) Capsules (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Tafinlar Additional Criteria Quantity Limits Apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Dasatinib (Sprycel) (Implemented 01/13/2015) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual PA Sprycel Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Deferiprone Tablet (Ferriprox) (Implemented 07/09/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Ferriprox tablet Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Denosumab Syringe (Prolia) (Implemented 01/13/2015) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Prolia Prolia® will continue to be covered through a manual review PA on a case-bycase basis for the initial dose. POS PA continuation approval criteria for Prolia® will apply as follows: 1 Prolia® claim is found in Medicaid drug history in the previous 12 months. In addition, a therapeutic duplication edit will reject an incoming Prolia® claim if an Xgeva® (denosumab) claim is found in the medical claims history in previous 6 months. A quantity edit for Prolia® of 1 injection per 175 days will be implemented. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Desmopressin (DDAVP) Nasal Spray and Solution (Implemented 03/26/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis in medicaid history of diabetes insipidus in the past three years. Denial criteria Diagnosis in medicaid history of nocturnal enuresis in the past three years. Diagnosis in medicaid history of urinary incontinence in the past three years. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Dexamethasone Dose Pak (Dexpak and Zema-Pak) (Implemented 10/11/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Dexamethasone 1.5mg Tablet Drugs that require manual review for prior authorization Dexpak Zema-Pak Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Dextromethorphan HBr/Quinidine Capsule (Nuedexta) (Implemented 06/21/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Nuedexta capsule Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Dihydroergotamine Mesylate Nasal Spray (Migranal) (Implemented 07/09/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Migranal Nasal Spray Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Direct Renin Inhibitors (Implemented 08/17/2010) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents with criteria Aliskiren (Tekturna) Aliskiren/HCTZ (Tekturna HCT) Aliskiren/Valsartan (Valturna) Nonpreferred agents Aliskiren/Amlodipine (Tekamlo) Aliskiren/Amlodipine (Amturnide) Approval criteria for preferred agent with criteria New starts will require a paid claim in history of an ACE-Inhibitor product (ACEInhibitor, ACE-Inhibitor/HCTZ, or ACE-Inhibitor/Calcium Channel Blocker) in the previous 27-45 days OR Continuation of therapy will require at least one paid claim in history of a “Preferred with criteria” angiotensin receptor blocker or direct renin inhibitor in the past 60 days Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Disopyramide CR (Norpace CR) (Implemented 04/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria >/= 90 days of Disopyramide CR therapy in the past 120 days Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Dornase Alfa inhalation Solution (Pulmozyme) (Implemented 01/09/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of cystic fibrosis in medical history Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Dorzolamide-Timolol 2%-0.5% PF (Cosopt PF) (Implemented 07/09/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Dorzolamide-Timolol 2%--0.5% (Cosopt) Drugs that require manual review for prior authorization Dorzolamide-Timolol 2%--0.5% preservative free (Cosopt PF) Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Doxepin 5% cream (Zonalon, Prudoxin) (Implemented 09/21/2009) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria History of > two claims for a steroidal product (from very high, high, or moderate strength) in the past 60 days Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Doxycycline/Minocycline (Implemented 06/19/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require a manual PA Generic MAC’d solid dosage forms of doxycycline and minocycline including: Doxycycline hyclate 20 mg tablet (Periostat®) Doxycycline hyclate 50 mg capsule (Vibramycin®) Doxycycline hyclate 100 mg capsule (Vibramycin®) Doxycycline hyclate 100 mg tablet (Vibra-tab®) Doxycycline monohydrate 50 mg capsule (Monodox®) Doxycycline monohydrate 100 mg capsule (Monodox®) Minocycline HCl 50 mg capsule (Minocin®) Minocycline HCl 75 mg capsule (Dynacin®) Minocycline HCl 100 mg capsule (Minocin®) Drugs that require manual PA Doxycycline hyclate 75 mg delayed-release capsule & tablet (Doryx®) Doxycycline hyclate 100 mg delayed-release capsule & tablet (Doryx®) Doxycycline monohydrate 40 mg extended-release capsule (Oracea®) Doxycycline monohydrate 75 mg capsule (Monodox®) Doxycycline monohydrate 150 mg capsule (Adoxa®) Doxycycline monohydrate 50 mg tablet (Adoxa®) Doxycycline monohydrate 75 mg tablet (Adoxa®) Doxycycline monohydrate 100 mg tablet (Adoxa®) Doxycycline monohydrate 150 mg tablet (Adoxa®) Minocycline HCl 50 mg tablet (Dynacin®) Minocycline HCl 75 mg tablet (Dynacin®) Minocycline HCl 100 mg tablet (Dynacin®) Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Doxylamine 5mg Chewable Tablet (Aldex AN) (Implemented 09/28/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Patients </= 6 years of age that cannot swallow a solid oral dosage form. Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Doxylamine Succinate and Pyridoxine (Diclegis DR 1010) (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Diclegis Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Dronabinol (Marinol) (Implemented 06/27/2007) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Criterion 1: Age > 18 years of age, AND Submitted ICD-9 diagnosis HIV within the past 730 days, AND Submitted ICD-9 diagnosis for cachexia within the past 730 days, AND At least three paid drug claims in history identifying antiretrovirals (either as single entity or combo drug) within the past 31 days, AND Paid claim for megestrol acetate (Megace) within the past 31 days (four weeks) (Showing concomitant treatment) Criterion 2: Age > 18 years of age, AND Submitted ICD-9 diagnosis HIV within the past 730 days, AND Submitted ICD-9 diagnosis for cachexia within the past 730 days , AND At least three paid drug claims in history identifying antiretrovirals (either as single entity or combo drug) within the past 31 days. Criterion 3: Age > 18 years of age, AND Submitted ICD-9 diagnosis malignant cancer within the past 365 days AND o Paid drug claim in history for antineoplastic agents within the past 45 days OR o Procedure code indicating radiation treatment within the past 45 days AND Paid drug claim in history within the past 45 days for an oral 5-HT3 (serotonin) receptor antagonist, OR Paid drug claim in history within the past 45 days for a NK1 (neurokinin-1) receptor antagonist Denial criteria Absence of approval criteria Submitted ICD-9 diagnosis bipolar in medical history. Submitted ICD-9 diagnosis depression in medical history. Submitted ICD-9 diagnosis schizophrenia in medical history. Submitted ICD-9 diagnosis substance or alcohol abuse in medical history. Additional criteria Quantity limits apply Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Droxidopa (Northera) Capsule (Implemented 01/13/2015) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Northera Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Efinaconazole 10% Topical Solution (Jublia) (Implemented 06/11/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Itraconazole Capsule [Sporanox] Terbinafine Tablet [Lamisil] Drugs that require manual PA Ciclopirox 8% Topical Solution [Penlac] Efinaconazole 10% Topical Solution [Jublia] Tavaborole 5% Topical Solution [Kerydin] Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Eliglustat (Cerdelga) Capsule (Implemented 01/13/2015) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual PA Eliglustat (Cerdelga) Capsule Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Eltrombopag Olamine Tablet (Promacta) (Implemented 07/09/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Promacta Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Emtricitabine (Emtriva) (Implemented 01/12/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria for treatment of HIV-1 infection A billed diagnosis of HIV/AIDS in the Medicaid history in previous 2 years; OR Paid drug claim(s) in Medicaid history of other antiretroviral therapy (ART), such as non-nucleoside reverse transcriptase inhibitors (NNRTI), OR protease inhibitors (PI), OR integrase strand transfer inhibitor (INSTI) in previous 6 months. If there is no HIV/AIDS diagnosis in Medicaid history and no records of other ART in the Medicaid drug profile, prescriber will be required to follow the manual review process and submit documentation confirming positive HIV diagnosis. Denial criteria Therapeutic duplication edit: paid claim within previous 30 days for Truvada; OR Absence of approval criteria. Approval criteria for PrEp will require a manual review PA process based upon the following: Documentation from prescriber that patient is at high risk for acquiring HIV infection; AND Negative HIV test before starting and every 3 months thereafter; AND Pregnancy test before starting and every 3 months thereafter. If pregnant, provide documentation of patient understanding of potential risks and benefits of using Truvada®, including contraindication with breastfeeding; AND Serum creatinine lab tests obtained prior to initiation, then every 6 months. Creatinine clearance should be >60 mL/min; AND Documented testing for Hepatitis B Virus (HBV) and results submitted. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Entacapone (Comtan) (Implemented 01/12/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria At least 1 paid Medicaid claim for Sinemet (carbidopa/levodopa) in the past 60 days, AND No therapeutic duplication with Stalevo. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Entecavir (Baraclude) (Implemented 09/24/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria No history of HIV/AIDS diagnosis in medical history, OR HIV/AIDS diagnosis in medical history, AND At least one paid Medicaid drug claim for antiretroviral in past 45 days Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Enzalutamide (Xtandi) (Implemented 12/19/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Xtandi Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Eslicarbazepine (Aptiom) (Implemented 07/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Aptiom 200 mg Tablet Aptiom 400 mg Tablet Aptiom 600 mg Tablet Aptiom 800 mg Tablet Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Erythropoiesis stimulating agents (Implemented 03/26/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local) Drugs that require manual PA Darbepoetin alfa [Aranesp] Epoetin alfa [Epogen] Epoetin alfa [Procrit] PA criteria Prescriber must fax labwork with a recent hemoglobin level for approval consideration. Fax documentation to 501-372-2971. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Estrogen-replacement agents (Implemented 07/11/2008) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents Estradiol 0.5mg, 1mg, 2mg oral tablet (Estrace) Estropipate oral tablet (Ogen) Nonpreferred agents Estradiol acetate tablet (Femtrace) Estradiol acetate vaginal ring (Femring) Estradiol 1.5mg oral tablet (Estrace) Estradiol spray (Evamist) Estradiol topical gel (Divigel) Estradiol transdermal (Alora, Climara) Estradiol vaginal ring (Estring) Estradiol vaginal tablet (Vagifem) Estrogens, conjugated (Cenestin, Ejuvia, Premarin) Estrogens, conjugated/Bazedoxifene (Duavee) Estrogens, esterified (Menest) Nonpreferred agents with criteria Estradiol/drospirenone (Angeliq) Estradiol/levonorgestrel (Climara Pro) Estradiol/norethindrone (Activella) Estradiol/norgestimate (Prefest) Estrogens, conjugated/medroxyprogesterone (Premphase, Prempro) Ethinyl estradiol/norethindrone acetate (Femhrt) Approval criteria for nonpreferred agents with criteria ≥ 120 days of therapy in the previous 180 days for the same drug, strength, and dosage form Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Everolimus Tablet (Afinitor) (Implemented 07/23/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drug that requires a manual review for prior authorization Afinitor Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Famotidine 40mg/5ml oral suspension (Pepcid) (Implemented 09/24/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria At least two paid Medicaid drug claims for ranitidine syrup in the past 60 days, AND ≤ 6 years of age, OR NPO (Appendix A) Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Febuxostat Tablet (Uloric) (Implemented 07/09/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Uloric Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Fenofibrate and Fenofibric Acid Products (Implemented 01/18/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization FENOFIBRATE 54 MG ORAL TABLET [LOFIBRA] FENOFIBRATE 67 MG ORAL CAPSULE [LOFIBRA] FENOFIBRATE 160 MG ORAL TABLET [LOFIBRA] GEMFIBROZIL 600 MG ORAL TABLET [LOPID] Drugs that require manual review for prior authorization ANTARA 30 MG, 43 MG, 90 MG, 130 MG ORAL CAPSULE (fenofibrate micronized) FIBRICOR 35 MG, 105 MG ORAL TABLET (fenofibric acid) LIPOFEN 50 MG, 150 MG ORAL CAPSULE (fenofibrate) LOFIBRA 134 MG, 200 MG ORAL CAPSULE (fenofibrate micronized) TRICOR 48 MG, 145 MG ORAL TABLET (fenofibrate nanocrystallized) TRIGLIDE 50 MG, 160 MG ORAL TABLET (fenofibrate nanocrystallized) TRILIPIX DR 45 MG, 135 MG ORAL CAPSULE (fenofibric acid) Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Fentanyl Buccal Tablet (Fentora and Onsolis) (Implemented 04/27/2005) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria >/=18 years of age, AND Opioid Tolerance of a ceiling dose for ≥ seven days during the past 30 days of one of the following: Fentanyl patch, Hydromorphone LA, Morphine LA, or Oxycodone LA AND Cancer with malignancies as determined by: o Diagnosis in past two years, OR o Antineoplastic agents in the past 365 days Denial criteria < 18 years of age, OR Remaining estimated days supply of fentanyl buccal tablet in history is > 25% Thereapeutic duplication with Abstral Therapeutic duplication with of Actiq Therapeutic duplication with other strengths of Fentora Therapeutic duplication with other strengths of Onsolis Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Fentanyl Nasal Spray (Lazanda) (Implemented 09/23/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Lazanda 100mcg Nasal Spray Lazanda 400mcg Nasal Spray Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Fentanyl 100mcg Sublingual Tablet (Abstral) (Implemented 01/31/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria >/=18 years of age, AND Opioid Tolerance of a ceiling dose for ≥ seven days during the past 30 days of one of the following: Fentanyl patch, Hydromorphone LA, Morphine LA, or Oxycodone LA AND Cancer with malignancies as determined by: o Diagnosis in past two years, OR o Antineoplastic agents in the past 365 days Denial criteria < 18 years of age, OR Remaining estimated days supply of fentanyl buccal tablet in history is > 25% Thereapeutic duplication with other strengths of Abstral Therapeutic duplication with Actiq Therapeutic duplication with Fentora Therapeutic duplication with Onsolis Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Fentanyl Sublingual Spray (Subsys) (Implemented 01/31/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria >/=18 years of age, AND Opioid Tolerance of a ceiling dose for ≥ seven days during the past 30 days of one of the following: Fentanyl patch, Hydromorphone LA, Morphine LA, or Oxycodone LA AND Cancer with malignancies as determined by: o Diagnosis in past two years, OR o Antineoplastic agents in the past 365 days Denial criteria < 18 years of age, OR Remaining estimated days supply of fentanyl sublingual spray in history is > 25% Thereapeutic duplication with Abstral Therapeutic duplication with Actiq Therapeutic duplication with Fentora Therapeutic duplication with Onsolis Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Fentanyl citrate oral transmucosal (Actiq) (Implemented 04/27/2005) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria >/= 16 years of age, AND Opioid tolerance of a ceiling dose for ≥ seven days during the past 30 days of one of the following: Fentanyl patch, Hydromorphone LA, Morphine LA, or Oxycodone LA AND Cancer with malignancies as determined by: o Diagnosis in past two years, OR o Antineoplastic Agents in the past 365 days Denial criteria < 16 years of age, OR Remaining estimated days supply of fentanyl citrate transmucosal in history is > 25% Therapeutic duplication with other strengths of Actiq Therapeutic duplication with Fentora Therapeutic duplication with Onsolis Thereapeutic duplication with Abstral Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Fibromyalgia Agents (Implemented 09/20/2011) The non-preferred antiepileptic medications will be considered non-preferred for treating fibromyalgia and neuropathic pain only. Medications listed as either preferred or non-preferred status in this category may or may not include an FDA approved indication for fibromyalgia or neuropathic pain. Use of these medications for fibromyalgia, neuralgias, and neuropathic pain has been reviewed through the evidence-based review process. Medications listed in this category as either preferred or nonpreferred status are not to be construed as endorsements for marketing of off-label use by the manufacturer or by Medicaid. Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents Amitriptyline HCl (Elavil) Cyclobenzaprine 10mg tablet (Flexeril) Gabapentin 100mg, 300mg, 400mg capsule (Neurontin) Nortriptyline HCl (Pamelor) Preferred agents with criteria Citalopram hydrobromide (Celexa) – see Second generation antidepressant Fluoxetine 10mg, 20mg capsule, 20mg/5ml solution (Prozac) – see Second generation antidepressant Paroxetine HCl immediate-release (Paxil) - Second generation antidepressant Nonpreferred agents with criteria: Buproprion - all dosage forms (Aplenzin, Wellbutrin) – see Second generation antidepressant Carbamazepine - all dosage forms (Tegretol) – see Neuropathic pain agents Cyclobenzaprine 5mg, 7.5mg tablet, extended-release capsule (Amrix, Fexmid, Flexeril) – see Skeletal muscle relaxants Desipramine HCl (Norpramin) Desvenlafaxine succinate (Pristiq) – see Second generation antidepressant Duloxetine HCl (Cymbalta) – see Second generation antidepressant Escitalopram oxalate (Lexapro) – see Second generation antidepressant Ethotoin (Peganone) Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Fluoxetine HCl 10mg, 15mg, 20mg Tablet; 40mg capsule; and 90mg weekly capsule (Prozac) – see Second generation antidepressant Fluvoxamine maleate – all dosage forms (Luvox) – see Second generation antidepressant Gabapentin 250mg/5ml solution, 600mg, 800mg tablet – see Neuropathic pain agents Imipramine HCl and pamoate (Tofranil) Lacosamide (Vimpat) – see Neuropathic pain agents Lamotrigine (Lamictal) – see Neuropathic pain agents Levetiracetam (Keppra) Milnacipran HCl (Savella) – see Second generation antidepressant Mirtazapine (Remeron) – see Second generation antidepressant Nefazodone HCl (Serzone) – see Second generation antidepressant Oxcarbazepine (Trileptal) – see Neuropathic pain agents Paroxetine HCl controlled-release tablet, and 10mg/5ml suspension (Paxil CR) – see Second generation antidepressant Paroxetine mesylate (Pexeva) – see Second generation antidepressant Phenytoin 100mg extended-release capsule (Dilantin) Pregabalin (Lyrica) – see Neuropathic pain agents Sertraline HCl (Zoloft) – see Second generation antidepressant Tiagabine (Gabitril) – Valproic Acid (Depakene, Stavzor) – see Neuropathic pain agents Venlafaxine, all dosage forms (Effexor, Effexor XR) – see Second generation antidepressant Zonisamide (Zonegran) Approval criteria for nonpreferred agents with criteria No diagnosis of myalgia and myositis, unspecified (ICD-9 729.1) in the past three years Drugs with a link to neuropathic pain agents, second generation antidepressants, or skeletal muscle relaxants have additional criteria to meet for approval Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Fidaxomicin (Dificid) (Implemented 01/12/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria >/= 18 years of age, AND At least 1 paid claim in Medicaid history for Vancomycin (oral or injectable compounded for oral use) in the previous 10-30 days. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Finasteride Tablet (Proscar) (Implemented 07/09/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of Benign Prostatic Hypertrophy in the past 3 years Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Fluorouracil Solution/Cream (Efudex) (Implemented 06/21/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval for Fluorouracil 2% Solution: Diagnosis of Actinic Keratosis in Medicaid history in the past 2 months, AND No therapeutic duplication with Diclofenac Sodium 3% Gel [Solaraze], OR No therapeutic duplication with other strengths of Fluorouracil Cream or Solution [Carac, Efudex, Fluoroplex], OR No therapeutic duplication with Imiquimod Cream [Aldara, Zyclara], OR No therapeutic duplication with Ingenol Gel [Picato] Approval for Fluorouracil 5% Cream or Solution: Diagnosis of Actinic Keratosis or Basal Cell Carcinoma in Medicaid history in the past 2 months AND No therapeutic duplication with Diclofenac Sodium 3% Gel [Solaraze], OR No therapeutic duplication with other strengths of Fluorouracil Cream or Solution [Carac, Efudex, Fluoroplex], OR No therapeutic duplication with Imiquimod Cream [Aldara, Zyclara], OR No therapeutic duplication with Ingenol Gel [Picato] Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Fluorouracil Cream (Carac 0.5% and Fluoroplex 1%) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval for fluorouracil 0.5% and 1% Cream: Diagnosis of Actinic Keratosis in Medicaid history in the past 2 months, AND No therapeutic duplication with Diclofenac Sodium 3% Gel [Solaraze], OR No therapeutic duplication with other strengths of Fluorouracil Cream or Solution [Carac, Efudex, Fluoroplex], OR No therapeutic duplication with Imiquimod Cream [Aldara, Zyclara], OR No therapeutic duplication with Ingenol Gel [Picato] Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Fosamprenavir Calcium (Lexiva) Tablet (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval for Lexiva Tablet: If the Medicaid recipient does not have a ritonavir claim in Medicaid history in the previous 45 days, a maximum quantity of 4 tablets per day will be allowed and a cumulative quantity of 124 tablets per 31 days. If the Medicaid recipient does have a ritonavir claim in Medicaid history in the previous 45 days, a maximum quantity of 2 tablets per day will be allowed and a cumulative quantity of 62 per 31 days. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Fosamprenavir Calcium (Lexiva) 50mg/5ml Suspension (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval for Oral suspension 50 mg/5 ml: < 6 years of age NPO diagnosis in Medicaid history. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Galantamine ER (Razadyne ER) (Implemented 04/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria >/= 90 days of Galantamine ER therapy in the past 120 days Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Galantamine Solution (Razadyne) (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval for Razadyne: Age > 50 years of age, AND Diagnosis of NPO in the Medicaid History Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Glycerol Phenylbutyrate Liquid (Ravicti) (Implemented 07/09/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Ravicti Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Glycophos 20ml Vial (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Glycophos 20ml vial Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Glycopyrrolate 0.2 mg/ml vial (Implemented 07/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Glycopyrrolate 1 mg/5 ml oral solution (Cuvposa) Drugs that require manual review for prior authorization Glycopyrrolate 0.2 mg/ml vial Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Glycopyrrolate 1.5mg Tablet (Glycate) (Implemented 03/18/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drug that requires a manual review for prior authorization Glycate 1.5mg Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Hemorrhoid Preparations (Implemented 01/12/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria History of at least three claims for three different products that do not require prior authorization within the previous 60 days. LABELER CODE 65649 68220 10631 64980 68220 Link to Memorandum Top of the document LABEL NAME ANUSOL-HC 2.5% CREAM PROCTOFOAM-HC 1%-1% FOAM PROCTOSOL-HC 2.5% CREAM PROCTOZONE-HC 2.5% CREAM CORTIFOAM 10% AEROSOL PA STATUS No PA No PA No PA No PA PA Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Hepatitis C Medications (Implemented 10/21/2009) Prescribers are required to complete the HCV Statement of Medical Necessity and fax to 501-683-4124. Drugs that require a manual PA Incivek 375 mg tablet Infergen 9mcg/0.3mls Infergen 15mcg/0.5mls Olysio (Simeprevir) Pegasys 180mcg (1 ml vial) Pegasys 180mcg (1 kit) – four doses Peg Intron 50mcg (1 kit) Peg Intron 80mcg (1 kit) Peg Intron 120mcg (1 kit) Peg Intron 150mcg (1 kit) Peg Intron 50mcg Redipen (1 kit) Peg Intron 80mcg Redipen (1 kit) Peg Intron 120mcg Redipen (1 kit) Peg Intron 150mcg Redipen (1 kit) Ribavirin 200mg capsule Ribavirin 200mg tablet Rebetol 40mg/ml solution Ribasphere 400mg tablet Ribasphere 600mg tablet Ribapak 400-400mg dosepack Ribapak 400-600mg dosepack Sovaldi (Sofosbuvir) Victrelis 200mg capsule Link to Hepatitis C prior authorization form—Microsoft Word format (.doc) Link to Hepatitis C prior authorization form—Portable Document Format (.pdf) Link to Memorandum Link to Memorandum Link to Updated Criteria Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria HMG-CoA Reductase Inhibitors (Implemented 06/10/2008) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents Atorvastatin calcium (Lipitor) Pravastatin sodium (Pravachol) Simvastatin 5mg, 10mg, 20mg, 40mg (Zocor) Preferred agents with criteria Simvastatin 80mg (Zocor) Nonpreferred agents Atorvastatin calcium/Ezetimibe (Liptruzet) Fluvastatin sodium (Lescol) Lovastatin (Mevacor) Lovastatin/Niacin extended-release (Advicor) Pitavastatin calclium (Livalo) Rosuvastatin calcium (Crestor) Simvastatin/Ezetimibe (Vytorin) Simvastatin/Niacin (Simcor) Simvastatin/Sitagliptin (Juvisync) Approval criteria for agents with criteria Simvastatin 80mg Ten or more claims for Sivastatin 80mg within past twelve months Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Hydrocodone ER Capsule (Zohydro ER) (Implemented 07/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Zohydro ER Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Hydroxypropyl Cellulose 5mg Eye Insert (Lacrisert) (Implemented 01/18/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of one of the following diagnoses associated with dry eye in the past two years: Keratoconjunctivits sicca, non-Sjogren’s syndrome Keratoconjunctivits sicca, Sjogren’s syndrome Keratoconjunctivitis, exposure Tear film insufficiency, unspecified (Dry eye syndrome) Xerosis Denial criteria Therapeutic duplication with Restasis (Cyclosporine) Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Ibrutinib (Imbruvica) Capsule (Implemented 03/18/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drug that requires a manual review for prior authorization Imbruvica 140mg Capsule Additional Criteria Quantity Limits Apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Icatibant (Firazyr) (Implemented 01/12/2012) Providers requesting a Prior Authorization (PA) should call the Medicaid Pharmacy Program, 501-683-4120. Drugs that require manual review for prior authorization Firazyr Additional criteria Quantity limits apply Age >17 years of Age Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Icosapent Ethyl Capsule (Vascepa) (Implemented 07/09/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Vascepa Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Idelaisib (Zydelig) Tablet (Implemented 01/13/2015) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Zydelig Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Imiquimod (Aldara) (Implemented 06/27/2007) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria > 12 years of age AND, Submitted ICD-9 diagnosis for superficial basal cell carcinoma (sBCC) within past two months, OR Submitted ICD-9 diagnosis for actinic keratosis (AK) within past two months, OR Submitted ICD-9 diagnosis for Condyloma Acuminata (or commonly known as external genital or perianal warts) within past two months, AND No Therapeutic Duplication with Diclofenac Sodium 3% gel No Therapeutic Duplication with Fluorouracil Cream/Solution Topical No Therapeutic Duplication with other strengths of Imiquimod Cream Topical No Therapeutic Duplication with Ingenol gel Topical Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Imiquimod (Zyclara) (Implemented 04/27/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria > 12 years of age, AND Submitted ICD-9 diagnosis for actinic keratosis (AK) within past two months, OR Submitted ICD-9 diagnosis for Condyloma Acuminanta (commonly known as external genital or perianal warts) within past two months, AND No Therapeutic Duplication with Diclofenac Sodium 3% gel No Therapeutic Duplication with Fluorouracil Cream/Solution Topical No Therapeutic Duplication with other strengths of Imiquimod Cream Topical No Therapeutic Duplication with Ingenol gel Topical Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Immunologic Agents (Multiple Sclerosis) (Implemented 09/27/2011) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents Glatiramer acetate 20 mg injection (Copaxone®) Interferon Beta – 1A injection (Avonex®) Non-Preferred agents Dimethyl fumarate capsule (Tecfidera®) Glatiramer acetate 40 mg injection (Copaxone®) Fingolimod HCl capsule (Gilenya®) Interferon Beta – 1A/albumin (Rebif®) Interferon Beta – 1B injection (Betaseron®) Interferon Beta – 1B kit (Extavia®) Peginterferon Beta – 1A (Plegridy®) Teriflunomide tablet (Aubagio®) Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Ingenol Mebutate (Picato Gel) (Implemented 03/08/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria >/= to 18 Years of Age Submitted ICD-9 diagnosis for actinic keratosis (AK) within past two months, AND No Therapeutic Duplication with Diclofenac Sodium 3% gel No Therapeutic Duplication with Fluorouracil Cream/Solution Topical No Therapeutic Duplication with Imiquimod Cream Topical No Therapeutic Duplication with other strengths of Ingenol gel Topical Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Insulin Pens (Implemented 04/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria applies to all strengths of the following insulin pens Apridra® (insulin glulisine) Humalog® (insulin lispro) Humalog® Mix 50-50 Humalog® Mix 75-25 Humulin® (insulin human) N Humulin® R, Lantus® (insulin glargine) Levemir® (insulin detemir) Novolin® (human insulin) 70-30 Novolin® N Novolin® R NovoLog® (insulin aspart) NovoLog® Mix 70-30 Relion Humulin® 70-30 Relion Humulin® N Relion Humulin® R Relion Novolin® 70-30 Relion Novolin® N Approval criteria Criterion 1 ≤ 18 years of age, AND Not currently in LTC, AND Quantity </= 15 ml per 31-day supply. Criterion 2 ≤ 18 years of age, AND Currently in LTC, AND Medicaid paid claims for ≥ 90 days of the same insulin pen formulation in the previous 120 days, AND Quantity </= 15 ml per 31-day supply. Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Criterion 3 >18 years of age, AND Medicaid paid claims for ≥ 90 days of the same insulin pen formulation in the previous 120 days. Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Isosorbide Dinitrate/Hydralazine (BiDil) (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization BiDil Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Itraconazle (Onmel) 200mg Tablet (Implemented 03/19/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Onmel Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Isotretinoin Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require a manual PA Isotretinoin 10 mg (Amnesteem, Claravis) Isotretinoin 20 mg (Amnesteem, Claravis, Sotret) Isotretinoin 30 mg (Claravis) Isotretinoin 40 mg (Amnesteem, Claravis) Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Itrazonazole Oral Solution (Sporanox) (Implemented 10/11/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria History of at least two claims for fluconazole (tablets or suspension) in the previous 7-30 days, OR One claim each of Nystatin Suspension and fluconazole (tablets or suspension) in the previous 7-30 days, OR NPO diagnosis in Medicaid history Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Ivacaftor Tablet (Kalydeco) (Implemented 07/09/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Kalydeco Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Ivermectin 0.5% Lotion (Sklice) (Implemented 06/18/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Sklice 0.5% Lotion Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Kits (Implemented 08/17/2010) All requests for “kits” or “combo pack” products (products that consist of packaging multiple products under one NDC) require a manual review. The underlined individual active ingredients (listed next to the product) do not require a PA. Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require a manual PA CICLODAN 0.77% CREAM KIT (CICLOPIROX 0.77% CREAM – HAIR & BODY CLEANSER) CICLOPIROX 8% KIT (CICLOPIROX 8% TOPICAL SOLUTION NAIL LACQUER – VITAMIN E CAPSULES – LACQUER REMOVER) CENTANY AT 2% OINMENT KIT (MUPIROCON 2% OINTMENT – STERILE GAUZE – TAPE) CNL 8 NAIL KIT (CICLOPIROX 8% TOPICAL SOLUTION NAIL LACQUER – EMORY BOARD – LACQUER REMOVER) HALONATE PAC COMBO PACK (HALOBETASOL PROPIONATE 0.5% OINTMENT – AMMONIUM LACTATE 12% LOTION) KETODAN 2% FOAM KIT (KETOCONAZOLE 2% TOPICAL FOAM – HAIR & BODY CLEANSER) LIDOCAINE-PRILOCAINE 2.5%-2.5% CREAM KIT (LIDOCAINPRILOCAINE 2.5%-2.5% CREAM – OCCLUSIVE DRESSINGS) PEDIADERM AF KIT (NYSTATIN 100,000 UNITS/GRAM CREAM – EMOLLIENT DIAPER CREAM) PEDIADERM HC 2% KIT (HYDROCORTISONE 2% LOTION – EMOLLIENT DIAPER CREAM) PEDIADERM TA 0.1% KIT (TRIAMCINOLONE ACETONIDE 0.1% CREAM – EMOLLIENT DIAPER CREAM) PEDIPIROX-4 NAIL KIT (CICLOPIROX 8% TOPICAL SOLUTION NAIL LACQUER – ALCOHOL NAIL LACQUER REMOVAL PADS) ROSADAN 0.75% CREAM KIT (METRONIDAZOLE 0.75% CREAM – MOISTURIZING SKIN WASH) ROSADAN 0.75% GEL KIT (METRONIDAZOLE 0.75% GEL – MOISTURIZING SKIN WASH) ROWASA 4 GM/60 ML ENEMA KIT (MESALAMINE 4 GM/60 ML ENEMA – CLEANSING WIPES) Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria SYNALAR 0.025% CREAM KIT (FLUOCINOLONE ACETONIDE 0.025% TOPICAL CREAM – EMOLLIENT CREAM) SYNALAR 0.025% OINTMENT KIT (FLUOCINOLONE ACETONIDE 0.025% TOPICAL OINTMENT – EMOLLIENT CREAM) SYNAGLAR TS 0.01% KIT (FLUOCINOLONE ACETONIDE 0.01% TOPICAL SOLUTION – HAIR & BODY CLEANSER) TERBINEX KIT (TERBINAFINE HCL 250 ORAL TABLETS – HYDROXYPROPYL-CHITOSAN 1% NAIL LACQUER) ULTRAVATE X CREAM COMBO PACK (HALOBETASOL PROPIONATE 0.05% TOPICAL CREAM – AMMONIUM LACTATE 10% MOISTURIZING CREAM) ULTRAVATE X OINTMENT COMBO PACK (HALOBETASOL PROPIONATE 0.05% TOPICAL OINTMENT – AMMONIUM LACTATE 10% MOISTURIZING CREAM) Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Lamotrigine Kits (Lamictal Start and Patient Titration Kits) (Implemented 03/18/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization* if being used for approved diagnosis Lamotrigine Tablets* Drugs that require manual review for prior authorization Lamictal ODT Start Kit (Blue) contains 25mg and 50mg tablets Lamictal ODT Start Kit (Green)contains 50mg and 100mg tablets Lamictal ODT Start Kit (Orange)contains 25mg, 50mg, and 100mg tablets Lamictal Tablet Start Kit (Blue) contains 25mg tablets Lamictal Tablet Start Kit (Green) contains 25mg and 100mg tablets Lamictal Tablet Start Kit (Orange) contains 25mg and 100mg tablets Lamictal XR Start Kit (Blue) contains 25mg and 50mg extended-release tablets Lamictal XR Start Kit (Green) contains 50mg, 100mg, and 200mg extended-release tablets Lamictal XR Start Kit (Orange) contains 25mg, 50mg, and 100mg extended-release tablets Lamictal Start Kits are designed for titration during the first 5 weeks of therapy and contain a single entity drug that typically does not require prior authorization. Daily dosing instructions are determined by the patient’s current antiepileptic regimen. Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Lansoprazole, Amoxicillin, and Clarithromycin combination (Prevpac) (Implemented 01/12/2005) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Criterion 1: No history of lansoprazole, amoxicillin, and clarithromycin combination (Prevpac) in the last 365 days, AND, No history of omeprazole, amoxicillin, and clarithromycin combination (Prevpac) in the last 365 days, AND, No concurrent proton pump inhibitor therapy within the past 30 days. Criterion 2: No history of lansoprazole, amoxicillin, and clarithromycin combination (Prevpac) in the last 365 days, AND, No history of omeprazole, amoxicillin, and clarithromycin combination (Prevpac) in the last 365 days, AND, No concurrent proton pump inhibitor therapy within the past 30 days. Criterion 3: No history of metronidazole/tetracycline/bismuth combination (Helidac) in the last 365 days, OR No history of metronidazole/tetracycline/bismuth combination (Pylera) in the last 365 days. Denial criteria Criterion 1: History of lansoprazole, amoxicillin, and clarithromycin combination (Prevpac) in the last 365 days, OR Current proton pump inhibitor therapy within the past 30 days Criterion 2: No history of lansoprazole, amoxicillin, and clarithromycin combination (Prevpac) in the last 365 days, AND, No history of omeprazole, amoxicillin, and clarithromycin combination (Prevpac) in the last 365 days, AND, No concurrent proton pump inhibitor therapy within the past 30 days. Criterion 3: No history of metronidazole/tetracycline/bismuth combination (Helidac) in the last 365 days, OR No history of metronidazole/tetracycline/bismuth combination (Pylera) in the last 365 days. Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Leukotriene Receptor Antagonists (Implemented 08/11/2009) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agent with criteria Montelukast sodium (Singulair) Nonpreferred agents Zafirlukast (Accolate) Zileuton (Zyflo) Approval criteria for preferred agents Criterion 1: A. If a paid drug claim in history for an inhaled corticosteroid, long-acting beta2 agonist/inhaled corticosteroid, or short-acting beta2 agonist in the last 365 days, then B. One of the following criteria below: < Two claims for short-acting beta2 in the last 365 days, AND < One claim for an oral corticosteroid in the last 183 days OR IF the patient exceeds any of the above criteria, then the asthma patient must have a claim for an inhaled asthma controller (ICS or ICS/LABA) in Medicaid drug history in last 45 days. OR Criterion 2: A. If no paid drug claim in history for an inhaled corticosteroid, long-acting beta2 agonist/inhaled corticosteroid, or short-acting beta2 agonist in the last 365 days, then B. One of the following criteria below: > One claim for an inhaled nasal steroid from the 7th day to the124th day in Medicaid history, OR > One claim for a second generation antihistamine from the 7th day to the124th day in Medicaid history OR Criterion 3: A. A diagnosis code for COPD (Appendix G) in patient history in the past 2 years AND patient is greater than 18 years old AND B. One of the following criteria below: > One claim for an inhaled nasal steroid from the 7th day to the124th day in Medicaid history, OR Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria > One claim for a second generation antihistamine from the 7th day to the124th day in Medicaid history Denial criteria Failure to meet approval criteria Therapeutic duplication with a LTRA other than the one on the incoming claim if >25% of the days supply of the claim in history remains Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Levetiracetam ER (Keppra ER) (Implemented 04/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria >/= 90 days of Levetiracetam ER therapy in the past 120 days Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Levofloxacin 500mg/20ml U.D. Cup (Implemented 04/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Currently LTC Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Levothyroxine Capsule (Tirosint) (Implemented 08/17/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization All strengths of generic levothyroxine tablet Drugs that require manual review for prior authorization Tirosint Capsule Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Levothyroxine Vial (Implemented 04/17/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Levothyroxine 200 mcg vial Levothyroxine 500 mcg vial Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Lidocaine-Prilocaine 2.5%-2.5% Cream (Emla) (Implemented 07/09/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Lithium ER or SA (Implemented 04/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria >/= 90 days of Lithium ER or Lithium SA therapy in the past 120 days Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Linacoltide (Linzess) (Implemented 04/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Criterion 1 > 18 years of age, AND Paid drug claim for Linzess (Linacoltide) within the past 60 days, Criterion 2: > 18 years of age, AND Paid claim for 30 day supply of polyethylene glycol 3350 (Miralax, Glycolax) within previous 45 days, AND Denial criteria Absence of approval criteria History of mechanical gastrointestinal obstruction Age < 18 years of age >/=1 Paid claim for an opioid in the past 60 days Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Lomitapide Mesylate Capsule (Juxtapid) (Implemented 07/09/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Juxtapid Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Low Molecular Weight Heparins (Dalteparin [Fragmin], Enoxaparin [Lovenox], Tinzaparin [Innohep]) (Implemented 04/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria No Therapeutic Duplication with Xarelto 10mg, AND No Therapeutic Duplication with other Low Molecular Weight Heparins Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Lindane (Implemented 03/22/2006) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Lindane Shampoo History of at least one permethrin or pyrethrin/piperonyl in the last 28 days which can include a combination of any of the following: o Two permethrin claims OR, o Two pyrethrin-piperonyl claims OR, o One permethrin and one pyrethrin-piperonyl claims, OR o One permethrin claim and one malathion claim, OR, o One pyrethrin-piperonyl claim and one malathion claim, AND Patient >/= 13 years old Lindane Lotion History of at least one permethrin or pyrethrin-piperonyl in the last 28 days, AND Patient >/= 13 years old Denial criteria Patient < 13 years of age, OR Previous claim history of Lindane in the last 180 days Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Lubiprostone (Amitiza) (Implemented 06/27/2007) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Criterion 1: > 18 years of age, AND Paid drug claim for Amitiza (Lubiprostone) within the past 60 days Criterion 2: > 18 years of age, AND Paid claim for 30 day supply of polyethylene glycol 3350 (Miralax, Glycolax) within previous 45 days, AND Amitiza dose of one tablet per day Denial criteria Absence of approval criteria History of mechanical gastrointestinal obstruction Age < 18 years of age Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Leuprolied/Norethindrone (Lupaneta) 2.5-5mg 1 month kit and 11.25-5mg 3 month kit (Implemented 07/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Billed diagnosis of endometriosis or uterine leiomyoma (fibroids) AND <12 billed claims of 3.75mg leuprolide injection (which would include Lupaneta®) in the previous 3 year Medicaid history, OR < 4 billed claims of 11.25mg leuprolide injection (which would include Lupaneta®) in the previous 3 year Medicaid history, AND No Therapeutic Duplication with other strengths of Lupron. Denial Criterion Diagnosis of infertility in Medicaid history (3 year look back), OR Thrombophlebitis, OR Thromboembolic disorders, OR Cerebral apoplexy in Medicaid history; OR Carcinoma of the breast in Medicaid history. Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Lupron (Implemented 06/21/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Lupron depot 11.25 mg 3 mo kit Lupron depot 22.5 mg 3 mo kit Lupron depot 3.75 mg kit Lupron depot 7.5 mg kit Lupron depot-4 month kit Lupron depot-ped 11.25 mg kit Lupron depot-ped 15 mg kit Lupron depot-ped 7.5 mg kit Criterion 1 for Lupron-Depot PED® 7.5 mg, 11.25 mg, and 15 mg: Diagnosis of Central Precocious Puberty (CPP) in Medicaid History, AND Girls <11 Years of Age or Boys <12 Years of Age, AND Procedure code for CT scan of the head within last 2 years, AND Procedure code in Medicaid History for Ultrasound of pelvic/adrenal/testicular area within last 2 years, AND Procedure code in Medicaid History for Bone Age study in the last 2 years, AND No Therapeutic Duplication with other strengths of Lupron. Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Criterion 2 for Lupron-Depot® 3.75 mg, and 11.25 mg-3 month • Billed diagnosis of endometriosis or uterine leiomyoma (fibroids) AND • <12 billed claims of 3.75mg leuprolide injection (which would include Lupaneta®) in the previous 3 year Medicaid history, OR • < 4 billed claims of 11.25mg leuprolide injection (which would include Lupaneta®) in the previous 3 year Medicaid history AND No Therapeutic Duplication with other strengths of Lupron. Denial Criterion Diagnosis of infertility Criterion 3 for Lupron-Depot® 7.5 mg, 22.5 mg-3 month, 30 mg-4 month, 45mg-6 month, and Lupron 2 week Kit Diagnosis in Medicaid History of prostate cancer within last 2 years, AND No Therapeutic Duplication with other strengths of Lupron. Criterion 4 for Lupron-Depot 3.75 mg, 7.5 mg, 11.25 mg-3 month Diagnosis in Medicaid History of breast cancer or ovarian cancer in the last 2 years, AND No Therapeutic Duplication with other strengths of Lupron. Additional criteria Quantity limits apply Lupron-Ped® criteria revision The point-of-sale approval requirement of the GnRH stimulation test for CPP will be removed. Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Point-of-sale continuation criteria Lupron-Ped® injections: for those CPP recipients previously approved who have 3 Lupron-Ped® paid claims in the previous 6 months AND who are < age 11 years for females and < age of 12 years for males. The continuation criteria were implemented on Nov. 16, 2011. Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Malathion (Ovide) (Implemented 03/22/2006) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Malathion Lotion > 6 years of age, AND History of at least one paid drug claim of permethrin or pyrethrin-piperonyl in the last 28 days Denial criteria < 6 years of age, OR History of one paid claim of malathion lotion in the last seven days, OR History of two paid claims of malathion lotion in the last 28 days Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Macitentan (Opsumit) Tablet (Implemented 03/18/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drug that requires a manual review for prior authorization Opsumit 10 mg Additional Criteria Quantity Limits Apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Maraviroc (Selzentry) (Implemented 04/12/2011) Prescribers are required to fax a letter of medical necessity and include all supporting documentation for manual review to 501-683-4124. Drugs that require manual review for prior authorization Selzentry Link to Selzentry Form Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Mecamylamine HCL Tablet (Vecamyl) (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Vecamyl Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Meclorethamine HCL Gel (Valchlor) (Implemented 03/18/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drug that requires a manual review for prior authorization Valchlor 0.016% Gel Additional Criteria Quantity Limits Apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Medroxyprogesterone (Depo-Provera) (Implemented 02/12/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval Criteria No Therapeutic Duplication with any other injectable Depo-Provera DESCRIPTION MEDROXYPROGESTERONE ACETATE 104 MG/0.65 ML SYRINGE MEDROXYPROGESTERONE ACETATE 150 MG/ML SYRINGE MEDROXYPROGESTERONE ACETATE 150 MG/ML VIAL MEDROXYPROGESTERONE ACETATE 400 MG/ML VIAL Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Megestrol (Megace and Megace ES) (Implemented 10/18/2006) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria History of HIV/AIDS in the past two years, OR History of a paid claim for an antiviral: HIV agent in the past 60 days, OR History of malignancy in the past two years, OR History of a paid claim for an antineoplastic in the past 90 days Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Memantine XR (Namenda XR) (Implemented 04/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria >/= 90 days of Memantine XR therapy in the past 120 days Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Memantine Solution (Namenda) (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval Namenda: Age > 50 years of age, AND Diagnosis of NPO in the Medicaid History Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Meprobamate Tablet (Equanil) (Implemented 07/09/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Mepron (Implemented 09/21/2009) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Generic MAC’d sulfamethoxazole-trimethoprim tablets are available without a prior authorization. Drugs that require manual review for prior authorization Mepron suspension Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Mercaptopurine 20mg/ml Suspension (Purixan) (Implemented 01/13/2015) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual PA Purixan Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Mesalamine 1000mg Suppository (Canasa) (Implemented 06/21/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Methoxsalen Capsule (Oxsoralen-Ultra, 8-MOP) (Implemented 09/23/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Oxsoralen-Ultra 8-MOP Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Metreleptin 11.3mg Vial (Myalept) (Implemented 09/23/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Myalept Vial Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Metformin Oral Solution (Riomet) (Implemented 09/21/2009) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Recipient </= 6 years of age, OR Diagnosis of NPO in Medicaid history (Appendix A) Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Metformin Extended-Release (Fortamet ER, Glumetza ER) (Implemented 01/18/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Metformin 500 mg Metformin 850 mg Metformin 1000 mg Metformin ER 500 mg Metformin ER 750 mg Drugs that require manual review for prior authorization Fortamet ER Glumetza ER Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Methotrexate Auto Inj. (Otrexup) (Implemented 07/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual prior authorization Otrexup Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Methotrexate Sodium (Trexall) (Implemented 8/17/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Methotrexate 2.5mg tablet Drugs that require manual review for prior authorization Trexall 5mg Trexall 7.5mg Trexall 10mg Trexall 15mg Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Methscopolamine (Pamine, Pamine Forte, Pamine FQ) (Implemented 06/19/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria History of peptic ulcer disease in Medicaid medical history in previous 6 months, AND CPT code for H.Pylori in procedure history in the past 6 months, AND At least 112 days of PPI therapy in the last 120 days. Denial criteria History of glaucoma Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Methylnaltrexone Br Sub-Q Injection (Relistor) (Implemented 09/24/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Medical diagnosis of malignant cancer in the past three years, AND At least one paid Medicaid drug claim in the past 30 days for glycolax or lactulose, AND At least one paid Medicaid drug claim for an opioid in the past 30 days. Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Metoclopramide Orally Disentegrating Tablet (Metozolv ODT) (Implemented 01/12/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Generic MAC’d metoclopropramide tablets and syrup are available without a prior authorization. Drugs that require manual review for prior authorization Metozolv ODT tablet Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Metronidazole 375 mg capsule (Flagyl) (Implemented 08/17/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria o Diagnosis of NPO (Appendix A) in the previous 3 years, OR o </= 6 years of age Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Metronidazole ER 750mg (Flagyl) (Implemented 08/17/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Flagyl 750 mg Tablets Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Metronidazole-Tetracycline-Bismuth (Helidac and Pylera) (Implemented 01/12/2005) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Criterion 1: No history of lansoprazole, amoxicillin, and clarithromycin combination (Prevpac) in the last 365 days, AND No history of omeprazole, amoxicillin, and clarithromycin combination (Omeclamox-Pak) in the last 365 days, AND Criterion 2: No history of metronidazole,tetracycline, and bismuth combination (Helidac) in the last 365 days, OR No history of metronidazole, tetracycline, and bismuth combination (Pylera) in the last 365 days. Denial criteria Criterion 1: History of lansoprazole, amoxicillin, and clarithromycin combination (Prevpac) in the last 365 days, OR History of omeprazole, amoxicillin, and clarithromycin combination I (Omeclamox-Pak) in the last 365 days, Criterion 2: History of metronidazole, tetracyline, and bismuth combination (Helidac) in the last 365 days, OR History of metronidazole, tetracyline, and bismuth combination (Pylera) in the last 365 days Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Miconazole 50mg Buccal Tablet (Oravig) (Implemented 01/18/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Clotrimazole 10mg troches Nystatin 100,000 units/ml oral suspension Drugs that require manual review for prior authorization Oravig Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Miconazole Nitrate, Zinc Oxide, and White Petrolatum Ointment (Vusion) (Implemented 03/26/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria At least two paid claims for two different products from the following list in the past 30 days: clotrimazole 1% cream, nystatin cream or ointment nystatin-trimacinolone cream or ointment Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Mifepristone 300mg Tablet (Korlym) (Implemented 07/23/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Korlym Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Mig Lustat (Zavesca) Capsule (Implemented 01/13/2015) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Zavesca Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Mipomersen Sodium Syringe (Kynamro) (Implemented 07/09/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Kynamro Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Misoprostol (Cytotec) (Implemented 01/09/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Criterion 1: Female, AND Long Term Care, OR Current birth control drug claim (within the past 30 days), OR Current injectable birth control drug claim, OR Medical history of tubal ligation, OR Medical history of hysterectomy, OR Medical history of menopause, OR Hormone replacement therapy in the past 45 days, OR Age > 55 AND NSAID claim in past 30 days Criterion 2: Male, AND NSAID claim in the past 30 days Denial criteria Medical history of current pregnancy Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Modafinil (Provigil) (Implemented 08/17/2005) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Age > 16 years of age, AND History of at least one of the following in the last two years: o Narcolepsy, OR o Multiple sclerosis, OR o Shift work sleep disorder, OR o Obstructive sleep apnea/hypopnea, AND CPAP, OR BPAP, OR Nasal interface positive airway pressure device Denial criteria Age < 16 years of age, OR History of at least one of the following in the last 30 days: o Atomoxetine (Strattera), OR o CII Stimulants, OR o Other modafinil (Provigil) strengths, OR o Other armodafinil (Nuvigil) strengths, OR o Pemoline (Cylert) Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Mycophenolate (Cellcept, Myfortic) (Suspension Implemented 10/11/2011) (Capsules and Tablets Implemented 12/10/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria for Capsules and Tablets Diagnosis of Organ Transplant in Medicaid History in the past 3 years Approval criteria for Suspension Diagnosis of Organ Transplant in Medicaid History in the past 3 years < 6 years of age, OR NPO (Appendix A) Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Nabilone (Cesamet) (Implemented 06/27/2007) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Age > 18 years of age, AND Submitted ICD-9 diagnosis malignant cancer within the past 365 days, AND o Paid drug claim in history for antineoplastic agents within the past 45 days, OR o Procedure code indicating radiation treatment within the past 45 days, AND Paid drug claim in history within the past 45 days for an oral 5-HT3 (serotonin) receptor antagonist, OR Paid drug claim in history within the past 45 days for a NK1 (neurokinin-1) receptor antagonist, OR Paid drug claim in history within the past 45 days for Marinol. Denial criteria Absence of approval criteria Submitted ICD-9 diagnosis bipolar in medical history. Submitted ICD-9 diagnosis depression in medical history. Submitted ICD-9 diagnosis schizophrenia in medical history. Submitted ICD-9 diagnosis substance or alcohol abuse in medical history. Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Nafarelin Nasal Spray (Synarel) (Implemented 09/21/2009) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of central precocious puberty (CPP) in the previous three years, OR Diagnosis of endometriosis in the previous three years Denial criteria Diagnosis of infertility in the previous three years Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Naloxone Auto-Injector (Evzio) (Implemented 09/23/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Evzio 0.4mg Auto-Injector Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Nandrolone Decanoate Injection (Implemented 01/09/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis for anemia secondary to chronic renal failure in the past 90 days, AND At least three paid Medicaid claims in the past 90 days for erythropoietin, AND No therapeutic duplication with erythropoietin Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Neuropathic Pain Agents (Implemented 06/05/2008) The non-preferred antiepileptic medications will be considered non-preferred for treating fibromyalgia and neuropathic pain only. Medications listed as either preferred or non-preferred status in this category may or may not include an FDA approved indication for fibromyalgia or neuropathic pain. Use of these medications for fibromyalgia, neuralgias, and neuropathic pain has been reviewed through the evidence-based review process. Medications listed in this category as either preferred or nonpreferred status are not to be construed as endorsements for marketing of off-label use by the manufacturer or by Medicaid. Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents Amitriptyline HCl (Elavil) Carbamazepine chewable tablet (Tegretol Chewable) Carbamazepine immediate-release tablet (Tegretol) Gabapentin 100mg, 300mg, 400mg capsule (Neurontin) Nortriptyline HCl (Pamelor) Preferred agents with criteria Venlafaxine HCl (Effexor) – see Second Generation Antidepressant Nonpreferred agents: Gabapentin extended-release capsule (Gralise ER) Gabapentin enacarbil extended-release tablet (Horizant ER) Nonpreferred agents with criteria: Carbamazepine extended-release capsule (Carbatrol SA, Equetro) Carbamazepine extended-release tablet (Tegretol XR) Carbamazepine Suspension (Tegretol) Divalproex sodium (Depakote) Duloxetine HCl (Cymbalta) – see Second Generation Antidepressant Gabapentin 250mg/5ml solution (Neurontin) Gabapentin 100mg, 200mg, 400mg, 600mg, 800mg tablet (Neurontin) Lacosamide (Vimpat) Lamotrigine (Lamictal) Lidocaine patch (Lidoderm) Oxcarbazepine (Trileptal) Pregabalin (Lyrica) Topiramate (Topamax) Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Valprioc acid (Depakene, Stavzor) Venlafaxine HCl extended-release capsule (Effexor XR) – see Second Generation Antidepressant Approval criteria for pregabalin (Lyrica) One or more of the approved diagnosis (Appendix H), AND No therapeutic duplication with pregabalin, OR One therapeutic duplication (75% overlap of last fill) with different date of service and same prescriber ID between Lyrica GCNs in previous 93 days, AND No diagnosis of myalgia and myositis, unspecified (ICD-9 729.1) in the past three years Approval criteria for nonpreferred anti-epileptic agents Nonpreferred Antiepileptic Agents: Antiepileptic Carbamazepine extended-release capsule (Carbatrol ER, Equetro) Carbamazepine extended-release tablet (Tegretol XR) Carbamazepine suspension (Tegretol) Divalproex sodium (Depakote) Gabapentin 250mg/5ml solution (Neurontin) Gabapentin 600mg, and 800mg tablet (Neurontin) Lamotrigine (Lamictal) Oxcarbazepine (Trileptal) Pregabalin (Lyrica) Topiramate (Topamax) Valprioc acid (Depakene, Stavzor) One or more of the approved diagnoses (Appendix H) No diagnosis of myalgia and myositis, unspecified (ICD-9 729.1) in the past three years – see Fibromyalgia agent criteria Nonpreferred lamotrigine ODT (Lamictal ODT) and topiramate sprinkle capsules (Topamax Sprinkles) One or more of the approved diagnoses (Appendix H), AND Age ≤ 6, OR NPO (Appendix A) Nonpreferred extended-release products (Carbatrol ER, Depakote ER, Equetro,Gralise ER, Horizant ER, Lamictal XR, Oxtellar XR, Stavzor XR, Tegretol XR) Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria >/= 90 days of therapy of the same extended-release product in the past 120 days. Immediate-release products are covered via existing criteria Approval criteria for nonpreferred topical analgesia Submitted ICD-9 diagnosis post-herpetic neuralgia (Table 4) within the past 12 months, OR Paid claim in history identifying appropriate antiviral medication (Table 4.2) for post-herpetic neuralgia (Table 4) within the past 30 days Table 4 – Postherpetic neuralgia diagnoses ICD-9 53.12 53.13 Description Postherpetic trigeminal neuralgia Postherpetic polyneuropathy Table 4.2 – Anitvirals Antivirals Acyclovir 200mg Acyclovir 200mg Acyclovir 400mg Acyclovir 800mg Famciclovir 125mg Famciclovir 250mg Famciclovir 500mg Valacyclovir 1g caplet Valacyclovir 500mg caplet Link to Memorandum Link to Memorandum Lamictal ODT Link to Memorandum Lidoderm Patch Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Nevirapine XR (Viramune XR) (Implemented 04/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria >/= 90 days of Nevirapine XR therapy in the past 120 days Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Nevirapine Oral Supension (Viramune) (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval Razadyne: Age </= 6 Years of Age Diagnosis of NPO in the Medicaid History Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Nimodipine Solution (Nymalize) (Implemented 1210/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Nymalize Solution Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Nitisinone Capsule (Orfadin) (Implemented 09/23/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Orfadin Capsule Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Nitrofurantoin Suspension (Furadantin) (Implemented 10/11/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria < 6 years of age, OR NPO (Appendix A) Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Nitroglycerin 0.4% Rectal Ointment (Rectiv) (Implemented 07/09/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Rective Rectal Ointment Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Non-benzodiazepine Sedative Hypnotics (Implemented 03/01/2009) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents with criteria Zaleplon (Sonata) Zolpidem immediate-release (Ambien) Nonpreferred agents Eszopiclone (Lunesta) Doxepine (Silenor) Ramelton (Rozerem) Suvorexant (Belsomra) Zolpidem extended-release (Ambien CR) Zolpidem sublingual tablet (Edluar, Intermezzo) Approval criteria for preferred agents with criteria No therapeutic duplication with other sedative hypnotics Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Nizatadine Oral Solution (Axid) (Implemented 12/10/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria At least two paid Medicaid drug claims for ranitidine syrup in the past 60 days, AND < 6 years of age, OR NPO (Appendix A) Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Noxafil DR Oral Tablet and Noxafil 300mg Vial (Implemented 07/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Noxafil DR Oral Tablet Noxafil 300mg Vial Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Omega-3-acid ethyl esters (Lovaza) (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Lovaza Additional Criteria Quantity Limits Apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Omeprazole, Amoxicillin, and Clarithromycin combination (Omeclamox-Pak) (Implemented 05/21/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Criterion 1: No history of lansoprazole, amoxicillin, and clarithromycin combination (Prevpac) in the last 365 days, AND, No history of omeprazole, amoxicillin, and clarithromycin combination (Omeclamox-Pak) in the last 365 days, AND, No concurrent proton pump inhibitor therapy within the past 30 days. Criterion 2: No history of lansoprazole, amoxicillin, and clarithromycin combination (Prevpac) in the last 365 days, AND, No history of omeprazole, amoxicillin, and clarithromycin combination (Omeclamox-Pak) in the last 365 days, AND, No concurrent proton pump inhibitor therapy within the past 30 days. Criterion 3: No history of metronidazole/tetracycline/bismuth combination (Helidac) in the last 365 days, OR No history of metronidazole/tetracycline/bismuth combination (Pylera) in the last 365 days. Denial criteria Criterion 1: History of lansoprazole, amoxicillin, and clarithromycin combination (Prevpac) in the last 365 days, OR Current proton pump inhibitor therapy within the past 30 days Criterion 2: No history of omeprazole, amoxicillin, and clarithromycin combination (Omeclamox-Pak) in the last 365 days, AND, No concurrent proton pump inhibitor therapy within the past 30 days. Criterion 3: No history of metronidazole/tetracycline/bismuth combination (Helidac) in the last 365 days, OR No history of metronidazole/tetracycline/bismuth combination (Pylera) in the last 365 days. Additional criteria Quantity limits apply Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Opioids, Long-acting (Implemented 08/01/2008) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents with criteria Methadone HCl (Dolophine) Morphine sulfate long-acting tablet (MS Contin, Oramorph) Nonpreferred agents with criteria Buprenorphine patch (Butrans) Fentanyl patch (Duragesic) Hydromorphone HCl extended-release tablet (Exalgo ER) Morphine sulfate extended-release capsule (Avinza, Kadian) Morphine sulfate/naltrexone (Embeda) Oxycodone-Acetaminophen extended-release tablet (Xartemis XR) Oxycodone extended-release tablet (Oxycontin) Oxymorphone HCl extended-release tablet (Opana ER) Tapentadol HCl extended-release tablet (Nucynta ER) Approval criteria for preferred agents with criteria No therapeutic duplication in drug history between long-acting narcotics Approval criteria for nonpreferred agents with criteria Buprenorphine patch o NPO (Appendix A), OR o Currently LTC, OR o Cancer with malignancies (Appendix E) or antineoplastic drug history (Appendix F) in past 12 months AND o No therapeutic duplication in drug history between long-acting narcotics Fentanyl patch o NPO (Appendix A), OR o Currently LTC, OR o Cancer with malignancies (Appendix E) or antineoplastic drug history (Appendix F) in past 12 months AND o No therapeutic duplication in drug history between long-acting narcotics Morphine sulfate long-acting capsule or oxycodone long acting tablet o Currently LTC, OR Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Cancer with malignancies (Appendix E) or antineoplastic drug history (Appendix F) in past 12 months AND o No therapeutic duplication in drug history between long-acting narcotics o Denial criteria Paid claim for Suboxone or Subutex in the past 60 days Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Opioids, Short-acting (Implemented 11/12/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Accumulation quantity limit will allow up to a maximum of 124 units of any solid oral short acting opioid paid by Medicaid per the previous 31 calendar days. No drug claim in the past 60 days for Subutex, OR No drug claim in the past 60 days for Suboxone AND Therapeutic duplication between short-acting opioids with less than 25% of the days’ supply remaining on the previous claim, OR Therapeutic duplication between a short-acting opioid and tramadol IR (Ultram) with less than 25% of the days’ supply remaining on the previous claim, OR Therapeutic duplication between a short-acting opioid and tramadol/acetaminophen (Ultracet) with less than 25% of the days’ supply remaining on the previous claim Therapeutic duplication between a short-acting opioid and tramadol ODT (Rybix) with less than 25% of the days’ supply remaining on the previous claim Denial criteria Therapeutic duplication between two short-acting opioids with more than 25% of the days’ supply remaining on previous claim Therapeutic duplication between a short-acting opioid and tramadol (Utram and Ultracet) with more than 25% of the days’ supply remaining on previous claim Therapeutic duplication between a short-acting opioid and tramadol ODT (Rybix) with more than 25% of the days’ supply remaining on previous claim Drug claim in history for Subutex Drug claim in history for Suboxone Solid oral dosage forms for short-acting opioids will reject for children less than 6 years of age. Greater than 124 units of any solid oral short acting opioid paid by Medicaid per the previous 31 calendar days. Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Exemptions Patients who have a diagnosis of malignant cancer or a paid claim for an antineoplastic in the past 12 months are exempt from the therapeutic duplication requirement. Quantity restrictions still apply. Short-acting pain medications that process with point of sale prior authorization criteria POINT OF SALE APPROVED SHORT-ACTING PAIN MEDICATIONS ACETAMINOPHEN-CODEINE 300-15 MG TABLET COMMON or OLDER TRADE NAME ACETAMINOPHEN-CODEINE 120-12 MG/5 ML SOLUTION TYLENOL WITH CODEINE #2 ACETAMINOPHEN-CODEINE 300-30 MG TABLET TYLENOL WITH CODEINE #3 ACETAMINOPHEN-CODEINE 300-60 MG TABLET BUTALBITAL-ACETAMINOPHEN-CAFFEINE 50-325 MG/15 ML SOLUTION BUTALBITAL-ACETAMINOPHEN 50-325 MG TABLET BUTALBITAL-ACETAMINOPHEN-CAFFEINE 50-325-40 MG TABLET CODEINE SULFATE 15 MG TABLET TYLENOL WITH CODEINE #4 ACETAMINOPHEN-CODEINE 120-12 MG/5 ML SOLUTION ALAGESIC LQ SOLUTION PHRENILIN TABLET ESGIC TABLET CODEINE SULFATE 30 MG TABLET CODEINE SULFATE 15 MG TABLET CODEINE SULFATE 30 MG/5 ML ORAL SOLUTION CODEINE SULFATE 30 MG TABLET CODEINE SULFATE 60 MG TABLET CODEINE SULFATE 60 MG TABLET HYDROCODONE-ACETAMINOPHEN 5-325 MG TABLET NORCO 5-325 TABLET HYDROCODONE-ACETAMINOPHEN 7.5-325 MG TABLET NORCO 7.5-325 TABLET HYDROCODONE-ACETAMINOPHEN 7.5-325 MG/15 ML SOLUTION HYCET 7.5-325 MG/5 ML SOLUTION HYDROCODONE-ACETAMINOPHEN 10-300 MG/15 ML SOLUTION LORTAB 10-300 MG/15 ML SOLUTION HYDROCODONE-ACETAMINOPHEN 10-325 MG TABLET NORCO 10-325 TABLET HYDROCODONE-IBUPROFEN 5-200 MG TABLET IBUDONE 5-200 MG TABLET HYDROCODONE-IBUPROFEN 5-200 MG TABLET REPREXAIN 5-200 MG TABLET HYDROCODONE-IBUPROFEN 7.5-200 MG TABLET VICOPROFEN 7.5-200 TABLET HYDROCODONE-IBUPROFEN 10-200 MG TABLET IBUDONE 10-200 MG TABLET HYDROCODONE-IBUPROFEN 10-200 MG TABLET REPREXAIN 10-200 MG TABLET HYDROMORPHONE 1 MG/ML LIQUID DILAUDID LIQUID HYDROMORPHONE 2 MG TABLET DILAUDID 2 MG TABLET HYDROMORPHONE 4 MG TABLET DILAUDID 4 MG TABLET HYDROMORPHONE 8 MG TABLET DILAUDID 8 MG TABLET CODEINE SULFATE 30 MG/5 ML SOLUTION LEVORPHANOL TARTRATE 2 MG TABLET LEVO-DROMORAN 2 MG TAB MEPERIDINE 50 MG TABLET DEMEROL 50 MG TABLET MEPERIDINE 100 MG TABLET DEMEROL 100 MG TABLET MORPHINE SULFATE 10 MG/5 ML SOLUTION MSIR 10 MG/5 ML ORAL SOLUTION MORPHINE SULFATE 15 MG TABLET MSIR 15 MG TABLET MORPHINE SULFATE 20 MG/ML CONCENTRATED SOLUTION ROXANOL 20 MG/ML CONCENTRATE MORPHINE SULFATE 20 MG/5 ML SOLUTION MSIR 20 MG/5 ML ORAL SOLUTION MORPHINE SULFATE 30 MG TABLET MSIR 30 MG TABLET OXYCODONE 5 MG CAPSULE OXYIR 5 MG CAPSULE OXYCODONE 5 MG/5 ML SOLUTION OXYCODONE 5 MG/5 ML SOLUTION Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria POINT OF SALE APPROVED SHORT-ACTING PAIN MEDICATIONS OXYCODONE 5 MG TABLET COMMON or OLDER TRADE NAME ROXICODONE 5 MG TABLET OXYCODONE 10 MG TABLET OXYCODONE 10 MG TABLET OXYCODONE 15 MG TABLET ROXICODONE 15 MG TABLET OXYCODONE 20 MG TABLET OXYCODONE 20 MG TABLET OXYCODONE 30 MG TABLET OXYCODONE 30 MG TABLET OXYCODONE-ACETAMINOPHEN 5-325 MG/5 ML SOLUTION ROXICET 5-325 MG/5 ML SOLUTION OXYCODONE -ACETAMINOPHEN 5-325 MG TABLET PERCOCET 5-325 TABLET OXYCODONE-ACETAMINOPHEN 7.5-325 MG TABLET PERCOCET 7.5-325 TABLET OXYCODONE-ACETAMINOPHEN 10-325 MG TABLET PERCOCET 10-325 TABLET TRAMADOL 50 MG TABLET ULTRAM TABLET TRAMADOL-ACETAMINOPHEN 37.5-325 MG TABLET ULTRACET TABLET Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Short-Acting pain medications that require manual review for prior authorization MANUALLY REVIEWED SHORT-ACTING PAIN MEDICATIONS ACETAMINOPHEN-CODEINE 120-12 MG/5 ML SUSPENSION COMMON TRADE NAME CAPITAL WITH CODEINE SUSPENSION BUTALBITAL-ACETAMINOPHEN 50-300 MG TABLET BUPAP 50-300 MG TABLET BUTALBITAL-ACETAMINOPHEN-CAFFEINE 50-300-40 MG CAPSULE FIORICET CAPSULE BUTALBITAL-ACETAMINOPHEN-CAFFEINE 50-325-40 MG CAPSULE ESGIC CAPSULE BUTALBITAL-ASPIRIN-CAFFEINE 50-325-40 MG CAPSULE FIORINAL CAPSULE BUTORPHANOL 10 MG/ML NASAL SPRAY CODEINE-BUTALBITAL-ACETAMINOPHEN-CAFFEINE 30-50-325-40 MG CAPSULE CODEINE-BUTALBITAL-ASPIRIN-CAFFEINE 30-50-325-40 MG CAPSULE DIHYDROCODEINE-ASPIRIN-CAFFEINE 16-356-30 MG CAPSULE STADOL NASAL SPRAY HYDROCODONE-ACETAMINOPHEN 2.5-325 MG TABLET VERDROCET 2.5-325 MG TABLET HYDROCODONE-ACETAMINOPHEN 5-300 MG TABLET VICODIN 5-300 MG TABLET HYDROCODONE-ACETAMINOPHEN 7.5-300 MG TABLET VICODIN ES 7.5-300 MG TABLET HYDROCODONE-ACETAMINOPHEN 10-300 MG TABLET VICODIN HP 10-300 MG TABLET HYDROCODONE-ACETAMINOPHEN 10-325 MG/15 ML SOLUTION ZAMICET 10-325 MG/15 ML SOLUTION HYDROCODONE-IBUPROFEN 2.5-200 MG TABLET REPREXAIN 2.5-200 MG TABLET OPIUM TINCTURE 10 MG/ML LIQUID OXYCODONE-ACETAMINOPHEN 2.5-325 MG TABLET OPIUM TINCTURE 10 MG/ML ORAL LIQUID OXYCODONE 20 MG/ML CONCENTRATED ORAL SOLN PERCOCET 2.5-325 MG TABLET OXYCODONE-ACETAMINOPHEN 5-300 MG TABLET PRIMLEV 5-300 MG TABLET OXYCODONE-ACETAMINOPHEN 5-400 MG TABLET MAGNACET 5-400 MG TABLET OXYCODONE-ACETAMINOPHEN 7.5-300 MG TABLET PRIMLEV 7.5-300 MG TABLET OXYMORPHONE 5 MG TABLET OPANA 5 MG TABLET OXYMORPHONE 10 MG TABLET OPANA 10 MG TABLET PENTAZOCINE-NALOXONE 50-0.5 MG TABLET TALWIN NX TABLET TAPENTADOL 50 MG TABLET NUCYNTA 50 MG TABLET TAPENTADOL 75 MG TABLET NUCYNTA 75 MG TABLET TAPENTADOL 100 MG TABLET NUCYNTA 100 MG TABLET OXYCODONE 20 MG/ML CONCENTRATED SOLUTION Additional criteria Quantity limits apply Link to Memorandum Link to Memorandum FIORICET WITH CODEINE CAPSULE FIORINAL WITH CODEINE CAPSULE SYNALGOS-DC CAPSULE Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Link to Memorandum Link to updated criteria Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Opium Tincture (Implemented 10/11/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Opium Tincture Additional Criteria Quantity Limits Apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Oseltamivir Suspension (Tamiflu) (Implemented 10/11/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria ≤ 12 years of age, AND At least 1 year of age Additional criteria Quantity Limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Osteoporosis drugs (Implemented 08/17/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Alendronate sodium 5mg daily dose (Fosamax®) Alendronate sodium 10mg daily dose (Fosamax®) Alendronate sodium 35mg weekly dose (Fosamax®) Alendronate sodium 40mg weekly dose (Fosamax®) Alendronate sodium 70mg weekly dose (Fosamax®) Drugs that require manual review for prior authorization Actonel® tablet Binosto® effervescent tablet Boniva ® tablet Boniva® injection Fosamax® Plus D tablet Fosamax® oral solution Forteo® injection (new start only) Prolia® injection (see below): Prolia Prolia® will continue to be covered through a manual review PA on a case-bycase basis for the initial dose. POS PA continuation approval criteria for Prolia® will apply as follows: 1 Prolia® claim is found in Medicaid drug history in the previous 12 months. In addition, a therapeutic duplication edit will reject an incoming Prolia® claim if an Xgeva® (denosumab) claim is found in the medical claims history in previous 6 months. A quantity edit for Prolia® of 1 injection per 175 days will be implemented. Additional criteria Forteo® quantity limits apply Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Link to Memorandum Link to Memorandum (Prolia) Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Otic Preparations (Implemented 09/21/2009, 01/18/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). OTIC PREPARATIONS GENERIC NAME COMMON TRADE NAME* PA STATUS Acetic Acid 2% otic drops Acetic Acid 2% otic solution No PA Antipyrine-Benzocaine-Glycerin otic solution Aurodex® otic solution No PA Benzocaine 20% otic drops Pinnacaine® 20% otic drops No PA Ciprofloxacin 0.3% ophthalmic drops Ciloxan® 0.3% ophthalmic drops No PA Fluocinolone acetonide Oil otic drops Dermotic® Oil 0.01% otic drops No PA Neomycin sulfate-Polymyxin B-Hydrocortisone otic solution or suspension Cortisporin® Ear otic solution or suspension No PA Ofloxacin 0.3% otic drops Floxin® 0.3% otic drops No PA Sulfacetamide-Prednisolone 10-0.23% ophthalmic drops Sulf-Pred® 10-0.23% ophthalmic drops No PA GENERIC NAME COMMON TRADE NAME* PA STATUS Acetic Acid-Hydrocortisone otic drops Acetasol HC® otic drops Manual PA Acetic Acid-Benzocaine 5.5%-1.4% otic drops Aurax® otic solution Manual PA Antipyrine-Benzocaine-Zinc Acetate otic drops Otozin® otic solution Manual PA Ciprofloxacin-Dexamethasone otic suspension Ciprodex® otic suspension Manual PA Ciprofloxacin-Hydrocortisone otic suspension Cipro HC® otic suspension Manual PA Neomycin sulfate-Polymyxin B-HydrocortisoneThonzonium otic drops Coly-Mycin S® otic drops Manual PA Neomycin sulfate-Polymyxin B-HydrocortisoneThonzonium otic drops Cortisporin TC® otic suspension Manual PA Link to Memorandum Otic Preparations Implemented 01/18/2011 Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Overactive-bladder Agents (Implemented 07/14/2009) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents Fesoterodine fumarate (Toviaz) Oxybutynin chloride 5mg/5ml Syrup, 5mg tablet (Ditropan) Solfenacin succinate (Vesicare) Preferred agents with criteria Oxybutynin chloride extended-release tablet (Ditropan XL Tablet) Nonpreferred agents Darifenacin hydrobromide (Enablex) Flavoxate HCl (Urispas) Mirabegron extended-release (Myrbetriq) Oxybutynin chloride gel (Gelnique) Oxybutynin patch (Oxytrol) Tolterodine tartrate tablet (Detrol) Tolterodine tartrate extended-release capsule (Detrol LA) Trospium chloride extended-release (Sanctura XR) Trospium chloride immediate-release (Sanctura) Approval criteria for preferred agents with criteria Oxybutynin XL tablet Ages 6 – 18, AND Diagnosis of spina bifida (ICD-9: 741), OR Four paid claims of oxybutynin XL in the past 6 months and age between 6 – 18 Link to PDL Memorandum: Overactive-bladder agents Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Oxandrolone (Oxandrin) (Implemented 01/09/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Criterion 1: Diagnosis in the past two years for: HIV/AIDS, AND Cachexia AND Paid drug claims in history for at least three antiretrovirals (either as a single entity or combo drug) within the past 30 days, AND Paid drug claim in history for megestrol acetate within the last four weeks Criterion 2: Diagnosis code in Medicaid history in the past 365 days for one of the following: Weight loss secondary to severe trauma (burns, spinal cord injury), OR Weight loss due to protein catabolism associated with prolonged administration of high dose corticosteroids. Criterion 3: Diagnosis in Medicaid history for osteoporosis, AND Concurrent therapy in the past 45 days for one of the following: o Biphophonate Didronel (etidronate) Fosamax (alendronate) o Aminobisphosphonate Actonel (risedronate) o Selective estrogen-receptor modulators Evista (raloxifene) Calcitonin injection, OR History of Ibandronate in the past 100 days Criterion 4: Diagnosis in Medicaid history for hereditary angioedema in the past three years. Denial criteria Diagnosis in Medicaid history for any of the following in the past 365 days: Prostate cancer Breast cancer – male or female Pregnancy Nephrosis Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Oxymethalone (Anadrol—50 tablet) (Implemented 01/09/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Criterion 1: Diagnosis of acquired aplastic anemia in the past 365 days, AND Five claims in the past six months for Cyclosporine Criterion 2: Diagnosis of chronic renal failure in the past 365 days, AND History of three paid drug claims of recombinant erythropoietin in the past 90 days Criterion 3: Diagnosis of malignant cancer in the past 365 days, AND Paid drug claim for an antineoplastic in the past 365 days, AND History of three paid drug claims for Neupogen in the past 90 days, OR History of three paid drug claims for Neulasta in the past 90 days Criterion 4: Diagnosis of Fanconi’s anemia in the past 365 days Criterion 5: Diagnosis for congenital refractory pure red cell aplasia in the past 365 days, AND At least 30 days of corticosteroid therapy in the past 90 days Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Pain Medications, Injectable (Implemented 04/12/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Injectable agents Buprenorphine Injectable Butorphanol Injectable Hydromorphone Injectable Levorphanol Injectable Meperidine Injectable Morphine Injectable Nalbuphine Injectable Pentazocine Injectable Approval criteria No drug claim in the past 60 days for Subutex, OR No drug claim in the past 60 days for Suboxone AND Therapeutic duplication between short-acting opioids with less than 25% of the days’ supply remaining on the previous claim, OR Therapeutic duplication between a short-acting opioid and tramadol IR (Ultram) with less than 25% of the days’ supply remaining on the previous claim, OR Therapeutic duplication between a short-acting opioid and tramadol/acetaminophen (Ultracet) with less than 25% of the days’ supply remaining on the previous claim Therapeutic duplication between a short-acting opioid and tramadol ODT (Rybix) with less than 25% of the days’ supply remaining on the previous claim Denial criteria Therapeutic duplication between two short-acting opioids with more than 25% of the days’ supply remaining on previous claim Therapeutic duplication between a short-acting opioid and tramadol (Utram and Ultracet) with more than 25% of the days’ supply remaining on previous claim Therapeutic duplication between a short-acting opioid and tramadol ODT (Rybix) with more than 25% of the days’ supply remaining on previous claim Drug claim in history for Subutex Drug claim in history for Suboxone Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Exemptions Patients who have a diagnosis of malignant cancer or a paid claim for an antineoplastic in the past 12 months are exempt. Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Papaverine 30mg/ml (Implemented 08/10/2007) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Denial criteria Submitted ICD-9 diagnosis for erectile dysfunction Submitted ICD-9 diagnosis for Impotence Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Pasireotide Diaspartate (Signifor) Ampule (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Signifor Additional Criteria Quantity Limits Apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Pazopanib (Votrient) (Implemented 10/10/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Votrient Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria PegInterferon Alfa-2B (Sylatron) (Implemented 05/25/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Denial criteria Submitted ICD-9 diagnosis for Hepatitis C, Chronic and Acute Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Pegvisomant Injection (Somavert) (Implemented 04/17/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria No Therapeutic duplication allowed between different strengths of Somavert Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Penicillamine (Depen 250mg; Cuprimine 250mg) (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Depen 250mg Tablet Cuprimine 250mg Capsule Additional Criteria Quantity Limits Apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Perampanel Tablet (Fycompa) (Implemented 07/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Fycompa Additional Criteria Quantity Limits Apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Phosphate Removing Agents (Implemented 07/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Calcium acetate 667 mg Capsule [PhosLo] Calcium acetate 667 mg/5 ml Solution [Phoslyra] Calcium acetate 667 mg Tablet [Eliphos] Sevelamer HCl Tablet [Renagel] Sevelamer carbonate 800 mg Tablet [Renvela 800 mg Tablet] Drugs that require manual review for prior authorization Ferric Citrate Tablet Lanthanum carbonate Chewable Tablet [Fosrenol] Sevelamer carbonate Powder Packet [Renvela Powder Packet] Sucroferric oxyhydroxide Chewable Tablets [Velphoro] Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Pimecrolimus (Elidel) (Implemented 03/12/2007) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria No therapeutic duplication with Tacrolimus (Protopic), AND At least two paid Medicaid drug claims of topical corticosteroid agents, each containing a different drug entity, AND At least one of the claims for the topical corticosteroid being at least medium potency or higher filled in the previous 14-45 day period. Additional criteria Age > 2 years of age Quantity limits apply Denial criteria History of Psoriasis in previous two years Therapeutic duplication with Tacrolimus (Protopic) No claim for at least two topical corticosteroids in Medicaid history No claim for a medium potency or higher topical corticosteroid in the previous 14-45 day period. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Podofilox (Condylox 0.5% topical solution and gel) (Implemented 01/09/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Podofilox 0.5% Topical Solution (Condylox) ≥ 18 years of age, AND No therapeutic duplication with Podofilox 0.5% gel, AND Diagnosis of Condyloma Acuminanta (commonly known as external genital or perianal warts) within the past two months Podofilox 0.5% Topical Gel (Condylox) ≥ 18 years of age, AND No therapeutic duplication with Podofilox 0.5% solution, AND Diagnosis of of Condyloma Acuminanta (commonly known as external genital or perianal warts) within the past two months OR Denial criteria Absence of approval criteria < 18 years of age Therapeutic duplication of gel/solution Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Pomalidomide Capsule (Pomalyst) (Implemented 07/09/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Pomalyst Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Ponatinib HCl Tablet (Iclusig) (Implemented 07/09/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Iclusig Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Posaconazole (Noxafil) Suspension (Implemented 01/09/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Criterion 1: Diagnosis code in Medicaid history for at least one of the following in the past 365 days: ≥ 13 years of age, AND HIV/AIDS Organ transplant procedure Graft vs. host disease Neutropenia Criterion 2: The following drug claims in Medicaid history in the past 365 days: ≥ 13 years of age, AND HIV/AIDS pharmacotherapy drug claims in history, OR Anti-rejection/Immunosuppression medication Criterion 3: ≥ 13 years of age, AND At least one paid claim for Fluconazole in the past 30 days, AND At least one paid claim for Itraconazole in the past 30days. Criterion 4: ≥ 13 years of age, AND History of paid claim for requested drug (Noxafil) in the past 180 days. Denial criteria < 13 years of age Absence of approval criteria History of a paid drug claim for the any of the following in the last 30 days: o Ergot alkaloids o Pimozide o Quinidine Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Posaconazole (Noxafil DR 100mg Oral Tablet and Noxafil 300mg Vial) (Implemented 07/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Noxafil DR 100mg Tablet Noxafil 300mg Vial Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Potassium Chloride (KCl) Capsule and Packet (Implemented 10/10/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Potassium chloride 8 mEq extended release-tablet [Klor-Con 8 mEq tablet] Potassium chloride 10 mEq extended-release tablet [Klor-Con M10] Potassium chloride 10% (20 mEq/15 ml) oral solution Potassium chloride 20 mEq extended-release tablet [Klor-Con M20] Potassium chloride 25 mEq effervescent tablet [Effer-K, Klor-Con-EF] Drugs that require manual review for prior authorization Potassium chloride 8 mEq extended-release capsule [Micro-K] Potassium chloride 10 mEq extended-release capsule [Micro-K] Potassium chloride 20 mEq powder packet [Klor-Con 20 mEq packet]* Potassium chloride 25 mEq powder packet [Klor-Con 25 mEq packet) *Effective 4/14/2014 Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Pramipexole ER (Mirapex ER) (Implemented 03/19/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of Parkinson’s Disease in Medicaid history in previous 2 years, AND >/= 90 days of therapy of Pramipexole ER in the past 120 days Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Prednisolone Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Generic prednisolone sodium phosphate 15mg/5ml (same as Orapred ® Solution) is available without a prior authorization Methylprednisolone Dose Pack Methylprednisolone tablet Prednisone Dose Pack Prednisone tablet Drugs that require manual review for prior authorization Flo-Pred 16.7 (15) mg/5 ml suspension – Implemented 07/08/2011 Millipred 5 mg Dose Pack – Implemented 01/18/2011 Millipred 5 mg tablet – Implemented 01/18/2011 Millipred 10 mg/5 ml solution – Implemented 04/21/2009 Orapred ODT tablet – Implemented 08/17/2010 Veripred 20 mg/5 ml solution – Implemented 04/21/2009 Link to Memorandum: Millipred Tablets, Dose Pack Link to Memorandum: Millipred Solution Link to Memorandum: Orapred ODT Link to Memorandum: Veripred Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Prednisone (Rayos DR) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Rayos DR 1mg Rayos DR 2mg Rayos DR 5mg Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Propafenone SR (Rythmol SR) (Implemented 04/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria >/= 90 days of Propafenone SR therapy in the past 120 days Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Proton Pump Inhibitors (Implemented 08/17/2010) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents with criteria Omeprazole 20mg capsule (Rx Prilosec) Pantoprazole sodium (Protonix) Nonpreferred agents Dexlansoprazole (Dexilant) Esomeprazole magnesium capsule (Nexium) Esomeprazole magnesium packet (Nexium Packet) Esomeprazole magnesium/Naproxen tablet (Vimovo) Esomeprazole strontium capsule Omeprazole 10mg, 40mg capsule (Rx Prilosec) Omeprazole suspension (Prilosec Suspension) Omeprazole/sodium bicarbonate (Zegerid) Rabeprazole sodium (Aciphex) Nonpreferred agents with criteria Lansoprazole capsule (Prevacid capsule) Lansoprazole solutab (Prevacid Solutab) Approval criteria for preferred agents with criteria Approve up to 93 days of proton pump inhibitor therapy per year for all recipients age 15 months or older Approve treatment beyond 93 days for recipients 15 months or older who have a diagnosis in history for Zollinger-Ellison Syndrome, Barrett’s esophagus, or an endoscopy (Appendix I) in the past 24 months Approve treatment beyond 93 days for recipients 15 months or older who have a diagnosis in history for Cystic Fibrosis, pancreatic insufficiency, or pancreatic disease in the past 24 months Approval criteria for nonpreferred agents with criteria Prevacid capsules < 1 year of age Prevacid solutabs < 7 years of age Seven years of age and older with documented history of NPO (Appendix A) within past 365 days. Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Denial criteria Prevacid capsules > 1 year of age Prevacid solutabs ≥ 7 years of age No documented history of NPO (Appendix A) within past 365 days All Proton Pump Inhibitors > 93 days of PPI therapy in the past 365 days for recipients 15 months or older, unless there is a diagnosis in history for ZollingerEllison Syndrome, Barrett’s esophagus, Cystic Fibrosis, pancreatic insufficiency, pancreatic disease, or an endoscopy (Appendix I) in the past 24 months Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Pyridostigmine Timespan (Mestinon Timespan) (Implemented 04/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria >/= 90 days of Pyridostigmine ER therapy in the past 120 days Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Calcitriol (Vectical), Calcipotriene (Dovonex, Sorilux) (Implemented 06/19/2006) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drug Dovonex Sorilux Vectical Approval criteria Diagnosis of psoriasis in Medicaid history in previous 365 days 2 paid claims of a topical corticosteroid in previous 27-60 days At least one paid claim for a topical corticosteroid must be from the very high potency category Approval criteria Diagnosis of psoriasis in Medicaid history in previous 365 days The incoming claim matches claim in history in the previous 45 days for Vectical, Dovonex, Sorilux At least two claims of a topical corticosteroid in previous 60 days in drug claim history with at least one topical corticosteroid claim 14-60 days back in history Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Quinine Sulfate (Qualaquin) (Implemented 06/27/2007) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Submitted ICD-9 diagnosis of uncomplicated plasmodium falciparum malaria in the previous 6 months, AND Concurrent therapy with seven days of Tetracycline, OR Concurrent therapy with seven days Doxycycline, OR Concurrent therapy with seven days Clindamycin. Denial criteria Absence of approval criteria Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Raloxifene (Evista) (Implemented 08/21/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of postmenopause in the previous 2 years, AND o Diagnosis of breast cancer in the previous 2 years OR Diagnosis of postmenopause in the previous 2years, AND Diagnosis of osteoporosis in the previous 2 years, AND o Diagnosis of esophageal strictures in the previous 2 years , OR o Diagnosis of esophageal achalasia in the previous 2 years Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Ranolazine (Ranexa) (Implemented 10/18/2006) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Denial criteria Diagnosis of hepatic impairment in the last 12 months Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Regorafenib (Stivarga) 40mg Tablet (Implemented 03/19/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Stivarga Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Respiratory Syncytial Virus (RSV) Medications (Implemented 01/01/1999) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Synagis PA form PA Forms available during RSV season at www.medicaid.state.ar.us Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Rifaximin 550mg Tablets (Xifaxan) (Implemented 09/28/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of Hepatic Encephalopathy in the previous 2 years. Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Rilonacept Injection (Arcalyst) (Implemented 07/09/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that requires manual review for prior authorization Arcalyst Injection Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Riociguat Tablet (Adempas) (Implemented 03/18/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Adempas 0.5 mg Tablet Adempas 1 mg Tablet Adempas 1.5 mg Tablet Adempas 2 mg Tablet Adempas 2.5 mg Tablet Additional criteria Quantity edits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Rivaroxaban 10mg Tablet (Xarelto) (Implemented 04/17/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria One claim of up to 31 tablets of 10 mg will be allowed at POS for inferred surgery prophylaxis, AND One paid claim for Xarelto 10 mg will be allowed once every 186 days for inferred surgery prophylaxis, AND There are no paid claims for others strengths of Xarelto® in the previous 6 months of Medicaid drug history, AND No therapeutic duplication with overlapping days’ supply allowed between Xarelto 10 mg claim and a claim for a Low Molecular Weight heparin Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Rivaroxaban 15mg, 20mg Tablet, Starter Pack (Xarelto) (Implemented 04/17/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria No Therapeutic duplication allowed between different strengths of Xarelto One (1) therapeutic duplication with overlapping days’ supply will be allowed once per 186 days for inferred change in therapy between an Xarelto® claim and any of the following: a Pradaxa® claim, a warfarin claim, OR a Eliquis® claim, AND The Xarelto® claim and the warfarin claim, the Pradaxa® claim, or the Eliquis® claim cannot have the same date of service. Additional criteria Quantity limits apply Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Rivastigmine Solution (Exelon) (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval Exelon: Age > 50 years of age, AND Diagnosis of NPO in the Medicaid History Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Ropinirole XL (Requip XL) (Implemented 03/19/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of Parkinson’s Disease in Medicaid history in previous 2 years AND >/= 90 days of therapy of Ropinirole XL in the past 120 days Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Rotigotine (Neupro) Patch (Implemented 03/19/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of Parkinson’s Disease in Medicaid history in previous 2 years Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Rosacea Treatment (Implemented 06/19/2006) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drug Finacea 15% Gel Metrogel 1% Topical Mirvaso 0.33% Gel Noritate 1% Cream Approval criteria Diagnosis of rosacea in Medicaid history in previous 2 years 2 paid claims for generic metronidazole 0.75% cream, gel, or lotion in the previous 27-60 days Denial criteria History of acne vulgaris in the last 60 days Drugs that do not require a PA o Metronidazole 0.75% Topical Cream [MetroCream 0.75%] o Metronidazole 0.75% Topical Gel [Metrogel 0.75%] o Metronidazole 0.75% Topical Lotion [MetroLotion 0.75%] Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Ruxolitinib Tablets (Jakafi) (Implemented 04/17/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria No Therapeutic duplication allowed between different strengths of Jakafi Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Sapropterin Dihydrochloride (Kuvan) (Implemented 04/12/2011) Prescribers are required to fax a letter of medical necessity and include all supporting documentation for manual review to 501-683-4124. Drugs that requires manual review for prior authorization Kuvan Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Sedative Hypnotics (Implemented 06/19/2006) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drug Estazolam (Prosom) Flurazepam (Dalmane) Quazepam (Doral) Temazepam (Restoril) Triazolam (Halcion) Denial criteria Therapeutic duplication with any of the following sedative hypnotic Estazolam (Prosom) Eszopiclone (Lunesta)* Flurazepam (Dalmane) Quazepam (Doral) Ramelteon (Rozerem)* Temazepam (Restoril) Triazolam (Halcion) Zaleplon (Sonata)* Zolpidem (Ambien)* Zolpidem (Zolpimist)* Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Serostim (Implemented 10/18/2006) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that requires manual review for prior authorization Serostim Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Serotonin 5-HT 1 Receptor Agonists (Implemented 07/01/2010) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents with criteria Sumatriptan 4mg/0.5ml kit refill (Imitrex) Sumatriptan 5mg nasal spray (Imitrex) Sumatriptan 6mg/0.5ml kit refill (Imitrex) Sumatriptan 6mg/0.5ml kit syringe (Imitrex) Sumatriptan 6mg/0.5ml vial (Imitrex) Sumatriptan 20mg nasal spray (Imitrex) Sumatriptan succinate tablet (Imitrex) Nonpreferred agents Almotriptan malate (Axert) Eletriptan HBr (Relpax) Frovatriptan succinate (Frova) Naratriptan HCl (Amerge) Rizatriptan benzoate (Maxalt) Sumatriptan 4mg/0.5ml needle free injection (Sumavel Dosepro) Sumatriptan 4mg/0.5ml syringe (Non MAC’d) Sumatriptan 4mg/0.5ml vial Sumatriptan 6mg/0.5ml needle free injection (Sumavel Dosepro) Sumatriptan 6mg/0.5ml syringe (Non MAC’d) Sumatriptan succinate/naproxen sodium (Treximet) Zolmitriptan (Zomig) Nonpreferred agents with criteria Rizatriptan benzoate disintegrating (Maxalt MLT) Approval criteria for preferred agents with criteria Preferred Injection Any serotonin 5-HT 1 receptor antagonist within past 365 days Approval criteria for nonpreferred agents with criteria: Maxalt MLT tablet Any preferred serotonin 5-HT 1 receptor antagonist, Maxalt tablet, or Maxalt MLT tablet within past 365 days Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Denial criteria for all agents: Therapeutic duplication of any serotonin 5-HT 1 receptor agonist Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Sildenafil (Revatio) (Implemented 10/11/2005) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of pulmonary heart disease in the last 365 days, OR Diagnosis of persistent fetal circulation in the last 365 days, OR Diagnosis of chronic respiratory disease arising in the perinatal period in the last 365 days Denial criteria One of the following diagnoses in the last 180 days: o Cavernosal fibros, OR o Hypotension, OR o Leukemia, OR o Life-threatening arrhythmia, OR o Malignant hypertension, OR o Multiple myeloma, OR o Myocardial infarction, OR o Peyronie’s disease, OR o Retinitis pigmentosa, OR o Sickle cell disease, OR o Stroke, OR o Unstable angina History of any of the following in the last 45 days: o Alpha-adrenergic blockers o Nitrates o Tamsulosin Concurrent use of any the following: o Indinavir o Lopinavir-ritonavir o Ritonavir Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Siltuximab vial (Sylvant) (Implemented 09/23/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Sylvant Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Sinecatechins (Veregen ointment 15%) (Implemented 06/19/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis for of Condyloma Acuminanta (commonly known as external genital or perianal warts) within the past two months, AND ≤ 124 days of Veregen therapy in the past 365 days Additional criteria Limited to 18 years and older Max quantity per claim = 30 grams Limited to 60 grams per 365 days Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Skeletal Muscle Relaxants (Implemented 03/20/2006) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents Chlorzoxazone 500 mg tablet (Parafon) Cyclobenzaprine HCl 10mg tablet (Flexeril) Methocarbamol (Robaxin) Preferred agents with criteria Baclofen tablet (Lioresal) Tizanidine HCl tablet (Zanaflex Tablet) Nonpreferred agents Carisoprodol (Soma) Carisoprodol/Aspirin (Soma Compound) Carisoprodol/Aspirin/Codeine (Soma Compound w/ Codeine) Chlorzoxazone 375 mg, 750 mg tablet (Lorzone) Cyclobenzaprine HCl 5mg, 7.5mg tablet (Flexeril, Fexmid) Cyclobenzaprine HCl extended-release capsule (Amrix) Dantrolene sodium (Dantrium) Metaxalone (Skelaxin) Orphenadrine citrate (Norflex) Orphenadrine/aspirin/caffeine (Norgesic) Tizanidine HCL capsule (Zanaflex Capsule) Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Approval criteria for preferred agents with criteria Baclofen tablet (Lioresal) and tizanidine HCl tablet (Zanaflex tablet) One of the following diagnoses: ICD-9 333.7 340 342.1 343 Description Symptomatic torsion dystonia Multiple sclerosis Spastic hemiplegia Infantile cerebral palsy ICD-9 433.x 1 767.4 781.0 806 345.6 430 Infantile spasms Subarachnoid hemorrhage 851 900 431 432 Intracerebral hemorrhage Other and unspecified intracranial hemorrhage 907.2 952 Top of the document Description Occlusion and stenosis of precerebral arteries with cerebral infarction Injury to spine and spinal cord Abnormal involuntary movements Fracture of vertebral column with spinal cord injury Cerebral laceration and contusion Injury to blood vessels of head and neck Late effect of spinal cord injury Spinal cord injury without evidence of spinal bone injury Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Sodium chloride 7% Inhalation Solution (Hyper-Sal 7%) (Implemented 05/24/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of cystic fibrosis within the past three years Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Sodium Oxybate (Xyrem) (Implemented 10/10/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis for Narcolepsy with Cataplexy in past two years Denial criteria Claim for a Sedative/Hypnotic Agent in previous thirty days Diagnosis of Alcohol Abuse in the previous eighteen months Diagnosis of Fibromyalgia in the previous two years Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Somatropin (Genotropin) (Implemented 01/24/2007) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Must meet one of the following five criteria: Criterion 1: Age < 18 years of age, AND Submitted diagnosis of Prader-Willi Syndrome (PWS) in the last two years Criterion 2: Age < 18 years of age, AND Submitted diagnosis of Turner Syndrome in the last two years Criterion 3: Age < 18 years of age, AND Submitted ICD-9 diagnosis in the last two years: o Pituitary dwarfism, OR o Panhypopituitarism, OR o Iatrogenic pituitary disorder, OR o Unspecified disorder of the pituitary gland, OR o Craniopharyngioma, OR o Septo-optic dysplasia AND all of the following: Growth Hormone Deficiency (GHD) confirmed by provocative GH Stimulation Testing, AND MRI or CT scan within the past two years, AND Documented test for hypothyroidism (T3 or T4 level) within the past two years, AND Recipients > 14 years of age: X-ray of the femur or finger within 365 days Criterion 4: Age > 18 years of age, AND Submitted ICD-9 diagnosis for one of the following in the last two years: o Pituitary dwarfism, OR o Iatrogenic pituitary disorder, OR o Unspecified disorder of the pituitary gland AND o GHD confirmed by provocative GH stimulation testing within the last two years. Criterion 5: Age > 18 years of age, AND Submitted ICD-9 diagnosis of panhypopituitarism, AND Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Two of the following appropriate replacement therapies (five paid claims in the past 180 days): o Thyroid hormones o Hydrocortisone o Estrogen o Testosterone, OR GHD confirmed by GH Stimulation testing or IGF-1 within the past two years Denial criteria History of any of the following diagnoses: o Age > 65 years of age o History of malignancy in the past 365 days o History of renal transplant in the past 365 days o Pregnancy History of Prader-Willi Syndrome concurrently with any of the following diagnoses: o Severe obesity o Sleep apnea o History of severe respiratory impairment Growth Hormone Continuation Criteria Point-of-sale continuation criteria have been added to the current growth hormone criteria for certain diagnoses. The following point-of-sale continuation criteria were implemented on Nov. 16, 2011: For recipients < age 13 years for females and < age 14 for males with a billed diagnosis of pituitary dwarfism within the previous 2 years AND a paid claim in Medicaid history for growth hormone in the previous 6 months. For recipients < age 18 years with a billed diagnosis of panhypopituitarism, Turner’s syndrome, Prader-Willi syndrome OR septioptic dysplasia within the previous 2 years AND a paid claim in Medicaid history for growth hormone within the previous 6 months. Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Somatropin (Humatrope) (Implemented 01/24/2007) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Must meet one of the following four criteria: Criterion 1: Age < 18 years of age, AND Submitted diagnosis of Turner Syndrome in the last two years Criterion 2: Age < 18 years of age, AND Submitted ICD-9 diagnosis in the last two years of: o Pituitary dwarfism, OR o Panhypopituitarism, OR o Iatrogenic pituitary disorder, OR o Unspecified disorder of the pituitary gland, OR o Craniopharyngioma, OR o Septo-optic dysplasia AND all of the following in the last two years: o Growth Hormone Deficiency (GHD) confirmed by provocative GH Stimulation Testing, AND o MRI or CT scan within the past two years, AND o Documented test for hypothyroidism (T3 or T4 level) within the past two years, AND o Recipients > 14 years of age: X-ray of the femur or finger within 365 days Criterion 3: Age > 18 years of age, AND Submitted ICD-9 diagnosis for one of the following in the last two years: o Pituitary dwarfism OR o Iatrogenic pituitary disorder OR o Unspecified disorder of the pituitary gland AND o GHD confirmed by provocative GH stimulation testing within the last two years. Criterion 4: Age > 18 years of age, AND Submitted ICD-9 diagnosis of panhypopituitarism, AND Two of the following appropriate replacement therapies (five claims in the past 180 days): o Thyroid hormones Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria o o o o Hydrocortisone Estrogen Testosterone, OR GHD confirmed by GH stimulation testing or IGF-1 within the past two years Denial criteria History of any of the following diagnoses: o Age > 65 years of age o History of malignancy in the past 365 days o History of renal transplant in the past 365 days o Pregnancy Growth Hormone Continuation Criteria Point-of-sale continuation criteria have been added to the current growth hormone criteria for certain diagnoses. The following point-of-sale continuation criteria were implemented on Nov. 16, 2011: For recipients < age 13 years for females and < age 14 for males with a billed diagnosis of pituitary dwarfism within the previous 2 years AND a paid claim in Medicaid history for growth hormone in the previous 6 months. For recipients < age 18 years with a billed diagnosis of panhypopituitarism, Turner’s syndrome, Prader-Willi syndrome OR septioptic dysplasia within the previous 2 years AND a paid claim in Medicaid history for growth hormone within the previous 6 months. Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Somatropin (Norditropin and Saizen) (Implemented 01/24/2007) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Must meet one of the following three criteria: Criterion 1: Age < 18 years of age, AND Submitted ICD-9 diagnosis in the last two years: o Pituitary dwarfism OR o Panhypopituitarism OR o Iatrogenic pituitary disorder OR o Unspecified disorder of the pituitary gland OR o Craniopharyngioma OR o Septo-optic dysplasia AND all of the following in the last two years: o Growth Hormone Deficiency (GHD) confirmed by provocative GH stimulation testing, AND o MRI or CT scan within the past 2 years, AND o Documented test for hypothyroidism (T3 or T4 level) within the past two years, AND o Recipients > 14 years of age: X-ray of the femur or finger within 365 days Criterion 2: Age > 18 years of age, AND Submitted ICD-9 diagnosis for one of the following in the last two years: o Pituitary dwarfism, OR o Iatrogenic pituitary disorder, OR o Unspecified disorder of the pituitary gland AND o GHD confirmed by provocative GH stimulation testing within the last two years. Criterion 3: Age > 18 years of age, AND Submitted ICD-9 diagnosis of panhypopituitarism, AND Two of the following appropriate replacement therapies (five claims in the past 180 days): o Thyroid hormones o Hydrocortisone o Estrogen o Testosterone, OR Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria o GHD confirmed by GH stimulation testing or IGF-1 within the past two years, Denial criteria Age > 65 years of age History of malignancy in the past 365 days History of renal transplant in the past 365 days Pregnancy Growth Hormone Continuation Criteria Point-of-sale continuation criteria have been added to the current growth hormone criteria for certain diagnoses. The following point-of-sale continuation criteria were implemented on Nov. 16, 2011: For recipients < age 13 years for females and < age 14 for males with a billed diagnosis of pituitary dwarfism within the previous 2 years AND a paid claim in Medicaid history for growth hormone in the previous 6 months. For recipients < age 18 years with a billed diagnosis of panhypopituitarism, Turner’s syndrome, Prader-Willi syndrome OR septioptic dysplasia within the previous 2 years AND a paid claim in Medicaid history for growth hormone within the previous 6 months. Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Somatropin (Nutropin) (Implemented 01/24/2007) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Must meet one of the following six criteria: Criterion 1: Age <18 years of age, AND Submitted diagnosis of Turner Syndrome in the last two years Criterion 2: Age < 18 years of age, AND Submitted diagnosis of chronic renal Insufficiency Criterion 3: Age < 18 years of age, AND Submitted diagnosis of end-stage renal disease awaiting transplantation Criterion 4: Age < 18 years of age, AND Submitted ICD-9 diagnosis in the last two years: o Pituitary dwarfism, OR o Panhypopituitarism, OR o Iatrogenic pituitary disorder, OR o Unspecified disorder of the pituitary gland, OR o Craniopharyngioma, OR o Septo-optic dysplasia AND all of the following in the last two years: o Growth Hormone Deficiency (GHD) confirmed by provocative GH stimulation testing, AND o MRI or CT scan within past two years, AND o Documented test for hypothyroidism (T3 or T4 level) within past 2 years, AND o Recipients > 14 years of age: X-ray of the femur or finger within 365 days Criterion 5: Age > 18 years of age, AND Submitted ICD-9 diagnosis for one of the following in the last two years: o Pituitary dwarfism, OR o Iatrogenic pituitary disorder, OR o Unspecified disorder of the pituitary gland AND o GHD confirmed by provocative GH stimulation testing within the last two years. Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Criterion 6: Age > 18 years of age, AND Submitted ICD-9 diagnosis of panhypopituitarism, AND Two of the following appropriate replacement therapies (five claims in the past 180 days): o Thyroid hormones o Hydrocortisone o Estrogen o Testosterone, OR o GHD confirmed by GH stimulation testing or IGF-1 within the past two years. Denial criteria Age > 65 years of age History of malignancy in the past 365 days History of renal transplant in the past 365 days Pregnancy Growth Hormone Continuation Criteria Point-of-sale continuation criteria have been added to the current growth hormone criteria for certain diagnoses. The following point-of-sale continuation criteria were implemented on Nov. 16, 2011: For recipients < age 13 years for females and < age 14 for males with a billed diagnosis of pituitary dwarfism within the previous 2 years AND a paid claim in Medicaid history for growth hormone in the previous 6 months. For recipients < age 18 years with a billed diagnosis of panhypopituitarism, Turner’s syndrome, Prader-Willi syndrome OR septioptic dysplasia within the previous 2 years AND a paid claim in Medicaid history for growth hormone within the previous 6 months. Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Somatropin (Omnitrope) (Implemented 01/24/2007) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Must meet one of the following six criteria: Criterion 1: Age < 18 years of age, AND Submitted diagnosis of Prader-Willi Syndrome (PWS) in the last two years Criterion 2: Age < 18 years of age, AND Submitted diagnosis of Turner Syndrome in the last two years Criterion 3: Age < 18 years of age, AND Submitted diagnosis of chronic renal insufficiency Criterion 4: Age < 18 years of age, AND Submitted diagnosis of end-stage renal disease awaiting transplantation Criterion 5: Age < 18 years of age, AND Submitted ICD-9 diagnosis in the last two years: o Pituitary dwarfism, OR o Panhypopituitarism, OR o Iatrogenic pituitary disorder, OR o Unspecified disorder of the pituitary gland OR o Craniopharyngioma, OR o Septo-optic dysplasia AND all of the following in the last two years: o Growth Hormone Deficiency (GHD) confirmed by provocative GH stimulation testing, AND o MRI or CT scan within past 2 years, AND o Documented test for hypothyroidism (T3 or T4 level) within past two years, AND o Recipients > 14 years of age: X-ray of the femur or finger within 365 days Criterion 6: Age >18 years of age, AND Submitted ICD-9 diagnosis for one of the following in the last two years: o Pituitary dwarfism, OR o Iatrogenic pituitary disorder, OR o Unspecified disorder of the pituitary gland Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria AND o GHD confirmed by provocative GH stimulation testing within the last two years. Criterion 7: Age > 18 years of age, AND Submitted ICD-9 diagnosis of panhypopituitarism, AND Two of the following appropriate replacement therapies (five claims in the past 180 days): o Thyroid hormones o Hydrocortisone o Estrogen o Testosterone, OR o GHD confirmed by GH stimulation testing or IGF-1 within the past two years Denial criteria Age > 65 years of age History of malignancy in the past 365 days History of renal transplant in the past 365 days Pregnancy Growth Hormone Continuation Criteria Point-of-sale continuation criteria have been added to the current growth hormone criteria for certain diagnoses. The following point-of-sale continuation criteria were implemented on Nov. 16, 2011: For recipients < age 13 years for females and < age 14 for males with a billed diagnosis of pituitary dwarfism within the previous 2 years AND a paid claim in Medicaid history for growth hormone in the previous 6 months. For recipients < age 18 years with a billed diagnosis of panhypopituitarism, Turner’s syndrome, Prader-Willi syndrome OR septioptic dysplasia within the previous 2 years AND a paid claim in Medicaid history for growth hormone within the previous 6 months. Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Somatropin (Tev-tropin) (Implemented 01/24/2007) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Must meet one of the following criteria: Age < 18 years of age, AND Submitted ICD-9 diagnosis in the last two years: o Pituitary dwarfism, OR o Panhypopituitarism, OR o Iatrogenic pituitary disorder, OR o Unspecified disorder of the pituitary gland, OR o Craniopharyngioma, OR o Septo-optic dysplasia AND all of the following in the last two years: o Growth Hormone Deficiency (GHD) confirmed by provocative GH stimulation testing, AND o MRI or CT scan within the past 2 years, AND o Documented test for hypothyroidism (T3 or T4 level) within the past two years, AND o Recipients > 14 years of age: X-ray of the femur or finger within 365 days Denial criteria Age > 65 years of age History of malignancy in the past 365 days History of renal transplant in the past 365 days Pregnancy Growth Hormone Continuation Criteria Point-of-sale continuation criteria have been added to the current growth hormone criteria for certain diagnoses. The following point-of-sale continuation criteria were implemented on Nov. 16, 2011: For recipients < age 13 years for females and < age 14 for males with a billed diagnosis of pituitary dwarfism within the previous 2 years AND a paid claim in Medicaid history for growth hormone in the previous 6 months. Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria For recipients < age 18 years with a billed diagnosis of panhypopituitarism, Turner’s syndrome, Prader-Willi syndrome OR septioptic dysplasia within the previous 2 years AND a paid claim in Medicaid history for growth hormone within the previous 6 months. Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Somatropin (Zorbtive) (Implemented 01/24/2007) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Criterion 6: Age > 18 years of age, AND Submitted ICD-9 diagnosis of short bowel syndrome Additional criteria Quantity limits apply Denial criteria Age > 65 years of age History of malignancy in the past 365 days History of renal transplant in the past 365 days Pregnancy Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Spinosad 0.9% Topical Suspension (Natroba) (Implemented 06/21/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Natroba Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Sucralfate Suspension (Carafate) (Implemented 10/11/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria < 6 years of age, OR NPO (Appendix A) Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Sulfamethoxazole-Trimethoprim 800-160/20ml U.D. Cup (Implemented 04/08/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Currently LTC Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Sunitinib (Sutent) Capsule (Implemented 01/13/2015) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Sutent Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tacrolimus (Astagraf XL) (Implemented 12/10/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Astagraf XL Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tacrolimus (Protopic) (Implemented 03/12/2007) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria No therapeutic duplication with other strengths of Tacrolimus (Protopic), AND No therapeutic duplication Pimecrolimus (Elidel), AND At least two paid Medicaid drug claims of topical corticosteroid agents, each containing a different drug entity, AND At least one of the claims for the topical corticosteroid being at least medium potency or higher filled in the previous 14-45 day period. Denial criteria History of Psoriasis in previous two years Therapeutic duplication with other strengths of tacrolimus (Protopic) Therapeutic duplication with pimecrolimus (Elidel) No claim for at least two topical corticosteroids in Medicaid history No claim for a medium potency or higher topical corticosteroid in the previous 14-45 day period. Additional criteria Tacrolimus 0.03% o Age > 2 years of age o Quantity limits apply Tacrolimus 0.1% o Age > 16 years of age o Quantity limits apply Denial criteria Tacrolimus 0.03% o Therapeutic duplication with pimecrolimus (Elidel) o Therapeutic duplication with tacrolimus (Protopic) 0.1% Tacrolimus 0.1% o Therapeutic duplication with pimecrolimus (Elidel) o Therapeutic duplication with tacrolimus (Protopic) 0.03% History of Psoriasis in previous two years Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria No claim for at least two topical corticosteroids in Medicaid history No claim for a medium potency or higher topical corticosteroid in the previous 14-45 day period. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tadalafil (Adcirca) (Implemented 09/15/2009) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of pulmonary heart disease in the last 365 days, OR Diagnosis of persistent fetal circulation in the last 365 days, OR Diagnosis of chronic respiratory disease arising in the perinatal period in the last 365 days Denial criteria One of the following diagnoses in the last 180 days: o Cavernosal fibros, OR o Hypotension, OR o Leukemia, OR o Life-threatening arrhythmia, OR o Malignant hypertension, OR o Multiple myeloma, OR o Myocardial infarction, OR o Peyronie’s disease, OR o Retinitis pigmentosa, OR o Sickle cell disease, OR o Stroke, OR o Unstable angina History of any of the following in the last 45 days: o Alpha-adrenergic blockers o Tamsulosin Concurrent use of any the following: o Indinavir o Lopinavir-ritonavir o Ritonavir Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tamoxifen 10mg/5ml Oral Solution (Soltamox) (Implemented 03/19/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Soltamox Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Targeted Immune Modulators (Implemented 10/17/2007) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Preferred agents with criteria Adalimumab (Humira) Cetrolizumab (Cimzia) Etanercept (Enbrel) Nonpreferred agents Abatacept (Orencia) Anakinra (Kineret) Apremilast (Otezla) Golimumab (Simponi) Infliximab (Remicade) Ustekinumab (Stelara) Tocilizumab (Actemra) Tofacitinib (Xeljanz) Approval criteria for preferred agents with criteria Approval criteria for Cimzia Must meet one of the following five criteria: Criterion 1: Age ≥ 18, AND Submitted ICD-9 diagnosis for Crohn’s disease or regional enteritis (Table 5) in the past two years, AND Submitted ICD-9 diagnosis for fistula in the past 730 days (Table 5), AND Maximum of 1 Cimzia Starter Kit every 2 years or 1 syringe kit or 1 vial kit every 23 days Criterion 2: Age ≥18, AND Submitted ICD-9 diagnosis of Crohn’s disease or regional enteritis (Table 5) in the past two years, AND ≥180 days of drug therapy in the past 365 days with any of the following: systemic glucocorticoid AND mesalamine therapy, mercaptopurine, or azathioprine, AND Maximum of 1 Cimzia Starter Kit every 2 years or 1 syringe kit or 1 vial kit every 23 days Criterion 3: Age ≥18, AND Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Submitted ICD-9 diagnosis of Crohn’s disease or regional enteritis (Table 5) in the past two years, AND Paid drug claim for cetrolizumab (Cimzia) in the past 45 days, AND Maximum of 1 syringe kit or 1 vial kit every 23 days Criterion 4: Age ≥18, AND Submitted ICD-9 diagnosis of rheumatoid arthritis (Table 5) in the past two years, AND ≥6 claims of drug therapy in the past 365 days with any of the following: gold compounds, hydroxychloroquine, penicillamine, methotrexate, azathioprine, sulfasalazine, or leflunomide, AND Maximum of 1 Cimzia Starter Kit every 2 years or 1syringe kit or 1 vial kit every 23 days Criterion 5: Submitted ICD-9 diagnosis of rheumatoid arthritis (Table 5) in the past two years, AND Paid Drug claim for cetrolizumab (Cimzia) in the past 45 days (signifying above criteria previously met), AND Maximum of 1 syringe kit or 1 vial kit every 23 days Criterion 6: Submitted ICD-9 diagnosis of psoriatic arthropathy (Table 5) in the past two years, AND Age ≥18, AND ≥6 claims of drug therapy in the past 365 days with any of the following: gold compounds, hydroxychloroquine, penicillamine, methotrexate, azathioprine, sulfasalazine, or leflunomide, AND Maximum of 1 Cimzia Starter Kit every 2 years or 1syringe kit or 1 vial kit every 23 days Criterion 5: Submitted ICD-9 diagnosis of psoriatic arthropathy (Table 5) in the past two years, AND Age ≥18, AND Paid Drug claim for cetrolizumab (Cimzia) in the past 45 days (signifying above criteria previously met), AND Maximum of 1 syringe kit or 1 vial kit every 23 days Approval criteria for Enbrel Must meet one of the following six criteria: Criterion 1: Submitted ICD-9 diagnosis of psoriasis (Table 5) in the past two years, AND Age > 17, AND Paid Drug claim for etanercept (Enbrel) in the past 45 days (signifying above criteria previously met) Criterion 2: Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Submitted diagnosis of psoriasis (Table 5) in the past two years, AND Age > 17, AND During days 180 to 395 days ago, a total of > 180 days of topical drug therapy with: anthralin, calcipotriene, corticosteroids, or tazarotene in past 395 days, AND During days 1 to 210 ago, a total of > 180 days of systemic drug therapy with: cyclosporine, methotrexate, or acitretin, AND Topical drug therapy trial occurred before systemic drug therapy Criterion 3: Submitted ICD-9 diagnosis of ankylosing spondylosis (Table 5) in the past two years Criterion 4: Submitted ICD-9 diagnosis of rheumatoid arthritis (Table 5) or psoriatic arthropathy (Table 5) in the past two years, AND >180 days of drug therapy in the past 365 days with any of the following: gold compounds, hydroxychloroquine, penicillamine, methotrexate, azathioprine, sulfasalazine, or leflunomide Criterion 5: Submitted ICD-9 diagnosis of rheumatoid arthritis (Table 5) or psoriatic arthropathy (Table 5) in the past two years, AND Paid Drug claim for etanercept (Enbrel) in the past 45 days (signifying above criteria previously met) Criterion 6: Submitted ICD-9 diagnosis of rheumatoid arthritis (Table 5) in the past two years, AND Age < 18 Approval criteria for Humira Must meet one of the following eight criteria: Criterion 1: Submitted ICD-9 diagnosis of ankylosing spondylosis (Table 5) in the past two years Criterion 2: Submitted ICD-9 diagnosis for rheumatoid arthritis (Table 5) or psoriatic arthropathy (Table 5) in the past two years, AND One of the following : > Six claims for any of the following in the past 365 days: gold compounds, hydroxychloroquine, penicillamine, methotrexate, azathioprine, sulfasalazine, or leflunomide, OR Paid Drug claim for adalimumab (Humira) in the past 45 days (signifying above criteria previously met) Criterion 3: Age > 18 years, AND Submitted ICD-9 diagnosis code for Crohn’s disease (Table 5) or regional enteritis (Table 5) in the past two years, AND Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Submitted ICD-9 diagnosis code for fistula (Table 5) in the past two years Criterion 4: Age > 18 years, AND Submitted ICD-9 diagnosis code for Crohn’s disease (Table 5) or regional enteritis (Table 5) in the past two years, AND > 180 days drug therapy of one of the following regimens in the past 365 days: concurrent systemic glucocorticoid AND mesalamine therapy, OR mercaptopurine, OR azathioprine Criterion 5: Age > 18 years, AND Submitted ICD-9 diagnosis code for Crohn’s disease (Table 5) or regional enteritis (Table 5) in the past two years, AND Drug claim for adalimumab (Humira) in the past 45 days (signifying above criteria previously met) Criterion 6: Submitted ICD-9 diagnosis of psoriasis (Table 5) in the past two years, AND Age > 18, AND Paid drug claim for adalimumab (Humira) in the past 45 days (signifying above criteria previously met) Criterion 7: Submitted diagnosis of ICD-9 psoriasis (Table 5) in the past two years, AND Age > 18, AND During days 180 to 395 days ago, a total of >180 days of topical drug therapy with: anthralin, calcipotriene, corticosteroids, or tazarotene in past 395 days, AND During days 1 to 210 ago, a total of > 180 days of systemic drug therapy with: cyclosporine, methotrexate, or acitretin, AND Topical drug therapy trial occurred before systemic drug therapy Criterion 8: Submitted ICD-9 diagnosis of rheumatoid arthritis (Table 5) in the past two years, AND Age < 18 Table 5 – Targeted immune modulator diagnoses Drug Cimzia Cimzia Cimzia Cimzia Cimzia ICD-9 555 555 565.1 567.2 567.2 Description Crohn's disease Regional enteritis Anal fistula Peritonitis (acute) generalized Peritoneal abscess Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Cimzia Cimzia Cimzia Cimzia Cimzia Cimzia Cimzia Cimzia Cimzia Cimzia Cimzia Cimzia Enbrel Enbrel Enbrel Enbrel Humira Humira Humira Humira Humira Humira Humira Humira Humira Humira Humira Humira Humira Humira Humira Humira Humira Humira Humira Humira 567.2 567.2 567.3 567.3 567.3 567.3 567.8 567.8 567.8 569.8 696.0 714 696.0 696.1 714 720 555 555 556 565.1 567.2 567.2 567.2 567.3 567.3 567.3 567.3 567.3 567.8 567.8 567.9 569.8 696.0 696.1 714 720 Spontaneous bacterial peritonitis Other suppurative peritonitis Retroperitoneal infections Psoas muscle abscess Other retroperitoneal abscess Other retroperitoneal infections Choleperitonitis Sclerosing mesenteritis Other specified peritonitis Fistula of intestine excluding rectum and anus Psoriatic arthropathy Rheumatoid arthritis and other inflammatory polyarthropathies Psoriatic arthropathy Other psoriasis Rheumatoid arthritis and other inflammatory polyarthropathies Ankylosing spondylosis Crohn's disease Regional enteritis Ulcerative Colitis Anal fistula Peritonitis (acute) generalized Peritoneal abscess Spontaneous bacterial peritonitis Other suppurative peritonitis Retroperitoneal infections Psoas muscle abscess Other retroperitoneal abscess Other retroperitoneal infections Choleperitonitis Sclerosing mesenteritis Other specified peritonitis Fistula of intestine excluding rectum and anus Psoriatic arthropathy Other psoriasis Rheumatoid arthritis and other inflammatory polyarthropathies Ankylosing spondylosis Link to Memorandum: Humira Link to Memorandum: Enbrel Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tasimelteon Capsule (Hetlioz) (Implemented 09/23/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Hetlioz Capsule Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tavaborole 5% Topical Solution (Kerydin) (Implemented 01/13/2015) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Itraconazole Capsule [Sporanox] Terbinafine Tablet [Lamisil] Drugs that require manual PA Ciclopirox 8% Topical Solution [Penlac] Efinaconazole 10% Topical Solution [Jublia] Tavaborole 5% Topical Solution [Kerydin] Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tazarotene Gel/Cream (Tazorac) (Implemented 06/19/2006) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drug Tazorac Approval criteria (New Start) Diagnosis of psoriasis in Medicaid history in previous 365 days, AND 2 paid claims of a topical corticosteroid in previous 27-60 days, AND At least one paid claim for a topical corticosteroid must be from the very high potency category. Approval criteria (Continuation Criteria) Diagnosis of psoriasis in Medicaid history in previous 365 days, AND The incoming claim matches claim in history in the previous 45 days of Tazorac, AND At least two claims of a topical corticosteroid in previous 60 days in drug claim history with at least one topical corticosteroid claim 14-60 days back in history. Denial criteria History of acne vulgaris in the last 60 days Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tedizolid (Sivextro) (Implemented 01/13/2015) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria No therapeutic duplication between a claim of the tablets and claim of the vials within the same month Additional criteria Age >/= 18 years of age Quantity Limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Teduglutide Vial (Gattex) (Implemented 07/09/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Gattex Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Telithromycin (Ketek) (Implemented 09/12/2007) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Denial criteria Submitted ICD-9 diagnosis myasthenia gravis in the past 730 days. Submitted ICD-9 diagnosis hepatitis in the past 730 days. Submitted ICD-9 diagnosis hepatic impairment in the past 730 days. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Temazepam 7.5mg and 22.5mg Capsule (Restoril) (Implemented 07/09/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Temazepam 7.5mg capsule Temazepam 22.5 mg capsule Exceptions (Beneficiaries that do not require a Prior Authorization) Temazepam 7.5mg capsule Long Term Care Beneficiaries Beneficiaries that are 65 years of age of older *No PA required for requests for Temazepam 7.5mg Capsule for the Beneficiaries listed above* Approval criteria Up to 124 Units of any solid oral benzodiazepines paid by Medicaid per the previous 31 calendar days. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tenofovir (Viread) (Implemented 12/19/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria A billed diagnosis of HIV/AIDS in the Medicaid history in previous 2 years; OR Paid drug claim(s) in Medicaid history of other antiretroviral therapy (ART), such as non-nucleoside reverse transcriptase inhibitors (NNRTI), OR protease inhibitors (PI), OR integrase strand transfer inhibitor (INSTI) in previous 6 months. A billed diagnosis of Hepatitis in the Medicaid history in previous 2 years. Denial criteria Therapeutic duplication edit: paid claim within previous 30 days for Truvada; OR Absence of approval criteria. Approval criteria for PrEp will require a manual review PA process based upon the following: Documentation from prescriber that patient is at high risk for acquiring HIV infection; AND Negative HIV test before starting and every 3 months thereafter; AND Pregnancy test before starting and every 3 months thereafter. If pregnant, provide documentation of patient understanding of potential risks and benefits of using Truvada®, including contraindication with breastfeeding; AND Serum creatinine lab tests obtained prior to initiation, then every 6 months. Creatinine clearance should be >60 mL/min; AND Documented testing for Hepatitis B Virus (HBV) and results submitted. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tenofovir 300mg/Emtricitabine 200mg (Truvada) (Implemented 12/19/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria for treatment of HIV-1 infection A billed diagnosis of HIV/AIDS in the Medicaid history in previous 2 years; OR Paid drug claim(s) in Medicaid history of other antiretroviral therapy (ART), such as non-nucleoside reverse transcriptase inhibitors (NNRTI), OR protease inhibitors (PI), OR integrase strand transfer inhibitor (INSTI) in previous 6 months. If there is no HIV/AIDS diagnosis in Medicaid history and no records of other ART in the Medicaid drug profile, prescriber will be required to follow the manual review process and submit documentation confirming positive HIV diagnosis. Denial criteria Therapeutic duplication edit: paid claim within previous 30 days for Emtriva® (emtricitabine) or Viread® (tenofovir disoproxil fumarate); OR Absence of approval criteria. Approval criteria for PrEp will require a manual review PA process based upon the following: Documentation from prescriber that patient is at high risk for acquiring HIV infection; AND Negative HIV test before starting and every 3 months thereafter; AND Pregnancy test before starting and every 3 months thereafter. If pregnant, provide documentation of patient understanding of potential risks and benefits of using Truvada®, including contraindication with breastfeeding; AND Serum creatinine lab tests obtained prior to initiation, then every 6 months. Creatinine clearance should be >60 mL/min; AND Documented testing for Hepatitis B Virus (HBV) and results submitted. Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Terbinafine 125mg and 187.5mg Granules Packet (Lamisil) (Implemented 04/12/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria At least one paid claim in Medicaid history for fluconazole suspension in the past 14-90 days, AND At least 2 paid claims in Medicaid history for griseofulvin suspension in the previous 14-90 days AND No therapeutic duplication between two different strengths of Lamisil granules. Additional criteria Quantity edits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Testosterone Replacement Products (Implemented 01/18/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drug Testosterone Cypionate 100 mg/ml Injection Testosterone Cypionate 200 mg/ml Injection Testosterone Enanthate 200 mg/ml Injection Approval criteria Male Diagnosis of one of the following diagnoses in the previous 2 years: o Hypospadias o Klinefelter Syndrome o Kallmann Syndrome o Panhypopituitarism o Prader-Willi Syndrome Denial criteria Female Diagnosis of one of the following diagnoses in the previous 2 years: o Decreased libido o Impotence o Any other sexual dysfunction diagnoses Exceptions (Request through Manual Review Process) Approve for women with diagnosis of breast cancer or hormone-responsive tumor in history Testosterone Products which require a Manual Review Androderm 2 mg/24hr Patch Androderm 4 mg/24hr Patch Androgel 1% Gel Android 10 mg Capsule Androxy 10 mg Tablet Aveed 750 mg/3 ml Vial Axiron 30 mg Actuation Solution Fortesta 10 mg Gel Pump Methitest 10 mg Tablet Striant 30 mg Mucoadhesive Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Testim 1% Gel Testred Capsules Additional Criteria Quantity Limits Apply Link to Memorandum Link to Updated Criteria Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tetrabenazine Tablet (Xenazine) (Implemented 09/21/2009) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of Huntington’s Disease with Chorea in the past 3 years. Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Timolol Maleate/Dorzolamide HCL (Cosopt PF) (Implemented 07/09/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Cosopt PF Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tiotropium Bromide Inhaler (Spiriva) (Implemented 12/19/2012) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Approval criteria Diagnosis of COPD in Medicaid history in previous 2 years; AND No therapeutic duplication with overlapping days’ supply between Anoro Ellipta, Spiriva, and/or Tudorza; AND Medicaid recipient is > 18 years of age; AND Denial criteria Lack of approval criteria; OR Diagnosis of asthma in Medicaid history in previous 2 years Additional criteria Quantity edits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tobacco-cessation Products (Implemented 11/15/2005) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Bupropion (Zyban) Currently pregnant, OR < 60 days post partum, OR < 187 days of bupropion (Zyban) in the last 365 days. Nicotine Replacement Therapy (Nicotine Gum and Patches) Currently pregnant, OR < 60 days post partum, OR < 187 days of nicotine replacement therapy in the last 365 days. Varenicline (Chantix) Implemented 08/01/2007 Currently pregnant, OR < 60 days post partum, OR < 187 days of varenicline in the last 365 days. Denial criteria Bupropion (Zyban) Therapeutic duplication of Chantix. Nicotine Replacement Therapy (Gum and Patches) Therapeutic duplication of nicotine gum and nicotine patches. Therapeutic duplication of Chantix. Varenicline (Chantix) Therapeutic duplication of nicotine gum Therapeutic duplication of nicotine patches Therapeutic duplication of Zyban Link to Memorandum Link to Memorandum for Chantix Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tobramycin Inhalation Solution (TOBI – Brand Name) (Implemented 01/09/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of cystic fibrosis in medical history Denial criteria History of Cayston in the past 50 days History of J Code for Tobramycin Injection in the past 60 days History of Bethkis in the past 50 days Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tobramycin Inhalation Solution (Generic) (Bethkis) (Implemented 03/18/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Generic Tobramycin Inhalation Solution Bethkis Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tobramycin Inhalation Powder (TOBI Podhaler) (Implemented 12/10/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization TOBI Podhaler Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Topical Corticosteroids (Implemented 03/26/2008) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria A trial of at least two different products containing a different drug entity within the same potency category that do not require prior authorization within the previous 60 days. (Appendix C.) TOPICAL CORTICOSTEROID PA LIST BY POTENCY POTENCY PA STATUS DRUG NAME VERY HIGH No PA CLOBETASOL PROPIONATE 0.05% CREAM [TEMOVATE® CREAM] VERY HIGH No PA CLOBETASOL PROPIONATE 0.05% EMOLLIENT CREAM [TEMOVATE EMOLLIENT® CREAM] VERY HIGH No PA CLOBETASOL PROPIONATE 0.05% GEL VERY HIGH No PA CLOBETASOL PROPIONATE 0.05% OINTMENT [TEMOVATE® OINTMENT] VERY HIGH No PA CLOBETASOL PROPIONATE 0.05% SOLUTION VERY HIGH No PA HALOBETASOL PROPIONATE 0.05% CREAM [ULTRAVATE® CREAM] VERY HIGH No PA HALOBETASOL PROPIONATE 0.05% OINTMENT [ULTRAVATE® OINTMENT] VERY HIGH PA BETAMETHASONE DIPROPIONATE AUGMENTED 0.05% OINTMENT [DIPROLENE® OINTMENT] VERY HIGH PA CLOBETASOL PROPIONATE 0.05% FOAM [OLUX® FOAM] VERY HIGH PA CLOBETASOL PROPIONATE 0.05% EMOLLIENT FOAM [OLUX-E® FOAM] VERY HIGH PA CLOBETASOL PROPIONATE 0.05% LOTION [CLOBEX® LOTION] VERY HIGH PA CLOBETASOL PROPIONATE 0.05% SHAMPOO [CLOBEX® SHAMPOO] VERY HIGH PA CLOBETASOL PROPIONATE 0.05% SPRAY [CLOBEX® SPRAY] VERY HIGH PA DIFLORASONE DIACETATE 0.05% OINTMENT VERY HIGH PA FLUOCINONIDE 0.01% CREAM [VANOS® CREAM] Continued Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Approval criteria A trial of at least two different products containing a different drug entity within the same potency category that do not require prior authorization within the previous 60 days. (Appendix C.) POTENCY PA STATUS DRUG NAME HIGH No PA HIGH No PA HIGH HIGH HIGH HIGH HIGH HIGH HIGH HIGH HIGH HIGH HIGH HIGH HIGH HIGH HIGH HIGH HIGH HIGH HIGH HIGH No PA No PA No PA No PA No PA No PA No PA No PA No PA No PA No PA PA PA PA PA PA PA PA PA PA HIGH PA HIGH HIGH PA PA BETAMETHASONE DIPROPIONATE 0.05% CREAM BETAMETHASONE DIPROPIONATE AUGMENTED 0.05% CREAM [DIPROLENE AF® CREAM] BETAMETHASONE DIPROPIONATE 0.05% LOTION BETAMETHASONE VALERATE 0.1% OINTMENT DESOXIMETASONE 0.25% CREAM [TOPICORT® CREAM] DIFLORASONE DIACETATE 0.05% CREAM FLUOCINONIDE 0.05% CREAM FLUOCINONIDE 0.05% EMOLLIENT CREAM FLUOCINONIDE 0.05% GEL FLUOCINONIDE 0.05% OINTMENT FLUOCINONIDE 0.05% SOLUTION TRIAMCINOLONE ACETONIDE 0.5% CREAM TRIAMCINOLONE ACETONIDE 0.5% OINTMENT AMCINONIDE 0.1% CREAM AMCINONIDE 0.1% LOTION AMCINONIDE 0.1% OINTMENT BETAMETHASONE DIPROPIONATE 0.05% OINTMENT DESOXIMETASONE 0.05% CREAM [TOPICORT® CREAM] DESOXIMETASONE 0.05% GEL [TOPICORT® GEL] DESOXIMETASONE 0.05% OINTMENT [TOPICORT® OINTMENT] DESOXIMETASONE 0.25% OINTMENT [TOPICORT® OINTMENT] DESOXIMETASONE 0.25% SPRAY [TOPICORT® SPRAY] DIFLORASONE DIACETATE 0.05% EMOLLIENT CREAM [APEXICON® E CREAM] HALCINONIDE 0.1% CREAM [HALOG® CREAM] HALCINONIDE 0.1% OINTMENT [HALOG® OINTMENT] Continued Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Approval criteria A trial of at least two different products containing a different drug entity within the same potency category that do not require prior authorization within the previous 60 days. (Appendix C.) POTENCY PA STATUS DRUG NAME MEDIUM MEDIUM MEDIUM MEDIUM MEDIUM MEDIUM MEDIUM MEDIUM MEDIUM MEDIUM MEDIUM MEDIUM MEDIUM MEDIUM MEDIUM MEDIUM MEDIUM MEDIUM No PA No PA No PA No PA No PA No PA No PA No PA No PA No PA No PA No PA No PA No PA No PA No PA No PA PA MEDIUM PA MEDIUM MEDIUM MEDIUM PA PA PA MEDIUM PA MEDIUM MEDIUM PA PA MEDIUM PA MEDIUM MEDIUM MEDIUM MEDIUM PA PA PA PA MEDIUM PA MEDIUM PA BETAMETHASONE VALERATE 0.1% CREAM BETAMETHASONE VALERATE 0.1% LOTION FLUTICASONE PROPIONATE 0.005% OINTMENT FLUTICASONE PROPIONATE 0.05% CREAM HYDROCORTISONE BUTYRATE 0.1% CREAM HYDROCORTISONE BUTYRATE 0.1% OINTMENT HYDROCORTISONE BUTYRATE 0.1% SOLUTION HYDROCORTISONE VALERATE 0.2% CREAM MOMETASONE FUROATE 0.1% CREAM [ELOCON® CREAM] MOMETASONE FUROATE 0.1% OINTMENT [ELOCON® OINTMENT] MOMETASONE FUROATE 0.1% SOLUTION [ELOCON® SOLUTION] TRIAMCINOLONE ACETONIDE 0.025% CREAM TRIAMCINOLONE ACETONIDE 0.025% LOTION TRIAMCINOLONE ACETONIDE 0.025% OINTMENT TRIAMCINOLONE ACETONIDE 0.1% CREAM TRIAMCINOLONE ACETONIDE 0.1% LOTION TRIAMCINOLONE ACETONIDE 0.1% OINTMENT BETAMETHASONE DIPROPIONATE AUGMENTED 0.05% GEL BETAMETHASONE DIPROPIONATE AUGMENTED 0.05% LOTION [DIPROLENE® LOTION] BETAMETHASONE VALERATE 0.12% FOAM [LUXIQ® FOAM] CLOCORTOLONE PIVALATE 0.1% CREAM [CLODERM® CREAM] FLUOCINOLONE ACETONIDE 0.025% CREAM [SYNALAR® CREAM] FLUOCINOLONE ACETONIDE 0.025% OINTMENT [SYNALAR ® OINTMENT] FLURANDRENOLIDE 4 MCG/SQ CM TAPE [CORDRAN® TAPE] FLUTICASONE PROPIONATE 0.05% LOTION [CUTIVATE® LOTION] HYDROCORTISONE BUTYRATE 0.1% EMOLLIENT CREAM [LOCOID® LIPOCREAM] HYDDROCORTISONE PROBUTATE 0.1% CREAM [PANDEL® CREAM] HYDROCORTISONE VALERATE 0.2% OINTMENT PREDNICARBATE 0.1% CREAM [DERMATOP® CREAM] PREDNICARBATE 0.1% OINTMENT [DERMATOP® OINTMENT] TRIAMCINOLONE ACETONIDE 0.147 MG/GM AEROSOL SPRAY [KENALOG® SPRAY] TRIAMCINOLONE ACETONIDE 0.05% OINTMENT [TRIANEX® OINTMENT] Continued Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Approval criteria A trial of at least two different products containing a different drug entity within the same potency category that do not require prior authorization within the previous 60 days. (Appendix C.) POTENCY PA STATUS LOW No PA LOW LOW No PA No PA LOW No PA LOW LOW LOW LOW LOW LOW LOW LOW LOW LOW LOW LOW LOW No PA No PA No PA No PA No PA No PA No PA No PA PA PA PA PA PA LOW PA LOW PA LOW PA DRUG NAME ALCLOMETASONE DIPROPIONATE 0.05% CREAM ALCLOMETASONE DIPROPIONATE 0.05% OINTMENT DESONIDE 0.05% OINTMENT FLUOCINOLONE ACETONIDE 0.01% OIL [DERMA-SMOOTHE FS®] HYDROCORTISONE 0.5% CREAM HYDROCORTISONE 0.5% OINTMENT HYDROCORTISONE 1% CREAM HYDROCORTISONE 1% OINTMENT HYDROCORTISONE 2.5% CREAM HYDROCORTISONE 2.5% LOTION HYDROCORTISONE 2.5% OINTMENT HYDROCORTISONE ACETATE 0.5% CREAM DESONIDE 0.05% CREAM DESONIDE 0.05% FOAM [VERDESO® FOAM] DESONIDE 0.05% GEL [DESONATE® GEL] DESONIDE 0.05% LOT (DESOWEN ®) FLUOCINOLONE ACETONIDE 0.01% CREAM FLUOCINOLONE ACETONIDE 0.01% SHAMPOO [CAPEX® SHAMPOO] FLUOCINOLONE ACETONIDE 0.01% SOLUTION [SYNALAR® SOLUTION] HYDROCORTISONE 2.5% SOLUTION [TEXACORT® SOLUTION] Denial criteria Failure to meet approval criteria Link to Memorandum Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Topical Products Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require a manual PA Altabax 1% ointment Implemented 09/21/2009 o Generic mupirocin is available without prior authorization Bensal® HP (benzoic acid 6%, salicylic Acid 3%, Oak Bark Extract) Ointment Implemented April 6, 2010 Nucort lotion (hydrocortisone acetate-aloe vera) Implemented 01/12/2010 o o Generic hydrocortisone is available without prior authorization Nuzon gel (hydrocortisone acetate-aloe vera) Implemented 01/12/2010 o o Generic hydrocortisone is available without prior authorization Ulesfia 5% lotion Implemented 01/12/2010 o Permethrin products are available without prior authorization Link to Memorandum Ulesfia Link to Memorandum Bensal HP Link to Memorandum Altabax Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Topiramate XR Capsules (Qudexy XR and Trokendi XR) (Implemented 12/10/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Qudexy XR Trokendi XR Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tramadol Extended-Release (Conzip Capsule and Ryzolt ER Tablet) (Implemented 01/12/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require manual review for prior authorization Tramadol ER (Ultram ER) Drugs that require manual review for prior authorization Conzip capsule Ryzolt ER tablet Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tramadol Immediate-Release (Ultram, Ultracet) (Implemented 04/21/2009) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Diagnosis of cancer in the past 12 months, AND No claim in history for Subutex, AND No claim in history for Suboxone. Paid claim for an antineoplastic in the past 12 months, AND No claim in history for Subutex, AND No claim in history for Suboxone. Therapeutic duplication between short-acting opioids with less than 25% of the days’ supply remaining on the previous claim, OR Therapeutic duplication between a short-acting opioid and tramadol IR (Ultram) with less than 25% of the days’ supply remaining on the previous claim, OR Therapeutic duplication between a short-acting opioid and tramadol/acetaminophen (Ultracet) with less than 25% of the days’ supply remaining on the previous claim. Therapeutic duplication between a tramadol IR and tramadol/acetaminophen (Ultracet) with less than 25% of the days’ supply remaining on the previous claim. Therapeutic duplication between a short-acting opioid and tramadol ODT (Rybix) with less than 25% of the days’ supply remaining on the previous claim. Denial criteria Paid Medicaid claim for Suboxone or Subutex in the past 60 days Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Trametinib (Mekinist) Tablets (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Mekinist Additional Criteria Quantity Limits Apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Tranexamic Acid (Lysteda) (Implemented 06/21/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Lysteda Additional criteria Quantity limits apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Transdermal Scopolamine Patches (Implemented 03/09/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria ≥ 12 years of age, OR History of at least one paid claim in the past 60 days for transderm scopolamine Additional criteria Quantity limits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Trazodone HCL (Oleptro ER 150mg & 300mg, Trazodone 300mg) Tablet (Implemented 07/09/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Oleptro ER 150mg, 300mg tablet Trazodone 300mg tablet Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Trientine HCl (Syprine) Capsule (Implemented 09/18/2013) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Syprine Additional Criteria Quantity Limits Apply Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Uloric Tablet (Febuxostat) (Implemented 08/17/2010) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that do not require prior authorization Allopurinol tablets Drugs that require manual review for prior authorization Uloric tablet Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Umeclidinium-Vilanterol Inhalation Powder (Anoro Ellipta) Prescribers may request an override for nonpreferred drugs by calling the UAMS College of Pharmacy Evidence-Based Prescription Drug Program Help Desk at 1-866-250-2518 (toll-free) or 501-526-4200 (local). Approval criteria Diagnosis of COPD in Medicaid history in previous 2 years; AND No therapeutic duplication with overlapping days’ supply between Anoro Ellipta, Spiriva, and/or Tudorza; AND Medicaid recipient is > 18 years of age; AND Denial criteria Lack of approval criteria; OR Diagnosis of asthma in Medicaid history in previous 2 years Additional criteria Quantity edits apply Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Valganciclovir Oral Solution (Valcyte) (Implemented 01/18/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Approval criteria Less than 9 years of age, OR History of diagnosis of NPO in Medicaid History Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Vemurafenib Tablet (Zelboraf) (Implemented 04/17//2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Zelboraf tablet Information required for the manual review process Confirmation of BRAFV600E mutation-positive melanoma as detected by an FDA-approved test In addition to requirement of diagnosis and lab results showing BRAFV600E mutation-positive melanoma, the following data will be required for the manual review process: o Baseline EKG and then every 3 months thereafter to monitor for QTc o Liver function tests baseline and then periodic o Baseline and periodic dermatology evaluation for squamous cell carcinomas Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Vismodegib Capsule (Erivedge) (Implemented 07/09/2012) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Erivedge capsule Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Vorapaxar (Zontivity) (Implemented 09/23/2014) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Zontivity Link to Memorandum Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Xgeva (Denosumab) (Implemented 01/21/2011) Prescribers may request an override by calling the HP Help Desk at 1-800707-3854 (toll-free) or 501-374-6609 ext. 500 (local). Drugs that require manual review for prior authorization Xgeva Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Xolair (Implemented 08/01/2003) Prescribers are required to fax a letter of medical necessity and include all supporting documentation for manual review to 501-683-4124. Link to Xolair statement of necessity—Microsoft Word format (.doc): https://www.medicaid.state.ar.us/Download/provider/forms/pharm/xolair.doc Link to Xolair statement of necessity—Portable Document Format (.pdf): https://www.medicaid.state.ar.us/Download/provider/forms/pharm/xolair.pdf Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Appendix A – Nil per os (NPO) ICD-9/Procedure codes B4034, B4035, B4036 B4149, B4150-B4156 B4160-B4162 43.11 46.32 96.07 97.01 432.46 432.60 437.52 437.6 437.61 438.3 438.32 440.15 443.72 443.73 494.4 Top of the document Description Enteral feeding supplies Enteral formula Enteral formula for pediatrics PEG PEJ tube Nasogastric tube insertion Nasogastric tube placement PEG placement PEG placement Naso/Oro-gastric tube placement Gastrostomy tube G-tube repositioning Gastrostomy tube Gastrostomy tube J-Tube J-Tube J-Tube PEG placement Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Appendix B – Approved tracheostomy codes Code V44.0 V55.0 31.1 31.2X 31.74 519.0X 31600 31601 31603 31605 31610 Description Tracheostomy status Attention to tracheostomy Temporary tracheostomy Permanent tracheostomy Revision of tracheostomy Tracheostomy complications Tracheostomy, planned (separate procedure); Tracheostomy, planned (separate procedure); younger than two years Tracheostomy, emergency procedure; transtracheal Tracheostomy, emergency procedure; cricothyroid membrane Tracheostomy, fenestration procedure with skin flaps Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Appendix C – Topical corticosteroids, no PA required Very high potency – No PA required POTENCY Very high PA STATUS Very high No PA Very high No PA CLOBETASOL PROPIONATE 0.05% GEL Very high No PA CLOBETASOL PROPIONATE 0.05% OINTMENT [TEMOVATE® OINTMENT] Very high No PA CLOBETASOL PROPIONATE 0.05% SOLUTION Very high No PA HALOBETASOL PROPIONATE 0.05% CREAM [ULTRAVATE®] Very high No PA HALOBETASOL PROPIONATE 0.05% OINTMENT [ULTRAVATE® OINTMENT] No PA DRUG NAME CLOBETASOL PROPIONATE 0.05% CREAM [tEMOVATE ® CREAM) CLOBETASOL PROPIONATE 0.05% EMOLLIENT CREAM [TEMOVATE EMOLLIENT® CREAM] Top of the document Topical Corticosteroid Criteria Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria High potency – No PA required POTENCY PA STATUS DRUG NAME High No PA BETAMETHASONE DIPROPIONATE 0.05% CREAM High No PA BETAMETHASONE DIPROPIONATE AUGMENTED 0.05% CREAM [DIPROLENE AF® CREAM] High No PA BETAMETHASONE DIPROPIONATE 0.05% LOTION High No PA BETAMETHASONE VALERATE 0.1% OINTMENT High No PA DESOXIMETASONE 0.25% CREAM [TOPICORT® CREAM] High No PA DIFLORASONE DIACETATE 0.05% CREAM High No PA FLUOCINONIDE 0.05% CREAM High No PA FLUOCINONIDE 0.05% EMOLLIENT CREAM High No PA FLUOCINONIDE 0.05% GEL High No PA FLUOCINONIDE 0.05% OINTMENT High No PA FLUOCINONIDE 0.05% SOLUTION High No PA TRIAMCINOLONE ACETONIDE 0.5% CREAM High No PA TRIAMCINOLONE ACETONIDE 0.5% OINTMENT Top of the document Topical Corticosteroid Criteria Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Medium potency – No PA required POTENCY PA STATUS DRUG NAME Medium No PA BETAMETHASONE VALERATE 0.1% CREAM Medium No PA BETAMETHASONE VALERATE 0.1% LOTION Medium No PA FLUTICASONE PROPIONATE 0.005% OINTMENT Medium No PA Medium No PA FLUTICASONE PROPIONATE 0.05% CREAM HYDROCORTISONE BUTYRATE 0.1% CREAM Medium No PA HYDROCORTISONE BUTYRATE 0.1% OINTMENT Medium No PA HYDROCORTISONE BUTYRATE 0.1% SOLUTION Medium No PA HYDROCORTISONE VALERATE 0.2% CREAM Medium No PA MOMETASONE FUROATE 0.1% CREAM Medium No PA MOMETASONE FUROATE 0.1% OINTMENT Medium No PA MOMETASONE FUROATE 0.1% SOLUTION Medium No PA TRIAMCINOLONE ACETONIDE 0.025% CREAM Medium No PA TRIAMCINOLONE ACETONIDE 0.025% LOTION Medium No PA TRIAMCINOLONE ACETONIDE 0.025% OINTMENT Medium No PA TRIAMCINOLONE ACETONIDE 0.1% CREAM Medium No PA TRIAMCINOLONE ACETONIDE 0.1% LOTION Medium No PA TRIAMCINOLONE ACETONIDE 0.1% OINTMENT Top of the document Topical Corticosteroid Criteria Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Low potency – No PA required POTENCY PA STATUS DRUG NAME Low No PA ALCLOMETASONE DIPROPIONATE 0.05% CREAM Low No PA ALCLOMETASONE DIPROPIONATE 0.05% OINTMENT Low No PA Low No PA Low No PA DESONIDE 0.05% OINTMENT FLUOCINOLONE ACETONIDE 0.01% OIL (DERMA-SMOOTHE FS®) HYDROCORTISONE 0.5% CREAM Low No PA HYDROCORTISONE 0.5% OINTMENT Low No PA HYDROCORTISONE 1% CREAM Low No PA HYDROCORTISONE 1% OINTMENT Low No PA HYDROCORTISONE 2.5% CREAM Low No PA HYDROCORTISONE 2.5% LOTION Low No PA HYDROCORTISONE 2.5% OINTMENT Low No PA HYDROCORTISONE ACETATE 0.5% CREAM Top of the document Topical Corticosteroid Criteria Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Appendix D – Congestive heart failure diagnoses ICD-9 402.01 402.11 402.91 404.01 404.03 404.11 404.13 404.91 404.92 404.93 428 Description Hypertensive heart disease with heart failure Hypertensive heart disease with heart failure Hypertensive heart disease with heart failure Hypertensive heart and renal disease with heart failure Hypertensive heart and renal disease with heart and renal failure Hypertensive heart and renal disease with heart failure Hypertensive heart and renal disease with heart and renal failure Hypertensive heart and renal disease with heart failure Hypertensive heart and renal disease with renal failure Hypertensive heart and renal disease with heart and renal failure Congestive heart failure, unspecified Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Appendix E – Malignant cancer diagnoses ICD-9 140 142 146 147 148 149 150 151 152 153 154 155 156 157 158 159 160 161 162 163 164 165 170 171 172 174 175 176 179 180 181 182 183 184 185 186 187 Description Malignant neoplasm of lip Malignant neoplasm of major salivary gland Malignant neoplasm of oropharynx Malignant neoplasm of nasopharynx Malignant neoplasm of hypopharynx Malignant neoplasm of other and ill-defined sites within the lip, oral cavity, and pharynx Malignant neoplasm of esophagus Malignant neoplasm of stomach Malignant neoplasm of small intestine including duodenum Malignant neoplasm of colon Malignant neoplasm of rectum rectosigmoid junction Malignant neoplasm of liver and intrahep Malignant neoplasm of gall bladder and extrahepatic bile duct Malignant neoplasm of pancreas Malignant neoplasm of retroperitoneum and peritoneum Malignant neoplasm of other and ill-defined sites within the digestive organs and peritoneum Malignant neoplasm of nasal cavities middle ear and accessory sinuses Malignant neoplasm of larynx Malignant neoplasm of trachea bronchus and lung Malignant neoplasm of pleura Malignant neoplasm of thymus, heart, and mediastinum Malignant neoplasm of other and ill-defined sites within the respiratory system and intrathoracic organs Malignant neoplasm of bone and articular cartilage Malignant neoplasm of connective and other soft tissue Malignant melanoma of skin Malignant neoplasm of female breast Malignant neoplasm of male breast Kaposis sarcoma Malignant neoplasm of uterus, part unspecified Malignant neoplasm of cervix uteri Malignant neoplasm of placenta Malignant neoplasm of body of uterus Malignant neoplasm of ovary and other uterine adnexa Malignant neoplasm other and unspecified female genital organs Malignant neoplasm of prostate Malignant neoplasm of testis Malignant neoplasm of penis and other male genital Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria ICD-9 188 189 190 191 192 193 194 195 196 197 198 199 200 201 202 203 204 205 206 207 208 Description Malignant neoplasm of bladder Malignant neoplasm of kidney and other and unspecified urinary organs Malignant neoplasm of eye Malignant neoplasm of brain Malignant neoplasm other and unspecified parts nervous system Malignant neoplasm of thyroid gland Malignant neoplasm of other endocrine glands and related structures Malignant neoplasm of other and ill-defined sites Secondary and unspecified malignant neoplasm of lymph Secondary malignant neoplasm of respiratory and digestive Secondary malignant neoplasm of other specified sites Malignant neoplasm without specification Lymphosarcoma and reticulosarcoma Hodgkins disease Other malignant neoplasms lymphoid and histiocytic tissue Multiple myeloma and immunoproliferative neoplasms Lymphoid leukemia Myeloid leukemia Monocytic leukemia Other specified leukemia Leukemia of unspecified cell type Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Appendix F – Antineoplastics to infer malignant cancer Antineoplastics Aldesleukin Alemtuzumab Altretamine Amifostine Arsenic Asparaginase Bcg Bevacizumab Bleomycin Busulfan Capecitabine Carboplatin Carmustine Cisplatin Clofarabine Cytarabine Dacarbazine Dactinomycin Daunorubicin Denileukin Docetaxel Doxorubicin Estramustine Etoposide Exemestane Floxuridine Fludarabine Fluorouracil Fulvestrant Gefitinib Top of the document Gemcitabine Gemtuzumab Idarubicin Ifosfamide Ifosfamide/mesna Imatinib Lomustine Mechlorethamine Mitomycin Mitoxantrone Oxaliplatin Paclitaxel Paclitaxel,semi-synthetic Pegaspargase Pemetrexed Pentostatin Plicamycin Procarbazine Rasburicase Rituximab Streptozocin Temozolomide Teniposide Thiotepa Topotecan Trastuzumab Vinblastine Vincristine Vinorelbine Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Appendix G – Chronic obstruction pulmonary disease diagnoses ICD-9 492 496 Description Emphysema Chronic airway obstruction, not elsewhere classified Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Appendix H – Approved diagnoses for nonpreferred antiepileptic agents in neuropathic pain agent class ICD-9 46.2 54.3 238.7 253.5 291.81 292 293.81 Description Subacute sclerosing panencephalitis Herpes simplex meningoencephalitis Other lymphatic and hematopoietic tissues Diabetes insipidus Alcohol withdrawal Drug withdrawal syndrome Psychotic disorder with delusions in conditions classified elsewhere Psychotic disorder with hallucinations in conditions classified 293.82 elsewhere 294.1XX Dementia in conditions classified elsewhere 295.XX Schizophrenic disorders 296.XX Affective psychoses (manic, depressive, bi-polar) Psychoses with origin specific to childhood (pervasive developmental 299.XX disorders) 300.01 Panic disorder 300.23 Social phobia 300.3 Obsessive compulsive disorder 300.4 Neurotic depression 301.12 Chronic depressive personality disorder 301.2 Schizoid personality disorder 301.4 Obsessive compulsive disorder 301.83 Borderline personality disorder 304.2 Cocaine dependence 306.1 Physiological malfunction arising from mental factors, respiratory 307.2X Tics 307.81 Tension headache 308.2 Predominant psychomotor disturbance 309.XX Adjustment reaction Unspecified nonpsychotic mental disorder following organic brain 310.9 damage 311 Depressive disorder 312.X Disturbance of conduct 313.XX Disturbance of emotions specific to childhood adolescence 317.XX Mild mental retardation 318.XX Other specified mental retardation 319.XX Unspecified mental retardation 327.51 Periodic limb movement disorder 330.1 Cerebral lipidoses 330.8 Rett’s disorder Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria ICD-9 Description 332.XX Parkinson’s disease 333.XX Other extrapyramidal disease and abnormal movement disorder 340 Multiple sclerosis 345.XX Epilepsy 346.XX Migraine 353.6 Phantom limb syndrome 359.2 Myotonic disorder 368.16 Psychophysical visual disturbance 388.3X Tinnitis 392.XX Rheumatic chorea 438.81 Apraxia 588.1 Nephrogenic diabetes insipidus 625.4 Premenstrual tension syndromes 696.1 Erythrodermic psoriasis 759.81 Prader-willi syndrome 759.89 Angelman syndrome 779.XX Other and ill defined conditions originating in the perinatal period 780.3X Convulsions, seizures 781.XX Abnormal involuntary movements 784.69 Symbolic dysfunction, unspecified, other 786.8 Hiccough 854.XX Intracranial injury V11.0 Schizophrenia V11.1 Affective disorders V11.3 Alcoholism V17.2 Other neurological diseases V18.4 Mental retardation V79.0 Depression V79.2 Mental retardation Top of the document Arkansas Medicaid Prescription Drug Program Prior Authorization Criteria Appendix I – Approved endoscopy codes Endoscopy CPT 43201 43234 43235 43236 43237 43238 43239 43240 43241 43242 43243 43244 43245 43246 43247 43248 43249 43250 43251 43255 43256 43257 43258 43259 Top of the document Procedure ESPHGSC RGD/FLX DIRED SBMCSL NJX ANY SBST UPR GI NDSC SMPL PRIM XM SPX UPR GI NDSC DX +-COLLJ SPEC BR/WA SPX UPR GI NDSC DIRED SBMCSL NJX ANY SBST UPR GI NDSC NDSC US XM LMTD ESOPH UPR GI NDSC TNDSC US FINE NDL ASPIR/BX ESOPH UPR GI NDSC BX 1/MLT UPR GI NDSC TRANSMURAL DRG PSEUDOCST UPR GI NDSC TNDSC INTRAL TUBE/CATH PLMT UPR GI NDSC TNDSC US FINE NDL ASPIR/BX W/US XM UPR GI NDSC NJX SCLEROSIS ESOPHGL&/GSTR VARC UPR GI NDSC BAND LIG ESOPHGL&/GSTR VARC UPR GI NDSC DILAT GSTR OUTLET FOR OBSTRCJ UPR GI NDSC DIRED PLMT PRQ GASTROSTOMY TUBE UPR GI NDSC RMVL FB UPR GI NDSC INSJ GD WIRE DILAT ESOPH GD WIRE UPR GI NDSC BALO DILAT ESOPH <30 MM DIAM UPR GI NDSC RMVL LES HOT BX/BIPOLAR CAUT UPR GI NDSC RMVL TUM POLYP/OTH LES SNARE TQ UPR GI NDSC CTRL BLD ANY METH UPR GI NDSC TNDSC STENT PLMT W/PREDILAT UPR GI NDSC DLVR THERMAL NRG SPHNCTR/CARDIA UPR GI NDSC ABLTJ LES X RMVL FORCEPS/CAUT/SNARE UPR GI NDSC W/US XM
© Copyright 2026